Abstract
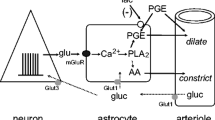
Glucose utilization (lCMRglc) increases linearly with spike frequency in neuropil but not perikarya of functionally activated neural tissues. Electrical stimulation, increased extracellular [K+] ([K+]0), or opening of Na+ channels with veratridine stimulates 1CMRglc in neural tissues; these increases are blocked by ouabain, an inhibitor of Na+,K+-ATPase. Stimulating Na+,K+-ATPase activity to restore ionic gradients degraded by enhanced spike activity appears to trigger these increases in lCMRglc. Cultured neurons behave similarly. Astrocytic processes that envelop synapses in neuropil probably contribute to the increased lCMRglc. lCMRglc in cultured astroglia is unaffected by elevated [K+]0 but is stimulated by increased intracellular [Na+] ([Na+]i), and this stimulation is blocked by ouabain or tetrodotoxin. L-Glutamate also stimulates lCMRglc in astroglia. This effect is unaffected by inhibitors of NMDA or non-NMDA receptors, blocked by ouabain, and absent in Na+-free medium; it appears to be mediated by increased [Na+]i due to combined uptake of Na+ with glutamate via Na+/glutamate co-transporters.
Similar content being viewed by others
REFERENCES
Sokoloff. L., Reivich, M., Kennedy, C., Des Rosiers, M. H., Patlak, C. S., Pettigrew, K. D., Sakurada, O., and Shinohara, M. 1977. The [14C]deoxyglucose method for the measurement of local cerebral glucose utilization: Theory, procedure, and normal values in the conscious and anesthetized albino rat. J. Neurochem. 28:897–916.
Sokoloff, L. 1981. Localization of functional activity in the central nervous system by measurement of glucose utilization with radioactive deoxyglucose. J. Cereb. Blood Flow Metab. 1:7–36.
Miyaoka, M., Shinohara, M., Batipps, M., Pettigrew, K. D., Kennedy, C., and Sokoloff, L. 1979. The relationship between the intensity of the stimulus and the metabolic response in the visual system of the rat. Acta. Neurol. Scand. 60(Suppl. 70):16–17.
Yarowsky, P., Kadekaro, M., and Sokoloff, L. 1983. Frequency-dependent activation of glucose utilization in the superior cervical ganglion by electrical stimulation of cervical sympathetic trunk. Proc. Natl. Acad. Sci., USA 80:4179–4183.
Kadekaro, M., Carne, A. M., and Sokoloff, L. 1985. Differential effects of electrical stimulation of sciatic nerve on metabolic activity in spinal cord and dorsal root ganglion in the rat. Proc. Natl. Acad. Sci., USA 82:6010–6013.
Shinohara, M., Dollinger, B., Brown, G., Rapoport, S., and Sokoloff, L. 1979. Cerebral glucose utilization: Local changes during and after recovery from spreading cortical depression. Science 203:188–190
Mata, M., Fink, D. J., Gainer, H., Smith, C. B., Davidsen, L., Savaki, H., Schwartz, W. J., and Sokoloff, L. 1980. Activity-dependent energy metabolism in rat posterior pituitary primarily reflects sodium pump activity. J. Neurochem. 34:213–215.
Kennedy, C., Des Rosiers, M. H., Sakurada, O., Shinohara, M., Reivich, M., Jehle, J. W., and Sokoloff, L. 1976. Metabolic mapping of the primary visual system of the monkey by means of the autoradiographic [14C]deoxyglucose technique. Proc. Natl. Acad. Sci., USA 73:4230–4234.
Nordmann, J. J. 1977. Ultrastructural morphometry of the rat neurohypophysis. J. Anat. 123:213–218.
Schwartz, W. J., Smith, C. B., Davidsen, L., Savaki, H., Sokoloff, L., Mata, M., Fink, D. J., and Gainer, H. 1979. Metabolic mapping of functional activity in the hypothalomo-neurohypophysial system of the rat. Science 205:723–725.
Smith, T. G, Jr. 1983. Sites of action potential generation in cultured neurons. Brain Res. 288:381–383.
Freygang, W. H. Jr. 1958. An analysis of extracellular potentials from single neurons in the lateral geniculate nucleus of the cat. J. Gen. Physiol. 41:543–564.
Freygang, W. H., Jr. and Frank, K. 1959. Extracellular potentials from single spinal motoneurones. J. Gen. Physiol. 42:749–760.
Orkand, R. K., Nicholls, J. G, and Kuffler, S. W. 1966. Effect of nerve impulses on the membrane potential of glial cells in the central nervous system of amphibia. J. Neurophysiol. 29:788–806.
Medzihradsky, F, Nandhasri, P. S., Idoyaga-Vargas, V, and Sellinger, O. Z. 1971. A comparison of ATPase activity of the glial cell fraction and the neuronal perikaryal fraction isolated in bulk from rat cerebral cortex. J. Neurochem. 18:1599–1603.
Henn, F. A., Haljamäe, H., and Hamberger, A. 1972. Glial cell function: active control of extracellular K+ concentration. Brain Res. 43:437–443.
Hertz, L. 1977. Drug-induced alterations of ion distribution at the cellular level of the central nervous system. Pharmacol. Rev. 29:35–65.
Erecinska, M. and Silver, I. A. 1994. Metabolism and role of glutamate in mammalian brain. Progress in Neurobiol. 43:37–71.
Yarowsky, P., Boyne, A. F., Wierwille, R., and Brookes, N. 1986. Effect of monensin on deoxyglucose uptake in cultured astrocytes: energy metabolism is coupled to sodium entry. J. Neurosci. 6:859–866.
Badar-Goffer, R. S., Ben-Yoseph, O., Bachelard, H. S., and Morris, P. G. 1992. Neuronal-glial metabolism under depolarizing conditions. A 13C-n.m.r. study. Biochem. J. 282:225–230.
Cummins, C. J., Glover, R. A., Sellinger, O. Z. 1979a. Neuronal cues regulate uptake in cultured astrocytes. Brain Res. 170:190–193.
Cummins, C. J., Glover, R. A., Sellinger, O. Z. 1979b. Astroglial uptake is modulated by extracellular K+. J. Neurochem. 33:779–785.
Brookes, N., Yarowsky, P. J. 1985. Determinants of deoxyglucose uptake in cultured astrocytes: the role of the sodium pump. J. Neurochem. 44:473–479.
Hertz, L., and Peng, L. 1992. Energy metabolism at the cellular level of the CNS. Can. J. Physiol. Pharmacol. 70(Suppl.):S145–S157.
Peng, L., Zhang, X., and Hertz, L. 1994. High extracellular potassium concentrations stimulate oxidative metabolism in a glutamatergic neuronal culture and glycolysis in cultured astrocytes but have no stimulatory effect in a GABAergic neuronal culture. Brain Res. 663:168–172.
Smith, C. B. 1983. Localization of activity-associated changes in metabolism of the central nervous system with the deoxyglucose method: Prospects for cellular resolution. Pages 269–317, in Barker, J. L., and McKelvy, J. F. (eds.), Current Methods in Cellular Neurobiology, Vol. I, Anatomical Techniques, John Wiley, New York.
Takahashi, S., Driscoll, B. F., Law, M. J., and Sokoloff, L. 1995. Role of sodium and potassium in regulation of glucose metabolism in cultured astroglia. Proc. Natl. Acad. Sci. USA 92:4616–4620.
Pellerin, L., and Magistretti, P. J. 1994. Glutamate uptake into astrocytes stimulates aerobic glycolysis: A mechanism coupling neuronal activity to glucose utilization. Proc. Natl. Acad. Sci. USA 91:10625–10629
Flott, B., and Seifert, W. 1991. Characterization of glutamate uptake in astrocyte primary cultures from rat brain. Glia 4:293–304
Magistretti, P. J., and Pellerin, L. 1996. Cellular bases of brain energy metabolism and their relevance to functional brain imaging: Evidence for a prominent role of astrocytes. Cerebral Cortex 6:50–61.
Tsacopoulos, M., and Magistretti, P. 1996. Metabolic coupling between glia and neurons. J. Neurosci. 16:877–885.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sokoloff, L. Energetics of Functional Activation in Neural Tissues. Neurochem Res 24, 321–329 (1999). https://doi.org/10.1023/A:1022534709672
Issue Date:
DOI: https://doi.org/10.1023/A:1022534709672