Abstract
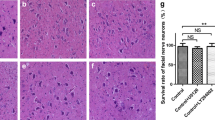
Immature rat facial motoneurons are very sensitive to injury with nearly 80% dying during the first week after axotomy. This motoneuron death is apoptotic, similar to that induced in neurons after tropic factor withdrawal. The diphenylpiperazines, flunarizine and cinnarizine, protect dorsal root ganglion neurons from death after withdrawal of trophic support, i.e., nerve growth factor withdrawal, in vitro. Similarly, the monoamine oxidase inhibitor, deprenyl, promotes survival of facial motoneurons after axotomy. These pharmacological agents were assessed both alone and in combination for their ability to prevent death in non-nerve growth factor dependent CNS motoneurons after facial nerve axotomy in newborn rats. Long-term experiments were done with the diphenylpiperazines to evaluate potential enhancement of regeneration. Facial nerve transection resulted in 78% neuronal loss in the injured compared with the contralateral, uninjured nucleus. Systemic administration of diphenylpiperazines for 1 week after facial nerve transection doubled the number of surviving motoneurons from 23% to 47%. Similar results were obtained with deprenyl. Combinations of diphenylpiperazines and deprenyl provide a similar degree of neuronal protection 1 week after injury as that obtained by either agent alone. We assessed the ability of diphenylpiperazines to protect facial motoneurons from death over a prolonged period and enhance subsequent regeneration. Motor neuron counts in rats treated with diphenylpiperazines for 1 month after injury and assessed 2 months later demonstrated long-term enhancement of neuronal protection with an increase of 45% in the number of horseradish peroxidase-labelled motoneurons. The diphenylpiperazines group had ∼80% more regenerated myelinated axons in the distal facial nerve than the control group. Thus, diphenylpiperazine treatment during the first month after injury provides long-term protection of non-nerve growth factor dependent CNS motoneurons with subsequent potentiation of long-term facial nerve regeneration.
Similar content being viewed by others
References
Altman, J. (1992) Programmed cell death: the paths to suicide. Trends in Neurosciences 15, 278–80.
Ansari, K. S., Yu, P. C., Kruck, P. A. & Tatton, W. G., (1993) Rescue of axotomized immature rat-facial motoneurons by R(–)-deprenyl: stereospecificity and independence from monamine oxidase inhibition. Journal of Neuroscience 13, 1043–52.
Carmignoto, G., Maffei, L., Candeo, P., Canella, R. & Comelli, C., (1989) Effect of NGF on the survival of rat retinal ganglion cells following optic nerve section. Journal of Neuroscience 9, 1263–72.
Contreras, M. L. & Guroff, G. (1987) Calcium-dependent, nerve growth factor-stimulated hydrolysis of phosphoinositides in PC-12 cells. Journal of Neurochemistry 48, 1466–72.
Crews, L. L. & Wigston, D. J. (1990) The dependence of motoneurons on their target muscle during postnatal development of the mouse. Journal of Neuroscience 10, 1643–50.
Eichler, M. & Rich, K. M. (1989) Death of sensory ganglion neurons after acute withdrawal of nerve growth factor in dissociated cell cultures. Brain Research 42, 340–6.
Eichler, M. Dubinsky, J. M., Tong, J. X. & Rich, K. M. (1994) The ability of diphenylpiperazines to prevent neuronal death in DRG neurons in vitro after NGF deprivation and in vivo after axotomy. Journal of Neurochemistry 62, 2148–57.
Eschweiler, G. W. & Bahr, M. (1993) Flunarizine enhances rat retinal ganglion cell survival after axotomy. Journal of the Neurological Sciences 116, 34–40.
Estus, S., Zaks, W. J., Freeman, R. S., Gruda, M., Bravo, R. & Johnson, E. M., Jr (1994) Altered gene expression in neurons during programmed cell death: identification of c-jun as necessary for neuronal apoptosis. Journal of Cell Biology 127, 1717–27.
Fink, D. W. Jr & Guroff, G. (1990) Nerve growth stimulation of arachidonic acid release from PC-12 cells: independence from phosphoinoside turnover. Journal of Neurochemistry 55, 1716–26.
Green, L. A. (1977a) Quantitative in vitro studies on the nerve growth factor (NGF) requirement of neurons. I. Sympathetic neurons. Developmental Biology 58, 96–105.
Green, L. A. (1977b) Quantitative in vitro studies on the nerve growth factor (NGF) requirement of neurons. II. Sensory neurons. Developmental Biology 58, 106–13.
Guha, A., Lau, N., Huvar, I., Gutmann, D., Provias, J. & Boss, G. (1996) Ras-GTP levels are elevated in human NF1 peripheral nerve tumors. Oncogene 12, 507–13.
Hamburger, V. (1934) The effects of wing bud extirpation on the development of the central nervous system in chick embryos. Journal of Experimental Zoology 68, 449–94.
Kharbanda, S., Saleem, A., Shafman, T., Emoto, Y. Taneja, M., Rubin, E., Weichselbaum, R., Woodgett, J., Avruch, J., Kyriakis, J. & Kufu, D. (1995) Ionizing radiation stimulates a Grb-2-mediated association of the stress-activated protein kinase with phosphatidylinositol 3-kinase. Journal of Biological Chemistry 270, 18871–4.
Kozak, A., Nikodijeveic, B., Yavin, E. & Guroff, G. (1992) Intracellular calcium levels regulate the actions of nerve growth factor on calcium uptake in PC-12 cells. Journal of Neuroscience Research 33, 30–6.
Laasberg, T., Philak, A., Neuman, T.& Saarma, M. (1988) Nerve growth increases the cyclic GMP level and activates the cyclic GMP phosphodiesterase in PC-12 cells. FEBS Letters 239, 367–70.
Lavallle, A. & Lavelle, F. W. (1958) The nucleolar apparatus and neuronal reactivity to injury during development. Journal of Experimental Zoology 137, 285–316.
Levi-Montalcini, R. (1987) Nerve growth factor: thirty five years later. The EMBO Journal 6, 1145–54.
Levi-Montalcini, R. & Angeletti, P. U. (1968) Nerve growth factor. Physiological Reviews 48, 534–69.
Lieberman, A. R. (1971) The axon reaction: a review of the principal features of perikaryal responses to axon injury. International Review of Neurobiology 14, 49–124.
Lieberman, A. L. (1974) Some factors affecting retrograde neuronal responses to axonal lesions. In Essays on the nervous system (edited by Bellaris, R. & Gray, E. G.) pp. 71–105. Oxford: Clarendon Press
Mesulam, M. M. & Mufson, E. J., (1980) The rapid anterograde transport of horseradish peroxidase. Neuroscience 5, 1277–86.
Mesulam, M. M., Hegarty, E., Barbas, H., Carson, K. A., Gower, E. C., Knapp, A. G., Moss, M. B. & Mufson, E. J. (1980) Additional factors influencing sensitivity in the tetramethyl benzidine method for HRP neurohistochemistry. Journal of Histochemistry and Cytochemistry 28, 1255–9.
Oppenheim, R. W. (1991) Cell death during development of the nervous system. Annual Review of Neuroscience 14, 453–501.
Oppenheim, R. W. (1996) Neurotrophic survival molecules for motoneurons: an embarrassment of riches. Neuron 17, 195–7.
Otto, D., Unsicker, K. & Grothe, C. (1987) Pharmacological effects of nerve growth factor and fibroblast growth factor applied to the transected sciatic nerve on neuron death in adult dorsal root ganglia. Neuroscience Letters 83, 156–60.
Pang, L., Zheng, C. F., Guan, K. L. & Saltiel, A. R. (1995) Nerve growth factor stimulates a novel protein kinase in PC-12 cells that phosphorylates and activates mitogen-activated protein kinase (MEK). Biochemistry 307, 513–19.
Pollin, M. M., McHanwell, S. & Slater, C. R. (1991) The effect of age on motor neuron death following axotomy in the mouse. Development 112, 83–90.
Ransom, S. W. (1906) Retrograde degeneration in the spinal nerves. Journal of Comparative Neurology 14, 265–93.
Rich, K. M. & Hollowell, J. P. (1990) Flunarizine protects neurons from death after axotomy or NGF deprivation. Science 248, 1419–21.
Rich, K. M., Luszczynski, J. R., Osborne, P. A. & Johnson, E. M. Jr (1987) Nerve growth factor protects adult sensory neurons from cell death and atrophy caused by nerve injury. Journal of Neurocytology 16, 261–8.
Romanes, G. J. (1946) Motor localization and the effects of nerve injury on the ventral horn cells of the spinal cord. Journal of Anatomy 80, 117–31.
Salo, P. T. & Tatton, W. G. (1992) Deprenyl reduces the death of motoneurons caused by axotomy. Journal of Neuroscience Research 31, 394–400.
Schubert, D. & Whitlock, C. (1977) Alteration of cellular adhesion by nerve growth factor. Proceedings of the National Academy of Science (USA) 74, 4055–8.
Schubert, D. Lecorbiere, M., Whitlock, C. & Stallcup, W. (1978) Alterations in the surface properties of cells responsive to nerve growth factor. Nature 273, 718–23
Seiler, S. M., Arnold, A. J. & Stanton, H. C. (1987) Inhibitors inositol triphosphate induced Ca2+ release from isolated platelet membrane vesicles. Biochemical Pharmacology 36, 3331–7.
Sendtner, M., Kreutzberg, G. W. & Thoenen, H. (1990) Ciliary neurotrophic factor prevents the degeneration of motor neurons after axotomy. Nature 345, 440–1.
Sendtner, M., Holtmann, B., Kolbeck, R., Thoenen, H. & Barde, Y-A. (1992) Brain-derived neurotrophic factor prevents the death of motoneurons in newborn rats after nerve section. Nature 360, 757–9.
Snider, W. D., Elliott, J. L. & Yan, Q. (1992) Axotomy-induced neuronal death during development. Journal of Neurobiology 23, 1231–46.
Tatton, W. G. & Greenwood, C. E. (1991) Rescue of dying neurons: a new action for deprenyl in MPTP Parkinsonism. Journal of Neuroscience Research 30, 666–72.
Thomas, P. G. & Verkeij, A. J. (1990) The dissimilar interactions of the calcium antagonist flunarizine with different phospholipid classes and molecular species: a differential scanning clorimetry study. Biochimica et Biophysica Acta 1030, 211–22.
Thomas, P. G., Zimmerman, A. G. & Verkeij, A. J. (1988) Prevention of calcium-induced membrane structural alterations in erythrocyte membranes by flunarizine. Biochimica et Biophyica Acta 946, 439–4.
Umemiya, M., Araki, I. & Kuno, M. (1993) Electrophysiological properties of axotomized facial motoneurons that are destined to die in neonatal rats. Journal of Physiology 462, 661–78.
Wu, R. M., Murphy, D. L. & Chiueh, C. C. (1996) Suppression of hydroxyl radical formation and protection of nigral neurons by 1-deprenyl (selegiline). Annals of the New York Academy of Sciences 786, 379–90.
Yan, O. Elliot, J. & Snider, W. D. (1992) Brain-derived neurotrophic factor rescues spinal motor neurons from axotomy-induced cell death. Nature 360, 753–5.
Yip, H. K., Rich, K. M., Lampe, P. A., & Johnson, E. M., Jr (1984) The effects of nerve growth factor or and its antiserum on the postnatal development and survival after injury of sensory neurons in the rat dorsal root ganglia. Neuroscience 4, 2986–92.
Yu, W. H. (1988) Sex difference in neuronal loss induced by axotomy in the rat brain stem motor nuclei. Experimental Neurology 102, 230–5.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tong, J.X., Rich, K.M. Diphenylpiperazines enhance regeneration after facial nerve injury. J Neurocytol 26, 339–347 (1997). https://doi.org/10.1023/A:1018508819191
Issue Date:
DOI: https://doi.org/10.1023/A:1018508819191