Abstract
Purpose: To assess the objective response rate, toxicityexperienced, progression-free survival, and overall survival ofpatients with previously untreated advanced soft tissue sarcomastreated with a liposomal doxorubicin formulation (Doxil).
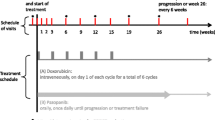
Methods: Patients with metastatic or recurrent soft tissuesarcoma who had received no prior chemotherapy for advanceddisease were treated with liposomal doxorubicin (Doxil) accordingto a two stage accrual design. Doxil was administered at 50mg/m2 every 4 weeks. A total of 15 patients were treated andare evaluable for response and toxicity.
Results: The male/female ratio was 7/8, the median age was60 years (34–75) and the ECOG performance status was 0-1 in>90% of patients. Leiomyosarcoma (7/15) and malignant fibroushistiocytoma (2/15) were the most common histologic diagnoses.No objective responses were observed in the 15 evaluablepatients. No lethal toxicity occurred. Grade 3–4 leukopenia orneutropenia were reported in 3/15 (20%) patients. Grade 3mucositis or hand-foot syndrome occurred in 2/15 (13%) and 1/15(7%) patients respectively and seemed more severe in olderpatients. The median time to progression was 1.9 months (range0.9–6.2). Twelve patients have now died. The Kaplan-Meierestimate of median overall survival is 12.3 months. As called forin the study design, accrual was terminated because no responseswere obtained in the first 15 patients.
Conclusion: Though well-tolerated, Doxil given accordingto this dose and schedule to patients with advanced soft tissuesarcoma had no significant therapeutic activity. A correlationbetween older age and skin/mucosal toxicity of Doxil is suggestedin this study but needs confirmation. Future investigations ofDoxil in soft tissue sarcomas should use a different schedule anddose.
Similar content being viewed by others
References
O'Bryan RM, Luce JK, Talley RW, Gottlieb JA, Baker LH, Bonadonna G: Phase II evaluation of adriamycin in human neoplasia. Cancer 32: 11–18, 1973
O'Bryan RM, Baker LH, Gottlieb JE, Rivkin SE, Balcerzak SP, Grumet GW, Salmon SE, Moon TE, Hoogstraten B: Dose response evaluation of adriamycin in human neoplasia. Cancer 39: 1940–1948, 1977
Benjamin RS, Wiernik PH, Bachur NR: Adriamycin chemotherapy-efficacy, safety, and pharmacologic basis of an intermittent single high-dosage schedule. Cancer 33: 19–27, 1974
Schoenfeld DA, Rosenbaum C, Horton J, Wolter JW, Falkson G, DeConti RC: A comparison of adriamycin versus vincristine and adriamycin, and cyclophosphamide versus vincristine, actinomycin-D, and cyclophosphamide for advanced sarcoma. Cancer 50: 2757–2762, 1982
Borden EC, Amato DA, Edmonson JK, Ritch PS, Shiraki M: Randomized comparison of doxorubicin and vindesine to doxorubicin for patients with metastatic soft-tissue sarcomas. Cancer 66: 862–867, 1990
Edmonson JH, Ryan LM, Blum RH, Brooks JS, Shiraki M, Frytak S, Parkinson DR: Randomized comparison of doxorubicin alone versus ifosfamide plus doxorubicin or mitomycin, doxorubicin, and cisplatin against advanced soft tissue sarcomas. J Clin Oncol 11: 1269–1275, 1993
Borden EC, Amato DA, Rosenbaum C, Enterline HT, Shiraki MJ, Creech RH, Lerner HJ, Carbone PP: Randomized comparison of three adriamycin regimens for metastatic soft tissue sarcomas. J Clin Oncol 5: 840–850, 1987
Baker LH, Frank J, Fine G, Balcerzalk SP, Stephens RL, Stuckey WJ, Rivkin S, Saiki J, Ward JH: Combination chemotherapy using adriamycin, DTIC, cyclophosphamide, and actinomycin D for advanced soft tissue sarcomas: a randomized comparative trial. A phase III, Southwest Oncology Group Study (7613). J Clin Oncol 5: 851–861, 1987
Mason M, Robinson W, Harmer C, Westbury G: Intra-arterial adriamycin, conventionally fractionated radiotherapy and conservative surgery for soft tissue sarcomas. Clin Oncol (R Coll Radiol) 4: 32–35, 1992
Didolkar MS, Kanter PW, Baffi RR, Schwartz HS, Lopez R: Comparison of regional versus systemic chemotherapy with adriamycin. Ann Surg 187: 332–336, 1978
Gabizon AA, Barenholz Y, Bialer M: Prolongation of the circulation time of doxorubicin encapsulated in liposomes containing a polyethylene glycol-derivatized phospholipid: pharmacokinetic studies in rodents and dogs. Pharm Res 10: 703–708, 1993
Gabizon A, Catane R, Uziely B, Kaufman B, Safra T, Cohen R, Martin F, Huang A, Barenhoiz Y: Prolonged circulation time and enhanced accumulation in malignant exudates of doxorubicin encapsulated in polyethylene-glycol coated liposomes. Cancer Res 54: 987–992, 1994
Northfelt DW, Martin FJ, Working P, Volberding PA, Russell J, Newman M, Amantea MA, Kaplan LD: Doxorubicin encapsulated in liposomes containing surface-bound polyethylene glycol: pharmacokinetics, tumor localization, and safety in patients with AIDS-related Kaposi's sarcoma. J Clin Pharmacol 36: 55–63, 1996
Alien TM, Hansen C, Martin F, Redemann C, Yau-Young A: Liposomes containing synthetic lipid derivatives of poly(ethylene glycol) show prolonged circulation half-lives in vivo. Biochim Biophys Acta 1066: 29–36, 1991
Hollander M, Wolfe D: Nonparametric Statistical Methods. New York, NY: John Wiley & Sons, Inc., 1973
Oudard S, Thierry A, Jorgensen TJ, Rahman A: Sensitization of multidrug-resistant colon cancer cells to doxorubicin encapsulated in liposomes. Cancer Chemother Pharmacol 28: 259–265, 1991
Thierry AR, Dritschilo A, Rahman A: Effect of liposomes on P-glycoprotein function in multidrug resistant cells. Biochem Biophys Res Commun 187: 1098–1105, 1992
Berry G, Billingham M, Alderman E, Richardson P, Torti F, Lum B, Patek A, Martin FJ: The use of cardiac biopsy to demonstrate reduced cardiotoxicity in AIDS Kaposi's sarcoma patients treated with pegylated liposomal doxorubicin. Ann Oncol 9: 711–716, 1998
Loehrer PJ Sr, Sledge GW Jr, Nicaise C, Usakewicz J, Hainsworth JW, Martelo OJ, Omura G, Braun TJ: Ifosfamide plus doxorubicin in metastatic adult sarcomas: a multiinstitutional phase II trial. J Clin Oncol 7: 1655–1659, 1989
Northfelt DW, Dezube BJ, Thommes JA, Miller BJ, Fischl MA, Friedman-Kien A, Kaplan LD, Du Mond C, Mamelok RD, Henry DH: Pegylated-liposomal doxorubicin versus doxorubicin, bleomycin, and vincristine in the treatment of AIDS-related Kaposi's sarcoma: results of a randomized phase III clinical trial. J Clin Oncol 16: 2445–2451, 1998
Uziely B, Jeffers S, Isacson R, Kutsch K, Wei-Tsao D, Yehoshua Z, Libson E, Muggia FM, Gabizon A: Liposomal doxorubicin: antitumor activity and unique toxicities during two complementary phase I studies. J Clin Oncol 13: 1777–1785, 1995
Stewart S, Jablonowski H, Goebel FD, Arasteh K, Spittle M, Rios A, Aboulafia D, Galleshaw J, Dezube BJ: Randomized comparative trial of pegylated liposomal doxorubicin versus bleomycin and vincristine in the treatment of AIDSrelated Kaposi's sarcoma. International Pegylated Liposomal Doxorubicin Study Group, J Clin Oncol 16: 683–691, 1998
Ranson MR, Carmichael J, O'Byrne K, Stewart S, Smith D, Howell A: Treatment of advanced breast cancer with sterically stabilized liposomal doxorubicin: results of a multicenter phase II trial [see comments]. J Clin Oncol 15: 3185–3191, 1997
Muggia FM, Hainsworth JD, Jeffers S, Miller R, Groshen S, Tan M, Roman L, Uziely B, Muderspach L, Garcia A, Burnett A, Greco FA, Morrow CP, Paradiso LJ, Liang LJ: Phase II study of liposomal doxorubicin in refractory ovarian cancer: antitumor activity and toxicity modification by liposomal encapsulation. J Clin Oncol 15: 987–993, 1997
Garcia AA, Kempf RA, Rogers M, Muggia FM: A phase II study of Doxil (liposomal doxorubicin): lack of activity in poor prognosis soft tissue sarcomas. Ann Oncol 9: 1131–1133, 1998
Skubitz K: A phase II trial of pegylated liposomal doxorubicin (Doxil) demonstrates activity in refractory sarcoma, mesothelioma, and head and neck cancer. Proc Am Soc Clin Oncol 18: 541a (abstract 2090), 1999
Toma S, Tucci A, Villani G, Carteni G, Palumbo R: Liposomal doxorubicin (CAELYX) in advanced pretreated soft tissue sarcomas (STS): a phase II trial of the Italian sarcoma group (ISG). Proc AmSoc Clin Oncol 18: 540a (abstract 2087), 1999
Judson I, Radford J, Blay J-Y, Van hoesel W, LeCesne A, van Oosterom A, Nielsen O, Camb C, Wall R, Harris M, Hermes C, Donate Di Paola E, Verweij J: A randomized Phase II trial of Caelyx/Doxil versus doxorubicin in advanced or metastatic soft tissue sarcomas (STS)-an EORTC soft tissue and bone sarcoma group (STBS) trial. Proc Am Soc Clin Oncol 18: 541a (abstract 2089), 1999
Casper ES, Schwartz GK, Sugarman A, Leung D, Brennan MF: Phase I trial of dose-intense liposome-encapsulated doxorubicin in patients with advanced sarcoma. J Clin Oncol 15: 2111–2117, 1997
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chidiac, T., Budd, G., Pelley, R. et al. Phase II Trial of Liposomal Doxorubicin (Doxil®) in Advanced Soft Tissue Sarcomas. Invest New Drugs 18, 253–259 (2000). https://doi.org/10.1023/A:1006429907449
Issue Date:
DOI: https://doi.org/10.1023/A:1006429907449