Abstract
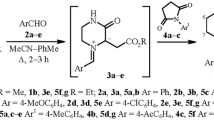
Iminiodichloromethanides generated by the reaction of O-acylsalicylaldehyde anils with dichlorocarbene undergo regioselective intramolecular 1,3-dipolar cycloaddition to the ester carbonyl group to give 2,5-epoxy-2,3,4,5-tetrahydro-1,4-benzoxazepin-2-ones. These compounds are expedient precursors for the synthesis of N-(2-hydroxybenzyl)ethanolamines.
Similar content being viewed by others
References
A. F. Khlebnikov, M. S. Novikov, and R. R. Kostikov, in Adv. Heterocycl. Chem., Ed. A. R. Katritzky, Academic, San Diego, 1996, 65, p. 93.
A. F. Khlebnikov and R. R. Kostikov, Izv. Akad. Nauk, Ser. Khim., 1993, 646 [Russ. Chem. Bull., 1993, 42, 603 (Engl. Transl.)].
R. R. Kostikov, A. F. Khlebnikov, and V. Ya. Bespalov, J. Phys. Org. Chem., 1993, 6, 83.
A. F. Khlebnikov, T. Yu. Nikiforova, and R. R. Kostikov, Zh. Org. Khim., 1996, 32, 746 [Russ. J. Org. Chem., 1996, 32, 715 (Engl. Transl.)].
A. F. Khlebnikov, M. S. Novikov, T. Yu. Nikiforova, and R. R. Kostikov, Zh. Org. Khim., 1999, 35, 98 [Russ. J. Org. Chem., 1999, 35, 91 (Engl. Transl.)].
A. F. Khlebnikov, M. S. Novikov, E. Yu. Kusei, Yu. Kopf, and R. R. Kostikov, Zh. Org. Khim., 2003, 39, 595 [Russ. J. Org. Chem., 2003, 39 (Engl. Transl.)].
M. S. Novikov, I. V. Voznyi, A. F. Khlebnikov, J. Kopf, and R. R. Kostikov, Chem. Soc., Perkin Trans. 1, 2002, 1628.
N. S. Kozlov, V. D. Pak, V. V. Mashevskii, and P. N. Plaksina, Khim.-farm. Zh., 1973, 7, 15 [Pharm. Chem. J., 1973, 7 (Engl. Transl.)].
M. S. Novikov, A. F. Khlebnikov, A. Krebs, and R. R. Kostikov, Eur. J. Org. Chem., 1998, 133.
Organic Synthesis, Ed. R. Adams, Wiley, New York, 1928, 8, 22.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Voznyi, I.V., Novikov, M.S., Khlebnikov, A.F. et al. Azomethine ylides derived from dichlorocarbene and O-acylsalicylaldehyde anils in the synthesis of 2,5-epoxy-2,3,4,5-tetrahydro-1,4-benzoxazepin-2-ones and 2-aminoethanols. Russian Chemical Bulletin 53, 1087–1091 (2004). https://doi.org/10.1023/B:RUCB.0000041304.68814.83
Issue Date:
DOI: https://doi.org/10.1023/B:RUCB.0000041304.68814.83