Abstract
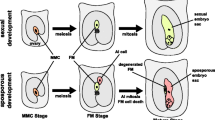
The isolation of genes associated with apomixis would improve understanding of the molecular mechanism of this mode of reproduction in plants as well as open the possibility of transfer of apomixis to sexual plants, enabling cloning of crops through seeds. Brachiaria brizantha is a highly apomictic grass species with 274 tetraploid apomicts accessions and only one diploid sexual. In this study we have compared gene expression in ovaries at megasporogenesis and megagametogenesis of sexual and apomictic accessions of B. brizantha by differential display (DD-PCR), with 60 primer combinations. Specificity of 65 cloned fragments, checked by reverse northern blot analysis, showed that 11 clones were differentially expressed, 6 in apomictic ovaries, 2 in sexual and 3 in apomictic and sexual, but at different stages. Of the 6 sequences isolated that were preferentially expressed in the apomictic accession: one sequence was from ovaries at megasporogenesis stage; three were from megagametogenesis stage; two were from both stages. Of the two sequences isolated from the sexual accessions, one showed expression in ovaries at megagametogenesis, while the other sequence was shown to be specific to both stages. Three sequences were from megasporogenesis stage in apomicts but were also detected at megagametogenesis in sexual plants. Sequence analysis showed that 5 of the 11 clones had no apparent homologues in the protein database. Some of the clones identified as apomictic-specific shared homology with known genes enabling their functional annotation. The relationships of these functions to the generation of the apomictic trait are discussed.
Similar content being viewed by others
References
Altschul, S.F., Gish, W., Miller, W., Myers, E.W. and Lipman, D.J. 1990. Basic local alignment search tool. J. Mol. Biol. 215: 403–410.
Altschul, S.F., Madden, T.L., Schaffer, A.A., Zhang, J., Zhang, Z., Miller, W. and Lipman, D.J. 1997. Gapped BLAST and PSIBLAST: a new generation of protein database search programs. Nucl. Acid Res. 25: 3389–3402.
Alves, E.R., Carneiro, V.T.C. and Araujo, A.C.G. 2001. Direct evidence of pseudogamy in an apomictic Brachiaria brizantha (Poaceae). Sex. Plant Reprod. 14: 207–212.
Araújo, A.C.G., Mukhambetzhanov, S., Pozzobon, M.T., Santana, E.F. and Carneiro, V.T.C. 2000. Female gametophyte development in apomictic and sexual Brachiaria brizantha (Poaceae). Rev. Cytol. Biol. Vég. Bot. 23: 13–28.
Aravind, L. and Koonin, E. 1999. Gleaning non-trivial structural, functional and evolutionary information about proteins by iterative database searches. J Mol Biol. 287: 1023–1040.
Aravind, L., Makarova, K.S. and Koonin, E.V. 2000. Holliday junction resolvases and related nucleases: identification of new families, phylogeny distribution and evolutionary trajectories. Nucl. Acids Res. 28: 3417–3432.
Aravind, L., Walker, D.R. and Koonin, E. 1999. Conserved domains in DNA repair proteins and evolution of repair systems. Nucl. Acids Res. 27: 1223–1242.
Bateman, A., Birney, E., Cerruti, L., Durbin, R., Etwiller, L., Eddy, S.R., Griffiths-Jones, S., Howe, K.L., Marshall, M. and Sonnhammer, E.L.L. 2002. The Pfam Protein Families Database. Nucl. Acids Res. 30: 276–280.
Bohl, F., Kruse, C., Frank, A., Ferring, D. and Jansen, R. 2000. Shep2, a novel RNA-binding protein, tethers ASH1 mRNA to the Myo4p myosin motor via She3p. EMBO J. 19: 5514–5524.
Borgnia, M., Nielsen, S., Engel, A. and Agre, P. 1999. Cellular and molecular biology of the aquaporin water channels. Annu. Rev. Biochem. 68: 425–458.
Bujnicki, J.M. 2000. Phylogeny of the restriction endonuclease-like superfamily inferred from comparison of protein structures. J. Mol. Evol. 50: 39–44.
Bujnicki, J.M., Elofsson, A., Fischer, D. and Rychlewski, L. 2001. Structure prediction meta server. Bioinformatics 17: 750–751.
Bullock, S.L. and Ish-Horowicz, D. 2001. Conserved signals and machinery for RNA transport in Drosophila oogenesis and embryogenesis. Nature 414: 611–616.
Callard, D., Lescure, B. and Mazzolini, L. 1994. A method for the elimination of false positives generated by the mRNA differential display technique. BioTechniques 16: 1096–1103.
Camerini-Otero, R.D. and Hsieh, P. 1995. Homologous recombination proteins in prokaryotes and eukaryotes. Annu. Rev. Genet. 29: 509–552.
Carman, J.G. 1997. Asynchronous expression of duplicate genes in angiosperms may cause apomixis, bispory, tetraspory, and polyembryony. Biol. J. Linnean Soc. 61: 51–94.
Chaudurhy, A.M., Ming, L., Miller, C., Craig, S., Dennis, E.S. and Peacock, W.J. 1997. Fertilization-independent seed development in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 94: 4223–4228.
Chen, L., Miyazaki, C., Kojima, A., Saito, A. and Adachi, T. 1999. Isolation and characterization of a gene expressed during early embryo sac development in apomictic guinea grass (Panicum maximum). J. Plant Physiol. 154: 55–62.
Deva, T. and Krishnaswamy, S. 2001. Structure-based sequence alignment of type-II restriction endonucleases. Biochim. Biophys. Acta 1544: 217–228.
Devos, D. and Valencia, A. 2000. Practical limits of function prediction. Proteins 41: 98–107.
Dingwall, C. and Laskey, R.A. 1991. Nuclear targeting sequences – a consensus? Trends Biochem. Sci. 16: 478–481.
Dusi, D.M.A. 2001. Apomixis in Brachiaria decumbens Stapf. Ph.D. Dissertation, University of Wageningen, Netherlands.
Dusi, D.M.A. and Willemse, M.T.M. 1999. Apomixis in Brachiaria decumbens Stapf.: gametophytic development and reproductive calendar. Acta Bot. Cracov. Ser. Bot. 41: 151–162.
Gobbe, J., Longly, B. and Louant, B.P. 1982. Calendrier des sporogénèse et gametogénèse femelles chez le diploïde et le tet-raploïde induit de Brachiaria rhuziziensis (Graminée). Can. J. Bot. 60: 2032–2037.
Hakman, I. and Oliviusson, P. 2002. High expression of putative aquaporin genes in cells with transporting and nutritive functions during seed development in Norway spruce (Picea abies). J. Exp. Bot. 53: 639–649.
Hanna, W.W. and Bashaw, E.C. 1987. Apomixis: its identification and use in plant breeding. Crop Sci. 27: 1136–1139.
Harvengt, P., Vlerick, A., Fuks, B., Wattiez, R., Ruysschaert, J.M. and Homble, F. 2000. Lentil seed aquaporins form a heterooligomer which is phosphorylated by a Mg(2+)-dependent and Ca(2+)-regulated kinase. Biochem J. 15: 183–190.
Hicks, J.L., Deng, W.M., Rogat, A.D., Miller, K.G. and Bownes, M. 1999. Class VI unconventional myosin is required for spermatogenesis in Drosophila. Mol. Biol. Cell 10: 4341–4353.
Kuhn, E. 2001. From library screening to microarray technology: strategies to determine gene expression profiles and to identify differentially regulated genes in plants. Ann. Bot. 87: 139–155.
Leblanc, O., Armstead, I., Pessino, S., Ortiz, J.P.A., Evans, C., Do Valle, C.B. and Hayward, M.D. 1997. Non-radioactive mRNA fingerprinting to visualise gene expression in mature ovaries of Brachiaria hybrids derived from B. brizantha, an apomictic tropical forage. Plant Sci. 126: 49–58.
Letunic, I., Goodstadt, L., Dickens, N.J., Doerks, T., Schultz, J., Mott, R., Ciccarelli, F., Copley, R.R., Ponting, C.P. and Bork, P. 2002. Recent improvements to the SMART domain-based sequence annotation resource. Nucl. Acids Res. 30: 242–244.
Liang, P. and Pardee, A.B. 1992. Differential display of eukaryotic messenger RNA by means of the polymerase chain reaction. Science 257: 967–971.
Lundstrom, J., Rychlewski, L., Bujnicki, J. and Elofsson, A. 2001. Pcons: a neural-network-based consensus predictor that improves fold recognition. Prot. Sci. 10: 2354–2362.
Luo, M., Bilodeau, P., Koltunow, A., Dennis, E.S., Peacock, W.J. and Chaudhury, A.M. 1999. Genes controlling fertilizationindependent seed development in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 96: 296–301.
Lutts, S., Ndikumana, J. and Louant, B.P. 1994. Male and female sporogenesis and gametogenesis in apomitic Brachiaria brizantha, Brachiaria decumbens and F1 hybrids with sexual colchicine induced tetraploid Brachiaria ruziziensis. Euphytica 78: 19–25.
Makarova, K.S., Aravind, L., Grishin, N.V., Rogozin, I.B. and Koonin, E.V. 2002. A DNA repair system specific for thermophilic Archaea and bacteria predicted by genomic context analysis. Nucl. Acids Res. 30: 482–496.
Marchler-Bauer, A., Panchenko, A.R., Shoemaker, B.A., Thiessen, P.A., Geer, L.Y. and Bryant, S.H. 2002. CDD: a database of conserved domain alignments with links to domain threedimensional structure. Nucl. Acids Res. 30: 281–283.
Martinez, E.J., Quarin, C.L. and Hayward, M.D. 1999. Genetic control of apospory in apomictic Paspalum species. Cytologia 64: 425–433.
Miles, J.W., Maass, B.L. and do Valle, C.B. 1996. Brachiaria: Biology, Agronomy and Improvement. CIAT Publishers, Cali, Colombia.
Mulder, N.J., Apweiler, R., Attwood, T.K., Bairoch, A., Barrell, D., Bateman, A., Binns, D., Biswas, M., Bradley, P., Bork, P., Bucher, P., Copley, R.R., Courcelle, E., Das, U., Durbin, R., Falquet, L., Fleischmann, W., Griffiths-Jones, S., Haft, D., Harte, N., Hulo, N., Kahn, D., Kanapin, A., Krestyaninova, M., Lopez, R., Letunic, I., Lonsdale, D., Silventoinen, V., Orchard, S.E., Pagni, M., Peyruc, D., Ponting, C.P., Selengut, J.D., Servant, F., Sigrist, C.J.A., Vaughan, R. and Zdobnov, E.M. 2003. The InterPro Database 2003 brings increased coverage and new features. Nucl. Acids Res. 31: 315–318.
Ngendahayo, M. 1988. Méchanismes de la réproduction dans le genre Brachiaria. Ph.D. Dissertation, Université Catholique de Louvain, Louvain, Belgium.
Nogler, G.A. 1984. Gametophytic apomixis. In: B.M. Johri (Ed.) Embryology of Angiosperms, Springer-Verlag, Berlin, pp. 475–518.
Pearson, W.R. and Lipman, D.J. 1998. Improved tools for biological sequence comparison. Proc. Natl. Acad. Sci. USA 85: 2444–2448.
Pessino, S.C., Espinoza, F., Martinez, E.J., Ortiz, J.P., Valle, E.M. and Quarin, C.L. 2001. Isolation of cDNA clones differentially expressed in flowers of apomictic and sexual Paspalum notatum. Hereditas 134: 35–42.
Rigden, D.J., Setlow, P., Setlow, B., Bagyan, I., Stein, R.A. and Jedrzejas, M.J. 2002. PrfA protein of Bacillus species: prediction and demonstration of endonuclease activity on DNA. Prot. Sci. 11: 2370–2381.
Sambrook, J., Fritsch, E.F. and Maniatis, T. 1989. Molecular Cloning: A Laboratory Manual, 2nd ed. Cold Spring Harbor Laboratory Press, Plainville, NY.
Savidan, Y. 1981. Genetics and utilization of apomixis for the improvement of Guinea grass (Pannicum maximum Jacq.). In: J. Smith and V.W. Hays (Eds.) Proceedings XIV International Grass Congress (15–24 June 1981, Lexington, KY). Westview Press, Boulder, CO, pp. 182–184.
Servant, F., Bru, C., Carrère, S., Courcelle, E., Gouzy, J., Peyruc, D., and Kahn, D. 2002. ProDom: automated clustering of homologous domains. Briefings Bioinformatics 3: 246–251.
Sherwood, R.T., Berg, C.C. and Young, B.A. 1994. Inheritance of apospory in bufflegrass. Crop Sci. 34: 1490–1494.
Tan, L., Chang, J.S., Costa, A. and Schedl, P. 2001. An autoregulatory feedback loop directs the localized expression of the Drosophila CPEB protein Orb in the developing oocyte. Development 128: 1159–1169.
Tucker, M.R., Araujo, A.C.G., Paech, N.A., Hecht, V., Schmidt, E.D.L., Rossel, J.B., de Vries, S.C. and Koltunow, A.M.G. 2003. Sexual and apomictic reproduction in Hieracium subgenus Pilosella are closely interrelated developmental pathways. Plant Cell 5: 1524–1537.
Valle, C.B. do 1986. Cytology, mode of reproduction and forage quality of selected species of Brachiaria Griseb. Ph.D. dissertation, University of Illinois, Urbana, Champaign, IL.
Valle, C.B. do., Glienke, C. and Leguisamon, G.O.C. 1994. Inheritance of apomixis in Brachiaria, a tropical forage grass. Apomixis Newsl. 7: 42–43.
Valle, C.B. do. and Savidan, Y. 1996. Genetics, cytogenetics and reproductive biology of Brachiaria. In: J.W. Miles, B.L. Maass and C.B. do Valle (Eds.) Brachiaria: Biology, Agronomy and Improvement, CIAT Publishers, Cali, Colombia, pp. 147–163.
Vielle-Calzada, J.-P., Nuccio, M.L., Budiman, M.A., Thomas, T.L., Burson, B.L., Hussey, M.A. and Wing, R.A. 1996. Comparative gene expression in sexual and apomictic ovaries of Pennisetum ciliari (L.) Link. Plant Mol Biol. 32: 1085–1092.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rodrigues, J.C., Cabral, G.B., Dusi, D.M. et al. Identification of differentially expressed cDNA sequences in ovaries of sexual and apomictic plants of Brachiaria brizantha . Plant Mol Biol 53, 745–757 (2003). https://doi.org/10.1023/B:PLAN.0000023664.21910.bd
Issue Date:
DOI: https://doi.org/10.1023/B:PLAN.0000023664.21910.bd