Abstract
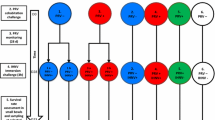
Infectious hematopoietic necrosis virus (IHNV) is one of the most significant viral pathogens of salmonids and is a leading cause of death among cultured juvenile fish. Although several vaccine strategies have been developed, some of which are highly protective, the delivery systems are still too costly for general use by the aquaculture industry. More cost effective methods could come from the identification of genes associated with IHNV resistance for use in selective breeding. Further, identification of susceptibility genes may lead to an improved understanding of viral pathogenesis and may therefore aid in the development of preventive and therapeutic measures. Genes of the major histocompatibility complex (MHC), involved in the primary recognition of foreign pathogens in the acquired immune response, are associated with resistance to a variety of diseases in vertebrate organisms. We conducted a preliminary analysis of MHC disease association in which an aquaculture strain of Atlantic salmon was challenged with IHNV at three different doses and individual fish were genotyped at three MHC loci using denaturing gradient gel electrophoresis (PCR-DGGE), followed by sequencing of all differentiated alleles. Nine to fourteen alleles per exon-locus were resolved, and alleles potentially associated with resistance or susceptibility were identified. One allele (Sasa-B-04) from a potentially non-classical class I locus was highly associated with resistance to infectious hematopoietic necrosis (p < 0.01). This information can be used to design crosses of specific haplotypes for family analysis of disease associations.
Article PDF
Similar content being viewed by others
References
Aoyagi, K., J.M. Kijkstra, C. Xia, I. Denda, M. Ototake, K. Hashimoto & T. Nakanishi. 2002. Classical MHC class I genes composed of highly divergent sequence lineages share a single locus in rainbow trout (Oncorhynchus mykiss). J. Immunol. 168: 260–273.
Apanius, V., D. Penn, P. Slev, L.R. Ruff & W.K. Potts. 1997. The nature of selection on the major histocompatibility complex. Crit. Rev. Immunol. 17: 179–224.
Balfry, S.K., A.G. Maule & G.K. Iwama. 2001. Coho salmon Oncorhynchus kisutch strain differences in disease resistance and non-specific immunity, following immersion challenges with Vibrio anguillarum. Dis. Aquat. Org. 47: 39–48.
Beacham, T.D. & T.P.T Evelyn. 1992a. Population and genetic variation in resistance of chinook salmon to vibriosis, furunculosis, and bacterial kidney disease. J. Aquat. Anim. Health 4: 153–167.
Beacham, T.D. & T.P.T. Evelyn. 1992b. Genetic variation in disease resistance and growth to chinook, coho, and chum salmon with respect to vibriosis, furunculosis and bacterial kidney disease. Trans. Am. Fish. Soc. 121: 456–485.
Bengtsson, B.O. & G. Thomson. 1981. Measuring the strength of associations between HLA antigens and diseases. Tissue Antigens 18: 356–363.
Bootland, L.M. & Leong, J.C. 1999. Infectious hematopoietic necrosis virus. pp. 57–121. In: P.T.K. Woo & D.W. Bruno (ed.) Fish Diseases and Disorders, Vol. 3: Viral, Bacterial and Fungal Infections, CAB International, New York, NY.
Brodsky, F.M., L. Lem, A. Solache & E.M. Bennett. 1999. Human pathogen subversion of antigen presentation. Immunol. Rev. 168: 199–215.
Grimholt, U., I. Olsaker, C. de Vries Lindstrom & O. Lie. 1994. A study of variability in the MHC class II ?1 and class I ?2 domain exons of Atlantic salmon (Salmo salar). Anim. Genet. 25: 147–153.
Grimholt, U., F. Drabløs, S.M. Jøorgensen, B. Høyheim & R.J.M. Stet. 2003. The major histocompatibility class I locus in Atlantic salmon (Salmo salar L.): polymorphism, linkage analysis and protein modelling. Immunogenetics 54: 570–581.
Hill, A.V.S. 1991. HLA associations with malaria in Africa: Some implications for MHC evolution. NATO ASI Ser, Vol. H 59. Molecular Evolution of the Major Histocompatibility Complex, Springer-Verlag, Berlin, Heidelberg.
Kaufman, J. & J. Salomonsen. 1997. The 'minimal essential MHC' revisited: Both peptide-binding and cell surface expression level of MHC molecules are polymorphisms selected by pathogens in chickens. Hereditas 127: 67–73.
Kaufman, J., H. Völk & H.J. Wallny. 1995. A 'Minimal essential MHC' and an 'unrecognized MHC': Two extremes in selection for polymorphism. Immunol. Rev. 143: 63–88.
Kent, M.L., G.S. Traxler, D. Kieser, J. Richard, S.C. Dawe, R.W. Shaw, G. Prosperi-Porta, J. Ketcheson & T.P.T. Evelyn. 1998. Survey of salmonid pathogens in ocean-caught fishes in British Columbia, Canada. J. Aquat. Anim. Health 10: 211–219.
Kono, T., M. Sakai & S. LaPatra. 2000. Expressed sequence tag analysis of kidney and gill tissues from rainbow trout (Oncorhynchus mykiss) infected with infectious hematopoietic necrosis virus. Mar. Biotech. 2: 493–498.
Kruglyak, L. 1999. Prospects for whole-genome linkage disequilibrium mapping of common disease genes. Nat. Genet. 22: 139–144.
Lamont, S.J. 1998. The chicken major histocompatibility complex and disease. Rev. Sci. Tech. Off. Int. Epizoot. 17: 128–142.
Malaga-Trillo E., Z. Zaleska-Rutczynska, B. McAndrew, V. Vincek, F. Figueroa, H. Sultmann & J. Klein. 1998. Linkage relationships and haplotype polymorphism among cichlid MHC class II B loci. Genetics 149: 1527–1537
Miller, K.M. & R.E. Withler. 1996. Sequence analysis of a polymorphic MHC class II gene in Pacific salmon. Immunogenetics 43: 337–351.
Miller K.M. & R.E. Withler 1998. The salmonid class I MHC: Limited diversity in a primitive teleost. Immunol. Rev. 166: 279–293
Miller, K.M., T.J. Ming, A.D. Schulze & R.E. Withler. 1999. Denaturing gradient gel electrophoresis (DGGE): A rapid and sensitive technique to screen nucleotide sequence variation in populations. BioTechniques 27: 1016–1030.
Miller, K.M., K.H. Kaukinen & A.D. Schulze. 2002. Expansion and contraction of major histocompatibility complex genes: A teleostean example. Immunogenetics 53: 941–963.
Murray B.W., P. Nilsson, Z. Zaleska-Rutczynska, H. Syltmann & J. Klein. 2000. Linkage relationships and haplotype variation of the major histocompatibility complex class I A genes in the cichlid fish Oreochromis niloticus. Mar. Biotechnol. 2: 437–448
Nei, M. & A.L. Hughes. 1991. Polymorphism and evolution of the major histocompatibility complex loci in mammals. pp. 222–247. In: R.K. Selander, A.G. Clark and T.S. Whittam (ed.) Evolution at the Molecular Level, Sinauer Associates Inc., Sunderland.
Palti, Y., K.M. Nichols, K.I. Waller, J.E. Parsons & G.H. Thorgaard. 2001. Association between DNA polymorphisms tightly linked to MHC class II genes and IHN virus resistance in backcrosses of rainbow and cut-throat trout. Aquaculture 194: 283–289.
Parham, P. 1994. The rise and fall of great class I genes. Seminars Immunol. 6: 373–382.
Parsons, J.E., R.A. Busch, G.H. Thorgaard & P.D. Scheerer. 1986. Increased resistance of triploid rainbow trout × coho salmon hybrids to infectious hematopoietic necrosis virus. Aquaculture 57: 337–343.
Phillips, R.B., A. Zimmerman, M.A. Noakes, Y. Palti, M.R. Morasch, L. Eiben, S.S. Ristow, G.H. Thorgaard & J.D. Hansen. 2003. Physical and genetic mapping of the rainbow trout major histocompatibility regions: evidence for duplication of the class I region. Immunogenetics 55(8): 561–569.
Potts, W.K. & P.R. Slev. 1995. Pathogen-based models favoring MHC genetic diversity. Immunol. Rev. 143: 181–197.
Raymond, M. & F. Rousset. 1995. GENEPOP (version 1.2): Population genetics software for exact tests and ecumenicism. J. Hered. 86: 248–249.
Sammut, B., L. Du Pasquier, P, Ducoroy, V, Laurens, A, Marcuz & A. Tournefier. 1999. Axolotl MHC architecture and polymorphism. Eur. J. Immunol. 29: 2897–2907.
Shum, B.P., R. Rajalingam, K.E. Magor, K. Azumi, W.H. Carr, B. Dixon, R.J.M. Stet, M.A. Adkison, R.P. Hedrick & P. Parham. 1999. A divergent non-classical class I gene conserved in salmonids. Immunogenetics 49: 479–490.
Shum, B.P., L. Guethlein, L.R. Flodin, M.A. Adkison, R.P. Hedrick, R.B. Nehring, R.J.M. Stet, C. Secombes & P. Parham. 2001. Modes of salmonid MHC class I and II evolution differ from the primate paradigm. J. Immunol. 166: 3297–3308.
Shum, B.P., P.M. Mason, K.E. Magor, L.R. Flodin, R.J.M. Stet & P. Parham. 2002. Structures of two major histocompatibility complex class I genes of rainbow trout (Oncorhynchus mykiss). Immunogenetics 54: 193–199.
Sidney, J., H.M. Grey, R.T. Kubo & A. Sette. 1996. Practical, biochemical and evolutionary implications of the discovery of HLA class I supermotifs. Immunol. Today 17: 261–266.
Svejgaard, A., C. Jersild, L.S. Nielson & W.F. Bodmer. 1974. HLA antigens and disease. Statistical and genetical considerations. Tissue Antigens 4: 95–105.
Svejgaard, A. & P. Ryder. 1994. HLA and disease associations: Detecting the strongest associations. Tissue Antigens 43: 18–27.
Takahata, N. 1995. MHC diversity and selection. Immunol. Rev. 143: 225–247.
Timon M., G. Elgar, S. Habu, K. Okumura & P.C.L Beverley. 1998. Molecular cloning of major histocompatibility complex class I cDNAs from the pufferfish Fugu rubripes. Immunogenetics 47: 170–173.
Traxler, G.S., J.R. Roome, K.A. Lauda & S. LaPatra. 1997. Appearance of infectious hematopoietic necrosis virus (IHNV) and neutralizing antibodies in sockeye salmon Oncorhynchus nerka during their migration and maturation period. Dis. Aquat. Org. 28: 31–38.
Winton, J.R. 1991. Recent advances in detection and control of infectious hematopoietic necrosis virus. Ann. Rev. Fish. Dis. 1: 83–93.
Winton, J.R. 1997. Immunization with viral antigens: Infectious hematopoietic necrosis. Dev. Biol. Standard. 90: 211–220.
Winton, J.R. 1998. Molecular approaches to fish vaccines. J. Appl. Ichthyol. 14: 153–158.
Withler, R.E. & T.P.T. Evelyn. 1990. Genetic variation in resistance to bacterial kidney disease within and between two strains of coho salmon from British Columbia. Trans. Am. Fish. Soc. 119: 1003–1009.
Wolf, K. 1988. Infectious hematopoietic necrosis. pp. 83–114. In: K. Wolf (ed.), Fish Viruses and Fish Viral Diseases, Cornell University Press, Ithaca, NY.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Miller, K.M., Winton, J.R., Schulze, A.D. et al. Major Histocompatibility Complex Loci are Associated with Susceptibility of Atlantic Salmon to Infectious Hematopoietic Necrosis Virus. Environmental Biology of Fishes 69, 307–316 (2004). https://doi.org/10.1023/B:EBFI.0000022874.48341.0f
Issue Date:
DOI: https://doi.org/10.1023/B:EBFI.0000022874.48341.0f