Abstract
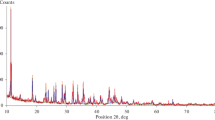
The thermal behaviour of halloysite fully expanded with hydrazine-hydrate has been investigated in nitrogen atmosphere under dynamic heating and at a constant, pre-set decomposition rate of 0.15 mg min-1. Under controlled-rate thermal analysis (CRTA) conditions it was possible to resolve the closely overlapping decomposition stages and to distinguish between adsorbed and bonded reagent. Three types of bonded reagent could be identified. The loosely bonded reagent amounting to 0.20 mol hydrazine-hydrate per mol inner surface hydroxyl is connected to the internal and external surfaces of the expanded mineral and is present as a space filler between the sheets of the delaminated mineral. The strongly bonded (intercalated) hydrazine-hydrate is connected to the kaolinite inner surface OH groups by the formation of hydrogen bonds. Based on the thermoanalytical results two different types of bonded reagent could be distinguished in the complex. Type 1 reagent (approx. 0.06 mol hydrazine-hydrate/mol inner surface OH) is liberated between 77 and 103°C. Type 2 reagent is lost between 103 and 227°C, corresponding to a quantity of 0.36 mol hydrazine/mol inner surface OH. When heating the complex to 77°C under CRTA conditions a new reflection appears in the XRD pattern with a d-value of 9.6 Å, in addition to the 10.2 Ĺ reflection. This new reflection disappears in contact with moist air and the complex re-expands to the original d-value of 10.2 Å in a few h. The appearance of the 9.6 Å reflection is interpreted as the expansion of kaolinite with hydrazine alone, while the 10.2 Å one is due to expansion with hydrazine-hydrate. FTIR (DRIFT) spectroscopic results showed that the treated mineral after intercalation/deintercalation and heat treatment to 300°C is slightly more ordered than the original (untreated) clay.
Similar content being viewed by others
References
R. A. Eggleton and D. B. Tilley, Clays Clay Miner., 46 (1998) 400.
B. Singh, Clays Clay Miner., 44 (1996) 191.
S. Komarneni, C. A. Fyfe and G. J. Kennedy, Clay Miner., 20 (1985) 327.
S. Yariv and S. Shoval, Clays Clay Miner., 23 (1985) 473.
B. Singh and I. D. R. Mackinnon, Clays Clay Miner., 44 (1996) 825.
J. H. Kirkman, Clays Clay Miner., 29 (1981) 1.
C. A. Hess and V. R. Saunders, J. Phys. Chem., 96 (1992) 4367.
D. R. Collins and C. R. A. Catlow, Acta Crystallogr., B47 (1991) 678.
R. L. Ledoux and J. L. White, J. Coll. Int. Sci., 21 (1966) 127.
C. T. Johnston and D. A. Stone, Clays Clay Miner., 38 (1990) 121.
R. L. Frost, J. T. Kloprogge, J. Kristóf and E. Horváth, Clays Clay Miner., 47 (1999) 732.
J. Kristóf, R. L. Frost, J. T. Kloprogge, E. Horváth and M. Gábor, J. Therm. Anal. Cal., 56 (1999) 885.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Horváth, E., Kristóf, J., Frost, R.L. et al. Hydrazine-hydrate intercalated halloysite under controlled-rate thermal analysis conditions. Journal of Thermal Analysis and Calorimetry 71, 707–714 (2003). https://doi.org/10.1023/A:1023301504396
Issue Date:
DOI: https://doi.org/10.1023/A:1023301504396