Abstract
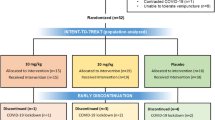
The objective of this study was to determine if an intravenous infusion of synthetic human secretin improves language and behavioral symptoms in children with autism. Forty-two children with the diagnosis of autism were randomized to one of two groups in this double-blind cross-over trial. One group received 2 IU/kg of intravenous synthetic human secretin at the first visit, followed by an equal volume of intravenous saline placebo at week 6. The other group received treatments in the reverse order. All children were evaluated at weeks 1, 3, 6, 9, and 12 with standardized assessments of language, behavior, and autism symptomatology. There were no significant differences in the mean scores on any measure of language, behavior, or autism symptom severity after treatment with secretin compared to treatment with placebo. The results of this study do not support secretin as a treatment for autism.
Similar content being viewed by others
REFERENCES
Aman, M., & Singh, M. (1994). Abberrant Behavior Checklist–Community. East Aurora, NY: Slosson Educational Publications, Inc.
American Psychiatric Association (1994). Diagnostic and Statistical Manual of Mental Disorders (4th ed). Washington, D. C.: Author.
Bristol-Power, M. M., & Spinella, G. (1999). Research on screening and diagnosis in autism: A work in progress. Journal of Autism and Developmental Disorders, 29(6), 435–438.
Bryson, S. E. (1996). Brief report: Epidemiology of autism. Journal of Autism and Developmental Disorders, 26, 165–167.
Chez, M. G., Buchanan, C. P., Bagan, B. T., Hammer, M. S., Mc-Carthy, K. S., Ovrutskaya, I., et al. (2000). Secretin and autism: A two-part clinical investigation [In Process Citation]. Journal of Autism and Developmental Disorders, 30(2), 87–94.
Coniglio, S. J., Lewis, J. D., Lang, C., Burns, T. G., Subhani-Siddique, R., Weintraub, A., et al. (2001). A randomized, double-blind, placebo-controlled trial of single-dose intravenous secretin as treatment for children with autism. Journal of Pediatrics, 138(5), 649–655.
Dunn, L., & Dunn, L. (1997). Peabody Picture Vocabulary Test. Circle Pines, MN: American Guidance Service.
Dunn-Geier, J., Ho, H. H., Auersperg, E., Doyle, D., Eaves, L., Matsuba, C., et al. (2000). Effect of secretin on children with autism: A randomized controlled trial. [In Process Citation]. Developmental Medicine and Child Neurology, 42(12), 796–802.
Fremeau, R. T., Jr., Korman, L. Y., & Moody, T. W. (1986). Secretin stimulates cyclic AMP formation in the rat brain. Journal of Neurochemistry, 46(6), 1947–1955.
Gilliam, J. E. (1995). Gilliam Autism Rating Scale. Austin, TX: Pro-ed.
Hammill, D., Pearson, N., & Voress, J. (1993). Developmental Test of Visual Perception (2nd ed.). Austin, TX: Pro-Ed.
Horvath, K., Papadimitriou, J. C., Rabsztyn, A., Drachenberg, C., & Tildon, J. T. (1999). Gastrointestinal abnormalities in children with autistic disorder [see comments]. Journal of Pediatrics, 135(5), 559–563.
Horvath, K., Stefanatos, G., Sokolski, K. N., Wachtel, R., Nabors, L., & Tildon, J. T. (1998). Improved social and language skills after secretin administration in patients with autistic spectrum disorders. Journal of the Association for Academic Minority Physicians, 9(1), 9–15.
Krug, D., Arick, J., & Almond, P. (1980). Autism Behavior Checklist. Austin, TX: Pro-Ed.
Mullen, E. M. (1995). Mullen Scales of Early Learning. Circle Pines, MN: American Guidance Service.
Owley, T., Steele, M., Corsello, C., Risi, S., McKaig, K., Lord, C., et al. (1999). A double-blind, placebo-controlled trial of secretin for the treatment of autistic disorder. Medscape General Medicine/ journal/1999/v01.n10.
Sandler, A. D., Sutton, K. A., DeWeese, J., Girardi, M. A., Sheppard, V., & Bodfish, J. W. (1999). Lack of benefit of a single dose of synthetic human secretin in the treatment of autism and pervasive developmental disorder [see comments]. New England Journal of Medicine, 341(24), 1801–1806.
Schopler, E. (1988). The Childhood Autism Rating Scale. Los Angeles, CA: Western Psychological Services.
Shipley, K., & McAfee, J. (1992). Assessment in Speech-Language Pathology. San Diego, CA: Singular Publishing Group.
Somogyi, L., Cintron, M., & Toskes, P. P. (1999). Synthetic human secretin compared to synthetic porcine secretin in pancreatic function testing [abstract]. Pancreas, 19, 439.
Somogyi, L., Cintron, M., & Toskes, P. P. (2000). Synthetic porcine secretin is highly accurate in pancreatic function testing in individuals with chronic pancreatitis. Pancreas, 21(3), 262–265.
Stutsman, R. (1981). Merrill-Palmer Scale of Mental Tests. Wood Dale, IL: Stoelting Co.
Ulrich, C. D., II, Holtmann, M., & Miller, L. J. (1998). Secretin and vasoactive intestinal peptide receptors: Members of a unique family of G protein-coupled receptors. Gastroenterology, 114(2), 382–397.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Molloy, C.A., Manning-Courtney, P., Swayne, S. et al. Lack of Benefit of Intravenous Synthetic Human Secretin in the Treatment of Autism. J Autism Dev Disord 32, 545–551 (2002). https://doi.org/10.1023/A:1021202930206
Issue Date:
DOI: https://doi.org/10.1023/A:1021202930206