Abstract
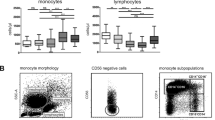
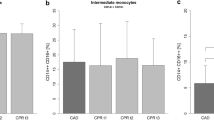
We have asked whether critically ill cardiac valve surgery patients identified by a high APACHE II score exhibit an increase in the number of proin-flammatory CD14+ CD16+ monocytes. A group of 12 patients was studied over a period of 5 days post cardiac valve surgery for changes in blood monocyte populations. Patients were selected on day 1 post surgery to either be in good clinical condition (APACHE II Score of ≤14; N = 9) or to be critically ill (APACHE II score of ≥24; N = 3). The ≤14 patients had an uneventful course and could leave the ICU after 2–3 days. Among the ≥24 patients two showed a decrease of the score to ≤14 within the 5 days of observation and they could leave the ICU thereafter. One ≥24 patient (patient #2) had a persistently high score and finally died on day 28. Analysis of blood monocytes on day 1 post surgery revealed that the ≤14 patients had normal values of CD14+CD16+ monocytes (44 ± 9/μl). By contrast the ≥24 patients had increased values of these cells with 243 ± 106 cells per μ1 on day 1. The numbers of CD14+CD16+ monocytes returned to the control range over the 5 days of observation in 2 of the ≥24 patients concomitant with the improvement of the APACHE II score. CD14+CD16+ monocytes remained, however, at a high level in patient #2, the patient with persistently high APACHE II score.
Similar content being viewed by others
REFERENCES
Passlick, B., D. Flieger, and H. W. L. Ziegler-Heitbrock. 1989. Identification and characterization of a novel monocyte subpopulation in human peripheral blood. Blood 74:2527–2534.
Ziegler-Heitbrock, H. W. L., G. Fingerle, M. StrÖbel, W. Schraut, F. Stelter, C. SchÜtt, B. Passlick, and A. Pforte. 1993. The novel subset of CD14+CD16+ blood monocytes exhibits features of tissue macrophages. Eur. J. Immunol. 23:2053–2058.
Frankenberger, M., T. Sternsdorf, H. Pechumer, A. Pforte, and H. W. L. Ziegler-Heitbrock. 1996. Differential cytokine expression in human blood monocyte subpopulations: a polymerase chain reaction analysis. Blood 87:373–377.
Fingerle, G., A. Pforte, B. Passlick, M. Blumenstein, M. StrÖbel, and H. W. L. Ziegler-Heitbrock. 1993. The novel subset of CD14+CD16+ blood monocytes is expanded in sepsis patients. Blood 82:3170–3176.
Nockher, W. A., L. Bergmann, and J. E. Scherberich. 1994. Increased soluble CD14 serum levels and altered CD14 expression of peripheral blood monocytes in HIV-infected patients. Clin. Exp. Immunol. 98:369–374.
Vanham, G., K. Edmonds, L. Qing, D. Hom, Z. Toossi, B. Jones, C. L. Daley, R. Huebner, L. Kestens, P. Gigase, and J. J. Ellner. 1996. Generalized immune activation in pulmonary tuberculosis: co-activation with HIV infection. Clin. Exp. Immunol. 103:30–34.
Ziegler-Heitbrock, H. W. L. 1996. Heterogeneity of human blood monocytes: the CD14+CD16+ subpopulation. Immunol. Today 17:424–428.
Physicians, MotACoC, and Committee SoCCMCC. 1992. Members of the American College of Chest Physicians/Society of Critical Care Medicine Consensus Conference: Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. Crit. Care. Med. 20:864–874.
Kirklin, J. K., S. Wesaby, E. H. Blackstone, J. W. Kirklin, D. E. Chenoweth, and A. D. Pacifico. 1983. Complement and the damaging effects of cardiopulmonary bypass. J. Thorac. Cardiovasc. Surg. 86:845–857.
Pilz, G., S. KÄÄb, E. Kreuzer, and K. Werdan. 1994. Evaluation of definitions and parameters for sepsis assessment in patients after cardiac surgery. Infection 22:8–17.
Knaus, W. A., E. A. Draper, D. P. Wagner, and J. E. Zimmerman. 1985. APACHE II: A severity of disease classification system. Crit. Care. Med. 13:818–829.
Kreuzer, E., S. KÄÄb, G. Pilz, and K. Werdan. 1992. Early prediction of septic complications after cardiac surgery by APACHE II score. Eur. J. Cardio-Thorac. Surg. 6:524–529.
Pilz, G., E. Kreuzer, S. KÄÄb, R. Appel, and K. Werdan. 1994. Early sepsis treatment with immunoglobulins after cardiac surgery in score-identified high-risk patients. Chest 105:76–82.
Pilz, G., R. Appel, E. Kreuzer, and K. Werdan. 1997. Comparison of early IgM-enriched immunoglobulin vs polyvalent IgG administration in score-identified postcardiac surgical patients at high risk for sepsis. Chest 111:419–426.
Morrow, A. G., C. T. Lambrew, and E. Braunwald. 1964. Idiopathic hypertrophic subaortic stenosis II. Operative treatment and the results of pre-and postoperative hemodynamic evaluations. Circulation 29/30(Suppl. 4): 120–141.
Ziegler-Heitbrock, H. W. L., B. Passlick, and D. Flieger. 1988. The monoclonal anti-monocyte antibody My4 stains B lymphocytes and two distinct monocyte subsets in human peripheral blood. Hybridoma 7:521–527.
Blumenstein, M., P. Boekstegers, P. Fraunberger, R. Andreesen, H. W. L. Ziegler-Heitbrock, and G. R. Fingerle-Rowson. 1997. Cytokine production precedes the expansion of CD14+CD16+-monocytes in human sepsis: a case report of a patient with self-induced septicaemia. Shock 8:73–75.
Weiner, L. M., W. Li, M. Holmes, R. B. Catalano, M. Dovnarsky, K. Padavic, and R. K. Alpaugh. 1994. Phase I trial of recombinant macrophage colony-stimulating factor and recombinant y-interferon: toxicity, monocytosis, and clinical effects. Cancer Res. 54:4084–4090.
Mackensen, A., C. Galanos, U. Wehr, and R. Engelhardt. 1992. Endotoxin tolerance: regulation of cytokine production and cellular changes in response to endotoxin application in cancer patients. Eur. Cytokine Netw. 3:571–579.
Volk, H.-D., P. Reinke, P. Falck, G. Staffa, H. Briedigkeit, and R. V. Baehr. 1989. Diagnostic value of an immune monitoring program for the clinical management of immunosuppressed patients with septic complications. Clin. Transplantation 3:246–252.
Hershman, M. J., W. G. Cheadle, S. R. Wellhausen, P. F. Davidson, and H. Polk, Jr., 1990. Monocyte HLA-DR antigen expression characterizes clinical outcome in the trauma patient. Br. J. Surg. 77:204–207.
Wakefield, C. H., P. D. Carey, S. Foulds, J. R. Monson, and P. J. Guillou. 1993. Changes in major histocompatibility complex class II expression in monocytes and T cells of patients developing infection after surgery. Br. J. Surg. 80:205–209.
Volk, H.-D., M. Thieme, S. Heym, W.-D. Docke, U. Ruppe, W. Tausch, D. Manger, S. Zuckermann, S. Golosubow, B. Nieter, P. Reinke, F. R. Seiler, W. Ax, and R. V. Baehr. 1991. Alterations in function and phenotype of monocytes from patients with septic disease-predictive value and new therapeutic strategies. Behring. Inst. Mitt. 88:208–215.
Ayala, A., W. Ertel, and I. H. Chaudry. 1996. Trauma-induced suppression of antigen presentation and expression of major histocompatibility class II antigen complex in leukocytes. Shock 5:79–90.
Andersen, L. W., L. Baek, H. Degn, J. Lehd, M. Krasnik, and J. P. Rasmussen. 1987. Presence of circulating endotoxins during cardiac operations. J. Thorac. Cardiovasc. Surg. 93:115–119.
Riddington, D. W., B. Venkatesh, C. M. Boivin, R. S. Bonser, T. S. Elliott, T. Marshall, P. J. Mountford, and J. F. Bion. 1996. Intestinal permeability, gastric intramucosal pH, and systemic endotoxemia in patients undergoing cardiopulmonary bypass. JAMA 275:1007–1012.
Munn, D. H., M. B. Garnick, and N.-K. V. Cheung. 1990. Effects of parenteral recombinant human macrophage colony-stimulating factor on monocyte number, phenotype, and antitumor cytotoxicity in nonhuman primates. Blood 75:2042–2048.
Munn, D. H., A. G. Bree, A. C. Beall, M. D. Kaviani, H. Sabio, R. G. Schaub, R. K. Alpaugh, L. M. Weiner, and S. J. Goldman. 1996. Recombinant human macrophage colony-stimulating factor in non-human primates: selective expansion of a CD16+ monocyte subset with phenotypic similarity to primate natural killer cells. Blood 88:1215–1224.
Saleh, M. N., S. J. Goldman, A. F. LoBuglio, A. C. Beall, H. Sabio, M. C. McCord, L. Minasian, R. K. Alpaugh, L. M. Weiner, and D. H. Munn. 1995. CD16+ monocytes in patients with cancer: spontaneous elevation and pharmacologic induction by recombinant human macrophage colony-stimulating factor. Blood 85:2910–2917.
Schmid, I., G. C. Baldwin, E. L. Jacobs, V. Isacescu, N. Neagos, J. V. Giorgi, and J. A. Glaspy. 1995. Alterations in phenotype and cell-surface antigen expression levels of human monocytes: differential response to in vivo administration of rhM-CSF or rhGM-CSF. Cytometry 22:103–110.
Evans, R., S. J. Kamdar, J. A. Fuller, and D. M. Krupke. 1995. The potential role of the macrophage colony-stimulating factor, CSF-1, in inflammatory responses: characterization of macrophage cytokine gene expression. J. Leukoc. Biol. 58:99–107.
Bukowski, R. M., G. T. Budd, J. A. Gibbons, R. J. Bauer, A. Childs, J. Antal, J. Finke, L. Tuason, V. Lorenzi, D. McLain, R. Tubbs, M. Edinger, and M. J. Thomassen. 1994. Phase I trial of subcutaneous recombinant macrophage colony-stimulating factor: clinical and immunomodulatory effects. J. Clin. Oncol. 12:97–106.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fingerle-Rowson, G., Auers, J., Kreuzer, E. et al. Expansion of CD14+CD16+Monocytes in Critically Ill Cardiac Surgery Patients. Inflammation 22, 367–379 (1998). https://doi.org/10.1023/A:1022316815196
Issue Date:
DOI: https://doi.org/10.1023/A:1022316815196