Abstract
So far, the 2019 novel coronavirus (COVID-19) is spreading widely worldwide. The early diagnosis of infection by the severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) is essential to provide timely treatment and prevent its further spread. Lateral flow assays (LFAs) have the advantages of rapid detection, simple operation, low cost, ease of mass production, and no need for special devices and professional operators, which make them suitable for self-testing at home. This review focuses on the early diagnosis of SARS-CoV-2 infection based on optical LFAs including colorimetric, fluorescent (FL), chemiluminescent (CL), and surface-enhanced Raman scattering (SERS) LFAs for the detection of SARS-CoV-2 antigens and nucleic acids. The types of recognition components, detection modes used for antigen detection, labels employed in different optical LFAs, and strategies to improve the detection sensitivity of LFAs were reviewed. Meanwhile, LFAs coupled with different nucleic acid amplification techniques and CRISPR–Cas systems for the detection of SARS-CoV-2 nucleic acids were summarized. We hope this review provides research mentalities for developing highly sensitive LFAs that can be used in home self-testing for the early diagnosis of SARS-CoV-2 infection.
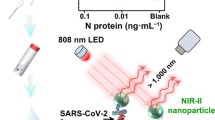
Graphical abstract






Copyright 2023, Elsevier) and b schematic diagram of the structure and operation process of the paint-programmed enhanced lateral flow assay (reprinted with permission from [49]. Copyright 2023, John Wiley and Sons)





Similar content being viewed by others
Data availability
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
References
Coronavirus Cases: Statistics and Charts-Worldometer. https://www.worldometers.info/coronavirus/coronavirus-cases/. Accessed 26 Dec 2023
J. Gao, J.-Q. Liu, H.-J. Wen, H. Liu, W.-D. Hu, X. Han, C.-X. Li, X.-J. Wang, Respir. Res. (2020). https://doi.org/10.1186/s12931-020-01363-7
S.A. Kiryanov, T.A. Levina, V.V. Kadochnikova, M.V. Konopleva, A.P. Suslov, D.Y. Trofimov, Diagnostics (2022). https://doi.org/10.3390/diagnostics12051091
E. Meyers, A. Coen, A. De Sutter, E. Padalko, S. Callens, L. Vandekerckhove, W. Witkowski, S. Heytens, P. Cools, J. Clin. Virol. (2022). https://doi.org/10.1016/j.jcv.2022.105270
S. Thongpradit, S. Prasongtanakij, S. Srisala, S. Chanprasertyothin, E. Pasomsub, B. Ongphiphadhanakul, Int. J. Environ. Res. Public Health (2022). https://doi.org/10.3390/ijerph19137783
M. Asif, M. Ajmal, G. Ashraf, N. Muhammad, A. Aziz, T. Iftikhar, J. Wang, H. Liu, Curr. Opin. Electrochem. (2020). https://doi.org/10.1016/j.coelec.2020.08.011
J. He, S. Zhu, J. Zhou, W. Jiang, L. Yin, L. Su, X. Zhang, Q. Chen, X. Li, Front. Bioeng. Biotechnol. (2023). https://doi.org/10.3389/fbioe.2022.1090281
F. Pei, S. Feng, W. Hu, B. Liu, X. Mu, Q. Hao, Y. Cao, W. Lei, Z. Tong, Talanta (2023). https://doi.org/10.1016/j.talanta.2022.124051
A. Ruggiero, C. Piubelli, L. Calciano, S. Accordini, M.T. Valenti, L.D. Carbonare, G. Siracusano, N. Temperton, N. Tiberti, S.S. Longoni, M. Pizzato, S. Accordini, T. Fantoni, L. Lopalco, A. Beretta, Z. Bisoffi, D. Zipeto, EBioMedicine (2022). https://doi.org/10.1016/j.ebiom.2022.103888
L. Spicuzza, D. Campagna, C. Di Maria, E. Sciacca, S. Mancuso, C. Vancheri, G. Sambataro, AIMS Microbiol. (2023). https://doi.org/10.3934/microbiol.2023020
J.J. Wang, N. Zhang, S.A. Richardson, J.V. Wu, Expert Rev. Mol. Diagn. (2021). https://doi.org/10.1080/14737159.2021.1913123
S.K. Vashist, Diagnostics (2020). https://doi.org/10.3390/diagnostics10040202
Q. Wang, Y. Zhang, L. Wu, S. Niu, C. Song, Z. Zhang, G. Lu, C. Qiao, Y. Hu, K.-Y. Yuen, Q. Wang, H. Zhou, J. Yan, J. Qi, Cell (2020). https://doi.org/10.1016/j.cell.2020.03.045
J. Cubuk, J.J. Alston, J.J. Incicco, S. Singh, M.D. Stuchell-Brereton, M.D. Ward, M.I. Zimmerman, N. Vithani, D. Griffith, J.A. Wagoner, G.R. Bowman, K.B. Hall, A. Soranno, A.S. Holehouse, Nat. Commun. (2021). https://doi.org/10.1038/s41467-021-21953-3
Y. Yamaoka, K. Miyakawa, S.S. Jeremiah, R. Funabashi, K. Okudela, S. Kikuchi, J. Katada, A. Wada, T. Takei, M. Nishi, K. Shimizu, H. Ozawa, S. Usuku, C. Kawakami, N. Tanaka, T. Morita, H. Hayashi, H. Mitsui, K. Suzuki, D. Aizawa, Y. Yoshimura, T. Miyazaki, E. Yamazaki, T. Suzuki, H. Kimura, H. Shimizu, N. Okabe, H. Hasegawa, A. Ryo, Cell Rep. Med. (2021). https://doi.org/10.1016/j.xcrm.2021.100311
J.S. Terry, L.B.R. Anderson, M.S. Scherman, C.E. McAlister, R. Perera, T. Schountz, B.J. Geiss, Virology (2021). https://doi.org/10.1016/j.virol.2021.01.003
R.-M. Lu, S.-H. Ko, W.-Y. Chen, Y.-L. Chang, H.-T. Lin, H.-C. Wu, Int. J. Mol. Sci. (2021). https://doi.org/10.3390/ijms222212412
J. Kim, D. Kim, K. Baek, M. Kim, B.M. Kang, S. Maharjan, S. Park, J.-K. Choi, S. Kim, Y.K. Kim, M.-S. Park, Y. Lee, H.-J. Kwon, Viruses Basel (2022). https://doi.org/10.3390/v15010028
D. Kim, J. Kim, S. Park, M. Kim, K. Baek, M. Kang, J.-K. Choi, S. Maharjan, M. Akauliya, Y. Lee, H.-J. Kwon, Front. Microbiol. (2021). https://doi.org/10.3389/fmicb.2021.726231
J.-H. Lee, Y. Jung, S.-K. Lee, J. Kim, C.-S. Lee, S. Kim, J.-S. Lee, N.-H. Kim, H.-G. Kim, Viruses Basel (2022). https://doi.org/10.3390/v14020255
J. Yang, V.M. Phan, C.-K. Heo, H.V. Nguyen, W.-H. Lim, E.-W. Cho, H. Poo, T.S. Seo, Sens. Actuator B Chem. (2023). https://doi.org/10.1016/j.snb.2023.133331
S. Tao, X. Zhao, D. Bao, X. Liu, W. Zhang, L. Zhao, Y. Tang, H. Wu, H. Ye, Y. Yang, D. Deng, A.C.S. Appl, Mater. Interfaces (2023). https://doi.org/10.1021/acsami.3c03434
N. Salcedo, A. Reddy, A.R. Gomez, I. Bosch, B.B. Herrera, Plos Neglect. Trop. Dis. (2022). https://doi.org/10.1371/journal.pntd.0010311
G. Li, A. Wang, Y. Chen, Y. Sun, Y. Du, X. Wang, P. Ding, R. Jia, Y. Wang, G. Zhang, Front. Immunol. (2021). https://doi.org/10.3389/fimmu.2021.635677
N.V. Antipova, T.D. Larionova, A.E. Siniavin, M.A. Nikiforova, V.A. Gushchin, I.I. Babichenko, A.V. Volkov, M.I. Shakhparonov, M.S. Pavlyukov, Int. J. Mol. Sci. (2020). https://doi.org/10.3390/ijms21239167
S.-H. Ko, W.-Y. Chen, S.-C. Su, H.-T. Lin, F.-Y. Ke, K.-H. Liang, F.-F. Hsu, M. Kumari, C.-Y. Fu, H.-C. Wu, J. Biomed. Sci. (2022). https://doi.org/10.1186/s12929-022-00891-2
B. Zhou, L. Cheng, S. Song, H. Guo, S. Shen, H. Wang, X. Ge, L. Liu, B. Ju, Z. Zhang, Virol. J. (2022). https://doi.org/10.1186/s12985-022-01827-w
M. Gransagne, G. Aymé, S. Brier, G. Chauveau-Le Friec, V. Meriaux, M. Nowakowski, F. Dejardin, S. Levallois, G. Dias De Melo, F. Donati, M. Prot, S. Brûlé, B. Raynal, J. Bellalou, P. Goncalves, X. Montagutelli, J.P. Di Santo, F. Lazarini, P. England, S. Petres, N. Escriou, P. Lafaye, J. Biol. Chem. (2022). https://doi.org/10.1016/j.jbc.2021.101290
J.W. Kim, A.H. Cho, H.G. Shin, S.H. Jang, S.Y. Cho, Y.R. Lee, S. Lee, Viruses Basel (2023). https://doi.org/10.3390/v15010174
P. Segovia-de Los Santos, C. Padula-Roca, X. Simon, C. Echaides, G. Lassabe, G. Gonzalez-Sapienza, Front. Immunol. (2023). https://doi.org/10.3389/fimmu.2023.1220477
L. Zhang, X. Fang, X. Liu, H. Ou, H. Zhang, J. Wang, Q. Li, H. Cheng, W. Zhang, Z. Luo, Chem. Commun. (2020). https://doi.org/10.1039/D0CC03993D
L.F. Yang, N. Kacherovsky, N. Panpradist, R. Wan, J. Liang, B. Zhang, S.J. Salipante, B.R. Lutz, S.H. Pun, Anal. Chem. (2022). https://doi.org/10.1021/acs.analchem.2c00554
L.F. Yang, N. Kacherovsky, J. Liang, S.J. Salipante, S.H. Pun, Anal. Chem. (2022). https://doi.org/10.1021/acs.analchem.2c01993
Y. Song, J. Song, X. Wei, M. Huang, M. Sun, L. Zhu, B. Lin, H. Shen, Z. Zhu, C. Yang, Anal. Chem. (2020). https://doi.org/10.1021/acs.analchem.0c01394
J. Li, Z. Zhang, J. Gu, H.D. Stacey, J.C. Ang, A. Capretta, C.D.M. Filipe, K.L. Mossman, C. Balion, B.J. Salena, D. Yamamura, L. Soleymani, M.S. Miller, J.D. Brennan, Y. Li, Nucleic Acids Res. (2021). https://doi.org/10.1093/nar/gkab574
N. Kacherovsky, L.F. Yang, H.V. Dang, E.L. Cheng, I.I. Cardle, A.C. Walls, M. McCallum, D.L. Sellers, F. DiMaio, S.J. Salipante, D. Corti, D. Veesler, S.H. Pun, Angew. Chem. Int. Edit. (2021). https://doi.org/10.1002/anie.202107730
J.-H. Lee, M. Choi, Y. Jung, S.K. Lee, C.-S. Lee, J. Kim, J. Kim, N.H. Kim, B.-T. Kim, H.G. Kim, Biosens. Bioelectron. (2021). https://doi.org/10.1016/j.bios.2020.112715
A.N. Baker, S.-J. Richards, C.S. Guy, T.R. Congdon, M. Hasan, A.J. Zwetsloot, A. Gallo, J.R. Lewandowski, P.J. Stansfeld, A. Straube, M. Walker, S. Chessa, G. Pergolizzi, S. Dedola, R.A. Field, M.I. Gibson, ACS Central Sci. (2020). https://doi.org/10.1021/acscentsci.0c00855
D. Prakashan, N.S. Shrikrishna, M. Byakodi, K. Nagamani, S. Gandhi, J. Med. Virol. (2023). https://doi.org/10.1002/jmv.28416
J.-H. Bong, T.-H. Kim, J. Jung, S.J. Lee, J.S. Sung, C.K. Lee, M.-J. Kang, H.O. Kim, J.-C. Pyun, BioChip J. (2021). https://doi.org/10.1007/s13206-021-00011-6
C. Wang, X. Cheng, L. Liu, X. Zhang, X. Yang, S. Zheng, Z. Rong, S. Wang, A.C.S. Appl, Mater. Interfaces (2021). https://doi.org/10.1021/acsami.1c11461
W. Wang, X. Yang, Z. Rong, Z. Tu, X. Zhang, B. Gu, C. Wang, S. Wang, Nano Res. (2023). https://doi.org/10.1007/s12274-022-5043-6
Z. Liu, C. Wang, S. Zheng, X. Yang, H. Han, Y. Dai, R. Xiao, Nanomed. Nanotechnol. Biol. Med. (2023). https://doi.org/10.1016/j.nano.2022.102624
W.W.-W. Hsiao, N. Sharma, T.-N. Le, Y.-Y. Cheng, C.-C. Lee, D.-T. Vo, Y.Y. Hui, H.-C. Chang, W.-H. Chiang, Anal. Chim. Acta (2022). https://doi.org/10.1016/j.aca.2022.340389
J. Szekely, J. Mongkolprasert, N. Jeayodae, C. Senorit, P. Chaimuti, P. Swangphon, N. Nanakorn, T. Nualnoi, P. Wongwitwichot, T. Pengsakul, Diagnostics (2022). https://doi.org/10.3390/diagnostics12020381
D. Hong, E.-J. Jo, C. Jung, M.-G. Kim, A.C.S. Appl, Mater. Interfaces (2022). https://doi.org/10.1021/acsami.2c13303
T. Peng, X. Jiao, Z. Liang, H. Zhao, Y. Zhao, J. Xie, Y. Jiang, X. Yu, X. Fang, X. Dai, Biosensors (2021). https://doi.org/10.3390/bios12010013
S. Ruantip, U. Pimpitak, S. Rengpipat, E. Pasomsub, C. Seepiban, O. Gajanandana, P. Torvorapanit, N. Hirankarn, P. Jaru-ampornpan, S. Siwamogsatham, P. Pongpaibool, S. Siwamogsatham, N. Thongchul, S. Chaiyo, Sens. Actuator B Chem. (2023). https://doi.org/10.1016/j.snb.2023.133898
D. Lee, T. Ozkaya-Ahmadov, A.F. Sarioglu, Small (2023). https://doi.org/10.1002/smll.202208035
L. Shen, Q. Zhang, X. Luo, H. Xiao, M. Gu, L. Cao, F. Zhao, Z. Chen, J. Clin. Lab. Anal. (2021). https://doi.org/10.1002/jcla.24091
H.-Y. Kim, J.-H. Lee, M.J. Kim, S.C. Park, M. Choi, W. Lee, K.B. Ku, B.T. Kim, E. Changkyun Park, H.G. Kim, S.I. Kim, Biosens. Bioelectron. (2021). https://doi.org/10.1016/j.bios.2020.112868
C. Liang, B. Liu, J. Li, J. Lu, E. Zhang, Q. Deng, L. Zhang, R. Chen, Y. Fu, C. Li, T. Li, Sens. Actuator B Chem. (2021). https://doi.org/10.1016/j.snb.2021.130718
Y. Sun, Z. Xie, F. Pei, W. Hu, S. Feng, Q. Hao, B. Liu, X. Mu, W. Lei, Z. Tong, Anal. Methods (2022). https://doi.org/10.1039/D2AY01530G
V.G. Panferov, N.A. Byzova, S.F. Biketov, A.V. Zherdev, B.B. Dzantiev, Biosensors Basel (2021). https://doi.org/10.3390/bios11070229
A.S. Lee, S.M. Kim, K.R. Kim, C. Park, D.-G. Lee, H.R. Heo, H.J. Cha, C.S. Kim, Sens. Actuator B Chem. (2023). https://doi.org/10.1016/j.snb.2022.133245
S.C. Razo, N.A. Panferova, V.G. Panferov, I.V. Safenkova, N.V. Drenova, Y.A. Varitsev, A.V. Zherdev, E.N. Pakina, B.B. Dzantiev, Sensors (2019). https://doi.org/10.3390/s19010153
V.G. Panferov, I.V. Safenkova, N.A. Byzova, Y.A. Varitsev, A.V. Zherdev, B.B. Dzantiev, Food Agric. Immunol. (2018). https://doi.org/10.1080/09540105.2017.1401044
W. Tian, X. Gao, J. Zhang, J. Yu, J. Zhang, Carbohydr. Polym. (2022). https://doi.org/10.1016/j.carbpol.2021.118863
K. Murakami, S. Nagatoishi, K. Kasahara, H. Nagai, Y. Sasajima, R. Sasaki, K. Tsumoto, Anal. Biochem. (2021). https://doi.org/10.1016/j.ab.2021.114337
R. Hu, T. Liao, Y. Ren, W. Liu, R. Ma, X. Wang, Q. Lin, G. Wang, Y. Liang, Nano Res. (2022). https://doi.org/10.1007/s12274-022-4351-1
R. Gupta, P. Gupta, S. Wang, A. Melnykov, Q. Jiang, A. Seth, Z. Wang, J.J. Morrissey, I. George, S. Gandra, P. Sinha, G.A. Storch, B.A. Parikh, G.M. Genin, S. Singamaneni, Nat. Biomed. Eng (2023). https://doi.org/10.1038/s41551-022-01001-1
C. Wang, X. Yang, S. Zheng, X. Cheng, R. Xiao, Q. Li, W. Wang, X. Liu, S. Wang, Sens. Actuator B Chem. (2021). https://doi.org/10.1016/j.snb.2021.130372
L.-D. Xu, J. Zhu, S.-N. Ding, Analyst (2021). https://doi.org/10.1039/D1AN01010G
M. Mao, F. Wu, X. Shi, Y. Huang, L. Ma, Biosensors Basel (2022). https://doi.org/10.3390/bios12070437
S.E. Seo, E. Ryu, J. Kim, C.J. Shin, O.S. Kwon, Sens. Actuator B Chem. (2023). https://doi.org/10.1016/j.snb.2023.133364
B. Xu, H. Tang, Y. Weng, V.S. Jones, S. Luo, C.Y. Cho, Y. Lin, J. Fang, X. Song, R. Huang, J. Clin. Lab. Anal. (2022). https://doi.org/10.1002/jcla.24513
J. Guo, S. Chen, S. Tian, K. Liu, J. Ni, M. Zhao, Y. Kang, X. Ma, J. Guo, Biosens. Bioelectron. (2021). https://doi.org/10.1016/j.bios.2021.113160
G.-Q. Zhang, Z. Gao, J. Zhang, H. Ou, H. Gao, R.T.K. Kwok, D. Ding, B.Z. Tang, Cell Rep. Phys. Sci. (2022). https://doi.org/10.1016/j.xcrp.2022.100740
W. Deng, F. Xie, H.T.M.C.M. Baltar, E.M. Goldys, Phys. Chem. Chem. Phys. (2013). https://doi.org/10.1039/c3cp50206f
Q. Ma, J. Wang, Z. Li, X. Lv, L. Liang, Q. Yuan, Small (2019). https://doi.org/10.1002/smll.201804969
S. Lüker, D.E. Reiter, Semicond. Sci. Technol. (2019). https://doi.org/10.1088/1361-6641/ab1c14
X. Cheng, X. Yang, Z. Tu, Z. Rong, C. Wang, S. Wang, J. Hazard. Mater. (2023). https://doi.org/10.1016/j.jhazmat.2023.132192
Y. Liu, Y. Lv, W. Chen, X. Yang, X. Cheng, Z. Rong, S. Wang, A.C.S. Appl, Mater. Interfaces (2023). https://doi.org/10.1021/acsami.3c05424
X. Yang, Q. Yu, X. Cheng, H. Wei, X. Zhang, Z. Rong, C. Wang, S. Wang, A.C.S. Appl, Mater. Interfaces (2023). https://doi.org/10.1021/acsami.2c21042
B. Jin, Z. Du, J. Ji, Y. Bai, D. Tang, L. Qiao, J. Lou, J. Hu, Z. Li, Talanta (2023). https://doi.org/10.1016/j.talanta.2023.124327
Y. Chen, J. Sun, Y. Xianyu, B. Yin, Y. Niu, S. Wang, F. Cao, X. Zhang, Y. Wang, X. Jiang, Nanoscale (2016). https://doi.org/10.1039/C6NR04017A
M. Chabi, B. Vu, K. Brosamer, M. Smith, D. Chavan, J.C. Conrad, R.C. Willson, K. Kourentzi, Analyst (2023). https://doi.org/10.1039/D2AN01499H
D. Liu, C. Ju, C. Han, R. Shi, X. Chen, D. Duan, J. Yan, X. Yan, Biosens. Bioelectron. (2021). https://doi.org/10.1016/j.bios.2020.112817
M. Fan, G.F.S. Andrade, A.G. Brolo, Anal. Chim. Acta (2020). https://doi.org/10.1016/j.aca.2019.11.049
S. Lai, Y. Liu, S. Fang, Q. Wu, M. Fan, D. Lin, J. Lin, S. Feng, J. Biophotonics (2023). https://doi.org/10.1002/jbio.202300004
Y. Joung, K. Kim, S. Lee, B.-S. Chun, S. Lee, J. Hwang, S. Choi, T. Kang, M.-K. Lee, L. Chen, J. Choo, ACS Sens. (2022). https://doi.org/10.1021/acssensors.2c01808
H. Han, C. Wang, X. Yang, S. Zheng, X. Cheng, Z. Liu, B. Zhao, R. Xiao, Sens. Actuator B Chem. (2022). https://doi.org/10.1016/j.snb.2021.130897
Z. Xie, S. Feng, F. Pei, M. Xia, Q. Hao, B. Liu, Z. Tong, J. Wang, W. Lei, X. Mu, Anal. Chim. Acta (2022). https://doi.org/10.1016/j.aca.2022.340486
D.R.P. Nicollete, R. Benedetti, B.A. Valença, K.K. Kuniyoshi, T.C.S. De Jesus, A. Gevaerd, E.B. Santiago, B.M.M. De Almeida, S.R.R. Júnior, M.V.M. Figueredo, Sci. Rep. (2023). https://doi.org/10.1038/s41598-023-31641-5
S.B. Park, J.H. Shin, BioChip J. (2022). https://doi.org/10.1007/s13206-022-00085-w
P. Srithong, S. Chaiyo, E. Pasomsub, S. Rengpipat, O. Chailapakul, N. Praphairaksit, Microchim. Acta (2022). https://doi.org/10.1007/s00604-022-05467-3
J. Kim, C. Kim, J.S. Park, N.E. Lee, S. Lee, S.-Y. Cho, C. Park, D.S. Yoon, Y.K. Yoo, J.H. Lee, Biosens. Bioelectron. (2023). https://doi.org/10.1016/j.bios.2022.114965
J.F.-W. Chan, K.-H. Kok, Z. Zhu, H. Chu, K.K.-W. To, S. Yuan, K.-Y. Yuen, Emerg. Microbes Infect. (2020). https://doi.org/10.1080/22221751.2020.1719902
T. Ji, Z. Liu, G. Wang, X. Guo, S. Akbar Khan, C. Lai, H. Chen, S. Huang, S. Xia, B. Chen, H. Jia, Y. Chen, Q. Zhou, Biosens. Bioelectron. (2020). https://doi.org/10.1016/j.bios.2020.112455
H. Chen, Y. Wang, H. Wei, Z. Rong, S. Wang, RSC Adv. (2022). https://doi.org/10.1039/D1RA07756B
X. Zhu, X. Wang, L. Han, T. Chen, L. Wang, H. Li, S. Li, L. He, X. Fu, S. Chen, M. Xing, H. Chen, Y. Wang, Biosens. Bioelectron. (2020). https://doi.org/10.1016/j.bios.2020.112437
J. Gomez-Martinez, S. Henry, E. Tuaillon, P. Van De Perre, C. Fournier-Wirth, V. Foulongne, J.-C. Brès, Front. Cell. Infect. Microbiol. (2022). https://doi.org/10.3389/fcimb.2022.902914
K. Kim, B. Lee, J.H. Park, J.-H. Park, K.J. Lee, T.J. Kwak, T. Son, Y.-B. Shin, H. Im, M.-G. Kim, Mater. Horizons (2023). https://doi.org/10.1039/D2MH01267G
S. Yu, S.B. Nimse, J. Kim, K.-S. Song, T. Kim, Anal. Chem. (2020). https://doi.org/10.1021/acs.analchem.0c03202
Y. Wang, H. Chen, H. Wei, Z. Rong, S. Wang, Lab Chip (2022). https://doi.org/10.1039/D1LC01167G
T. Notomi, Nucleic Acids Res. (2000). https://doi.org/10.1093/nar/28.12.e63
T. Huang, L. Li, X. Liu, Q. Chen, X. Fang, J. Kong, M.S. Draz, H. Cao, Anal. Methods (2020). https://doi.org/10.1039/D0AY01768J
Y.-P. Wong, S. Othman, Y.-L. Lau, S. Radu, H.-Y. Chee, J. Appl. Microbiol. (2018). https://doi.org/10.1111/jam.13647
S. Agarwal, C. Warmt, J. Henkel, L. Schrick, A. Nitsche, F.F. Bier, Anal. Bioanal. Chem. (2022). https://doi.org/10.1007/s00216-022-03880-4
C. Zhang, T. Zheng, H. Wang, W. Chen, X. Huang, J. Liang, L. Qiu, D. Han, W. Tan, Anal. Chem. (2021). https://doi.org/10.1021/acs.analchem.0c05059
Y.O. Jang, N.H. Kim, Y. Roh, B. Koo, H.J. Lee, J.Y. Kim, S.-H. Kim, Y. Shin, Sens. Actuator B-Chem. (2023). https://doi.org/10.1016/j.snb.2022.133193
J. Li, J. Macdonald, F. Von Stetten, Analyst (2019). https://doi.org/10.1039/C8AN01621F
F. Tu, X. Yang, S. Xu, D. Chen, L. Zhou, X. Ge, J. Han, Y. Zhang, X. Guo, H. Yang, Transbound. Emerg. Dis. (2021). https://doi.org/10.1111/tbed.13849
X. Meng, S. Zou, D. Li, J. He, L. Fang, H. Wang, X. Yan, D. Duan, L. Gao, Biosens. Bioelectron. (2022). https://doi.org/10.1016/j.bios.2022.114739
T.R. Shelite, A.C. Uscanga-Palomeque, A. Castellanos-Gonzalez, P.C. Melby, B.L. Travi, J. Virol. Methods (2021). https://doi.org/10.1016/j.jviromet.2021.114227
Y. Sun, P. Qin, J. He, W. Li, Y. Shi, J. Xu, Q. Wu, Q. Chen, W. Li, X. Wang, G. Liu, W. Chen, Biosens. Bioelectron. (2022). https://doi.org/10.1016/j.bios.2021.113771
G. Wang, X. Yang, H. Dong, Z. Tu, Y. Zhou, Z. Rong, S. Wang, Pathogens (2022). https://doi.org/10.3390/pathogens11111252
J. Qian, S.A. Boswell, C. Chidley, Z. Lu, M.E. Pettit, B.L. Gaudio, J.M. Fajnzylber, R.T. Ingram, R.H. Ward, J.Z. Li, M. Springer, Nat. Commun. (2020). https://doi.org/10.1038/s41467-020-19258-y
T. Zheng, X. Li, Y. Si, M. Wang, Y. Zhou, Y. Yang, N. Liang, B. Ying, P. Wu, Biosens. Bioelectron. (2023). https://doi.org/10.1016/j.bios.2022.114989
C.P. Mancuso, Z.-X. Lu, J. Qian, S.A. Boswell, M. Springer, Anal. Chem. (2021). https://doi.org/10.1021/acs.analchem.1c01576
M.J. Oishee, T. Ali, N. Jahan, S.S. Khandker, M.A. Haq, M.U. Khondoker, B.K. Sil, H. Lugova, A. Krishnapillai, A.R. Abubakar, S. Kumar, M. Haque, M.R. Jamiruddin, N. Adnan, Infect. Drug Resistance (2021). https://doi.org/10.2147/IDR.S289629
Z. Zhu, Y. Guo, C. Wang, Z. Yang, R. Li, Z. Zeng, H. Li, D. Zhang, L. Yang, Biosens. Bioelectron. (2023). https://doi.org/10.1016/j.bios.2023.115179
J. Liu, Y. Zhang, H. Xie, L. Zhao, L. Zheng, H. Ye, Small (2019). https://doi.org/10.1002/smll.201902989
M. Zou, F. Su, R. Zhang, X. Jiang, H. Xiao, X. Yan, C. Yang, X. Fan, G. Wu, Sens. Actuator B Chem. (2021). https://doi.org/10.1016/j.snb.2021.129899
A. Hernandez-Garcia, M.D. Morales-Moreno, E.G. Valdés-Galindo, E.P. Jimenez-Nieto, A. Quezada, Diagnostics (2022). https://doi.org/10.3390/diagnostics12061434
M. Azhar, R. Phutela, M. Kumar, A.H. Ansari, R. Rauthan, S. Gulati, N. Sharma, D. Sinha, S. Sharma, S. Singh, S. Acharya, S. Sarkar, D. Paul, P. Kathpalia, M. Aich, P. Sehgal, G. Ranjan, R.C. Bhoyar, K. Singhal, H. Lad, P.K. Patra, G. Makharia, G.R. Chandak, B. Pesala, D. Chakraborty, S. Maiti, Biosens. Bioelectron. (2021). https://doi.org/10.1016/j.bios.2021.113207
E. Xiong, L. Jiang, T. Tian, M. Hu, H. Yue, M. Huang, W. Lin, Y. Jiang, D. Zhu, X. Zhou, Angew. Chem. Int. Edit. (2021). https://doi.org/10.1002/anie.202014506
T. Marsic, Z. Ali, M. Tehseen, A. Mahas, S. Hamdan, M. Mahfouz, Nano Lett. (2021). https://doi.org/10.1021/acs.nanolett.1c00612
J.P. Broughton, X. Deng, G. Yu, C.L. Fasching, V. Servellita, J. Singh, X. Miao, J.A. Streithorst, A. Granados, A. Sotomayor-Gonzalez, K. Zorn, A. Gopez, E. Hsu, W. Gu, S. Miller, C.-Y. Pan, H. Guevara, D.A. Wadford, J.S. Chen, C.Y. Chiu, Nat. Biotechnol. (2020). https://doi.org/10.1038/s41587-020-0513-4
Lateral Flow Readout, CRISPR/Cas-based detection strategies. https://www.milenia-biotec.com/en/tips-lateral-flow-readouts-crispr-cas-strategies/. Accessed 4 Dec 2023
M. Patchsung, K. Jantarug, A. Pattama, K. Aphicho, S. Suraritdechachai, P. Meesawat, K. Sappakhaw, N. Leelahakorn, T. Ruenkam, T. Wongsatit, N. Athipanyasilp, B. Eiamthong, B. Lakkanasirorat, T. Phoodokmai, N. Niljianskul, D. Pakotiprapha, S. Chanarat, A. Homchan, R. Tinikul, P. Kamutira, K. Phiwkaow, S. Soithongcharoen, C. Kantiwiriyawanitch, V. Pongsupasa, D. Trisrivirat, J. Jaroensuk, T. Wongnate, S. Maenpuen, P. Chaiyen, S. Kamnerdnakta, J. Swangsri, S. Chuthapisith, Y. Sirivatanauksorn, C. Chaimayo, R. Sutthent, W. Kantakamalakul, J. Joung, A. Ladha, X. Jin, J.S. Gootenberg, O.O. Abudayyeh, F. Zhang, N. Horthongkham, C. Uttamapinant, Nat. Biomed. Eng (2020). https://doi.org/10.1038/s41551-020-00603-x
Q. Zhang, J. Li, Y. Li, G. Tan, M. Sun, Y. Shan, Y. Zhang, X. Wang, K. Song, R. Shi, L. Huang, F. Liu, Y. Yi, X. Wu, Biosens. Bioelectron. (2022). https://doi.org/10.1016/j.bios.2022.113978
Z. Li, X. Ding, K. Yin, L. Avery, E. Ballesteros, C. Liu, Biosens. Bioelectron. (2022). https://doi.org/10.1016/j.bios.2021.113865
M.D. Morales-Moreno, E.G. Valdés-Galindo, M.M. Reza, T. Fiordelisio, J. Peon, A. Hernandez-Garcia, CRISPR J. (2023). https://doi.org/10.1089/crispr.2022.0074
K. Dighe, P. Moitra, M. Alafeef, N. Gunaseelan, D. Pan, Biosens. Bioelectron. (2022). https://doi.org/10.1016/j.bios.2021.113900
D. Wang, S. He, X. Wang, Y. Yan, J. Liu, S. Wu, S. Liu, Y. Lei, M. Chen, L. Li, J. Zhang, L. Zhang, X. Hu, X. Zheng, J. Bai, Y. Zhang, Y. Zhang, M. Song, Y. Tang, Nat. Biomed. Eng (2020). https://doi.org/10.1038/s41551-020-00655-z
Acknowledgements
This research was supported by the National Natural Science Foundation of China (21475094).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liang, R., Fan, A., Wang, F. et al. Optical lateral flow assays in early diagnosis of SARS-CoV-2 infection. ANAL. SCI. (2024). https://doi.org/10.1007/s44211-024-00596-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44211-024-00596-6