Abstract
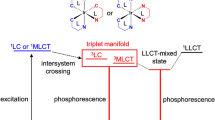
Two red-emitting heteroleptic iridium(III) complexes (Ir-p and Ir-q) were synthesized and their photophysical and biological properties were analyzed. After their structures have been confirmed by several techniques, such as 1H NMR, 13C NMR, FT–IR, UV–Vis, and MALDI TOF analyses, their luminescence behavior was investigated in ethanol and PBS (physiological medium, pH ~ 7.4) solutions. Emission spectra of both complexes are dominated by 3MLCT states at room temperature in ethanolic solution, but at 77 K the Ir-q exhibits the 3LC as the dominant emission state. The Ir-q complex shows one of the highest emission quantum yields, 11.5%, for a red emitter based on iridium(III) complexes in aerated PBS solution, with color coordinates (x;y) of (0.712;0.286). Moreover, both complexes display high potential to be used as a biological marker with excitation wavelengths above 400 nm, high water solubility (Ir-p 1838 μmol L−1, Ir-q 7601 μmol L−1), and distinct emission wavelengths from the biological autofluorescence. Their cytotoxicity was analyzed in CHO-k1 cells by MTT assays, and the IC50 was estimated as being higher than 131 μmol L−1 for Ir-p, and higher than 116 μmol L−1 for Ir-q. Concentrations above 70% of viability were used to perform cell imaging by confocal and fluorescence microscopies and the results suggest that the complexes were internalized by the cell membrane and they are staining the cytoplasm region.






Similar content being viewed by others
References
Wilbuer, A., Vlecken, D. H., Schmitz, D. J., Kräling, K., Harms, K., Bagowski, C. P., & Meggers, E. (2010). Iridium complex with antiangiogenic properties. Angewandte Chemie International Edition, 49(22), 3839–3842. https://doi.org/10.1002/anie.201000682
Li, C., Wang, H., Shen, J., & Tang, B. (2015). Cyclometalated iridium complex-based label-free photoelectrochemical biosensor for DNA detection by hybridization chain reaction amplification. Analytical chemistry, 87(8), 4283–4291. https://doi.org/10.1021/ac5047032
Liu, X., Maegawa, Y., Goto, Y., Hara, K., & Inagaki, S. (2016). Heterogeneous catalysis for water oxidation by an iridium complex immobilized on bipyridine-periodic mesoporous organosilica. Angewandte Chemie International Edition, 55(28), 7943–7947. https://doi.org/10.1002/anie.201601453
Li, T. Y., Wu, J., Wu, Z. G., Zheng, Y. X., Zuo, J. L., & Pan, Y. (2018). Rational design of phosphorescent iridium (III) complexes for emission color tunability and their applications in OLEDs. Coordination Chemistry Reviews, 374, 55–92. https://doi.org/10.1016/j.ccr.2018.06.014
Canisares, F. S., Bispo-Jr, A. G., Pires, A. M., & Lima, S. A. (2020). Syntheses and characterization of Schiff base ligands and their Ir (III) complexes as coating for phosphor-converted LEDs. Optik, 219, 164995. https://doi.org/10.1016/j.ijleo.2020.164995
Lamansky, S., Djurovich, P., Murphy, D., Abdel-Razzaq, F., Kwong, R., Tsyba, I., Bortz, M., Mui, B., Bau, R., & Thompson, M. E. (2001). Synthesis and characterization of phosphorescent cyclometalated iridium complexes. Inorganic Chemistry, 40(7), 1704–1711. https://doi.org/10.1021/ic0008969
Zanoni, K. P., Kariyazaki, B. K., Ito, A., Brennaman, M. K., Meyer, T. J., & Murakami Iha, N. Y. (2014). Blue-green iridium (III) emitter and comprehensive photophysical elucidation of heteroleptic cyclometalated iridium (III) complexes. Inorganic chemistry, 53(8), 4089–4099. https://doi.org/10.1021/ic500070s
Tsuzuki, T., Shirasawa, N., Suzuki, T., & Tokito, S. (2003). Color tunable organic light-emitting diodes using pentafluorophenyl-substituted iridium complexes. Advanced Materials, 15(17), 1455–1458. https://doi.org/10.1002/adma.200305034
Ma, D. L., Wu, C., Tang, W., Gupta, A. R., Lee, F. W., Li, G., & Leung, C. H. (2018). Recent advances in iridium (III) complex-assisted nanomaterials for biological applications. Journal of Materials Chemistry B, 6(4), 537–544. https://doi.org/10.1039/C7TB02859H
Ma, D. L., Ng, H. P., Wong, S. Y., Vellaisamy, K., Wu, K. J., & Leung, C. H. (2018). Iridium (III) complexes as reaction based chemosensors for medical diagnostics. Dalton Transactions, 47(43), 15278–15282. https://doi.org/10.1039/C8DT03492C
Yi, Q. Y., Wan, D., Tang, B., Wang, Y. J., Zhang, W. Y., Du, F., He, M., & Liu, Y. J. (2018). Synthesis, characterization and anticancer activity in vitro and in vivo evaluation of an iridium (III) polypyridyl complex. European Journal of Medicinal Chemistry, 145, 338–349. https://doi.org/10.1016/j.ejmech.2017.11.091
Kapuscinski, J. (1995). DAPI: A DNA-specific fluorescent probe. Biotechnic and Histochemistry, 70(5), 220–233. https://doi.org/10.3109/10520299509108199
Scaduto, R. C., Jr., & Grotyohann, L. W. (1999). Measurement of mitochondrial membrane potential using fluorescent rhodamine derivatives. Biophysical Journal, 76(1), 469–477. https://doi.org/10.1016/S0006-3495(99)77214-0
Pendergrass, W., Wolf, N., & Poot, M. (2004). Efficacy of MitoTracker Green™ and CMXrosamine to measure changes in mitochondrial membrane potentials in living cells and tissues. Cytometry Part A, 61(2), 162–169. https://doi.org/10.1002/cyto.a.20033
Zhao, Q., Yu, M., Shi, L., Liu, S., Li, C., Shi, M., Zhou, Z., Huang, C., & Li, F. (2010). Cationic iridium (III) complexes with tunable emission color as phosphorescent dyes for live cell imaging. Organometallics, 29(5), 1085–1091. https://doi.org/10.1021/om900691r
Murphy, L., Congreve, A., Pålsson, L. O., & Williams, J. G. (2010). The time domain in co-stained cell imaging: Time-resolved emission imaging microscopy using a protonatable luminescent iridium complex. Chemical Communications, 46(46), 8743–8745. https://doi.org/10.1039/c0cc03705b
Zhou, Y., Jia, J., Li, W., Fei, H., & Zhou, M. (2013). Luminescent biscarbene iridium (III) complexes as living cell imaging reagents. Chemical Communications, 49(31), 3230–3232. https://doi.org/10.1039/C3CC40845K
Smith, A. M., Mancini, M. C., & Nie, S. (2009). Second window for in vivo imaging. Nature Nanotechnology, 4(11), 710–711. https://doi.org/10.1038/nnano.2009.326
Liu, T., Zhu, L., Zhong, C., Xie, G., Gong, S., Fang, J., Ma, D., & Yang, C. (2017). Naphthothiadiazole-based near-infrared emitter with a photoluminescence quantum yield of 60% in neat film and external quantum efficiencies of up to 3.9% in nondoped OLEDs. Advanced Functional Materials, 27(12), 1606384. https://doi.org/10.1002/adfm.201606384
Dolan, C., Moriarty, R. D., Lestini, E., Devocelle, M., Forster, R. J., & Keyes, T. E. (2013). Cell uptake and cytotoxicity of a novel cyclometalated iridium (III) complex and its octaarginine peptide conjugate. Journal of Inorganic Biochemistry, 119, 65–74. https://doi.org/10.1016/j.jinorgbio.2012.11.001
Lee, L. C. C., Lau, J. C. W., Liu, H. W., & Lo, K. K. W. (2016). Conferring phosphorogenic properties on iridium (III)-based bioorthogonal probes through modification with a nitrone unit. Angewandte Chemie, 128(3), 1058–1061. https://doi.org/10.1002/ange.201509396
Wang, J., Hou, X., Jin, C., & Chao, H. (2016). A cyclometalated iridium (III) complex serves as a phosphorescent probe for specific mitochondrial imaging in living cells. Chinese Journal of Chemistry, 34(6), 583–588. https://doi.org/10.1002/cjoc.201500769
Lau, J. S. Y., Lee, P. K., Tsang, K. H. K., Ng, C. H. C., Lam, Y. W., Cheng, S. H., & Lo, K. K. W. (2009). Luminescent cyclometalated iridium (III) polypyridine indole complexes synthesis, photophysics, electrochemistry, protein-binding properties, cytotoxicity, and cellular uptake. Inorganic Chemistry, 48(2), 708–718. https://doi.org/10.1021/ic801818x
Li, Y., Liu, B., Xu, C. X., He, L., Wan, Y. C., Ji, L. N., & Mao, Z. W. (2020). Mitochondria-targeted phosphorescent cyclometalated iridium (III) complexes: Synthesis, characterization, and anticancer properties. JBIC Journal of Biological Inorganic Chemistry, 25(4), 597–607. https://doi.org/10.1007/s00775-020-01783-2
Yang, Y., Guo, L., Ge, X., Zhu, T., Chen, W., Zhou, H., Zhao, L., & Liu, Z. (2019). The fluorine effect in zwitterionic half-sandwich iridium (III) anticancer complexes. Inorganic Chemistry, 59(1), 748–758. https://doi.org/10.1021/acs.inorgchem.9b03006
Novohradsky, V., Vigueras, G., Pracharova, J., Cutillas, N., Janiak, C., Kostrhunova, H., Brabec, V., Ruiz, J., & Kasparkova, J. (2019). Molecular superoxide radical photogeneration in cancer cells by dipyridophenazine iridium (III) complexes. Inorganic Chemistry Frontiers, 6(9), 2500–2513. https://doi.org/10.1039/C9QI00811J
Qin, L. Q., Zou, B. Q., Qin, Q. P., Wang, Z. F., Yang, L., Tan, M. X., Liang, C.-J., & Liang, H. (2020). Highly cytotoxic, cyclometalated iridium (III)-5-fluoro-8-quinolinol complexes as cancer cell mitochondriotropic agents. New Journal of Chemistry, 44(19), 7832–7837. https://doi.org/10.1039/D0NJ00465K
Yang, Y., Guo, L., Ge, X., Chen, W., Zhou, H., Zhu, T., Li, X., Tuo, S., & Liu, Z. (2020). Fluorescent zwitterionic Iridium (III) complexes containing sulfonate groups: Synthesis, biological activity and tracking in live cells. Dyes and Pigments, 176, 108220. https://doi.org/10.1016/j.dyepig.2020.108220
Mayo, E. I., Kilså, K., Tirrell, T., Djurovich, P. I., Tamayo, A., Thompson, M. E., Lewis, N. S., & Gray, H. B. (2006). Cyclometalated iridium (III)-sensitized titanium dioxide solar cells. Photochemical and Photobiological Sciences, 5(10), 871–873. https://doi.org/10.1039/B608430C
Nonoyama, M. (1974). Benzo [h] quinolin-10-yl-N Iridium (III) complexes. Bulletin of the Chemical Society of Japan, 47(3), 767–768. https://doi.org/10.1246/bcsj.47.767
Mosmann, T. (1983). Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. Journal of Immunological Methods, 65(1–2), 55–63. https://doi.org/10.1016/0022-1759(83)90303-4
Canisares, F. S., Mutti, A. M., Cavalcante, D. G., Job, A. E., Pires, A. M., & Lima, S. A. (2022). Luminescence and cytotoxic study of red emissive europium (III) complex as a cell dye. Journal of Photochemistry and Photobiology A Chemistry, 422, 113552. https://doi.org/10.1016/j.jphotochem.2021.113552
Cavalcante, D. G., Da Silva, N. D., Marcarini, J. C., Mantovani, M. S., Marin-Morales, M. A., & Martinez, C. B. (2014). Cytotoxic, biochemical and genotoxic effects of biodiesel produced by different routes on ZFL cell line. Toxicology in Vitro, 28(6), 1117–1125. https://doi.org/10.1016/j.tiv.2014.05.008
Pannipara, M., Asiri, A. M., Alamry, K. A., Arshad, M. N., & El-Daly, S. A. (2014). Spectroscopic investigation, effect of solvent polarity and fluorescence quenching of a new D–π–A type chalcone derivative. Journal of Fluorescence, 24(6), 1629–1638. https://doi.org/10.1007/s10895-014-1449-1
Brouwer, A. M. (2011). Standards for photoluminescence quantum yield measurements in solution (IUPAC technical report). Pure and Applied Chemistry, 83(12), 2213–2228. https://doi.org/10.1351/PAC-REP-10-09-31
Jiang, W., Gao, Y., Sun, Y., Ding, F., Xu, Y., Bian, Z., Li, F., Bian, J., & Huang, C. (2010). Zwitterionic iridium complexes: Synthesis, luminescent properties, and their application in cell imaging. Inorganic Chemistry, 49(7), 3252–3260. https://doi.org/10.1021/ic9021239
Tammer, M., & Sokrates, G. (2004). Infrared and Raman characteristic group frequencies: tables and charts. Wiley. https://doi.org/10.1007/s00396-004-1164-6
Emmeluth, C., & Suhm, M. A. (2003). A chemical approach towards the spectroscopy of carboxylic acid dimer isomerism. Physical Chemistry Chemical Physics, 5(15), 3094–3099. https://doi.org/10.1039/B303816E
Papageorgiou, S. K., Kouvelos, E. P., Favvas, E. P., Sapalidis, A. A., Romanos, G. E., & Katsaros, F. K. (2010). Metal–carboxylate interactions in metal–alginate complexes studied with FTIR spectroscopy. Carbohydrate Research, 345(4), 469–473. https://doi.org/10.1016/j.carres.2009.12.010
Deiters, E., Song, B., Chauvin, A. S., Vandevyver, C. D., Gumy, F., & Bünzli, J. C. G. (2009). Luminescent bimetallic lanthanide bioprobes for cellular imaging with excitation in the visible-light range. Chemistry, 15(4), 885–900. https://doi.org/10.1002/chem.200801868
Ma, Y., Liu, S., Yang, H., Wu, Y., Yang, C., Liu, X., Zhao, Q., Wu, H., Liang, J., Li, F., & Huang, W. (2011). Water-soluble phosphorescent iridium (III) complexes as multicolor probes for imaging of homocysteine and cysteine in living cells. Journal of Materials Chemistry, 21(47), 18974–18982. https://doi.org/10.1039/C1JM13513A
Tsuboyama, A., Iwawaki, H., Furugori, M., Mukaide, T., Kamatani, J., Igawa, S., Moriyama, T., Miura, S., Takiguchi, T., Okada, S., & Hoshino, M. (2003). Homoleptic cyclometalated iridium complexes with highly efficient red phosphorescence and application to organic light-emitting diode. Journal of the American Chemical Society, 125(42), 12971–12979. https://doi.org/10.1021/ja034732d
Ning, X., Zhao, C., Jiang, B., Gong, S., Ma, D., & Yang, C. (2019). Green and yellow pyridazine-based phosphorescent iridium (III) complexes for high-efficiency and low-cost organic light-emitting diodes. Dyes and Pigments, 164, 206–212. https://doi.org/10.1016/j.dyepig.2019.01.031
Watts, R. J., & Missimer, D. (1978). Environmentally hindered radiationless transitions between states of different orbital parentage in iridium (III) complexes. Application of rigid-matrix induced perturbations of the pseudo-Jahn-Teller potential to the rigidochromic effect in d6 metal complexes. Journal of the American Chemical Society, 100(17), 5350–5357. https://doi.org/10.1021/ja00485a016
Li, G. N., Zou, Y., Yang, Y. D., Liang, J., Cui, F., Zheng, T., Xie, H., & Niu, Z. G. (2014). Deep-red phosphorescent iridium (III) complexes containing 1-(benzo [b] thiophen-2-yl) isoquinoline ligand: Synthesis, photophysical and electrochemical properties and DFT calculations. Journal of Fluorescence, 24(5), 1545–1552. https://doi.org/10.1007/s10895-014-1443-7
Serroni, S., Juris, A., Campagna, S., Venturi, M., Denti, G., & Balzani, V. (1994). Tetranuclear bimetallic complexes of ruthenium, osmium, rhodium, and iridium. Synthesis, absorption spectra, luminescence, and electrochemical properties. Journal of the American Chemical Society, 116(20), 9086–9091. https://doi.org/10.1021/ja00099a026
Calogero, G., Giuffrida, G., Serroni, S., Ricevuto, V., & Campagna, S. (1995). Absorption spectra, luminescence properties, and electrochemical behavior of cyclometalated iridium (III) and rhodium (III) complexes with a Bis (pyridyl) triazole ligand. Inorganic Chemistry, 34(3), 541–545. https://doi.org/10.1021/ic00107a004
Zhao, Q., Liu, S., Shi, M., Wang, C., Yu, M., Li, L., Li, F., Yi, T., & Huang, C. (2006). Series of new cationic iridium (III) complexes with tunable emission wavelength and excited state properties: Structures, theoretical calculations, and photophysical and electrochemical properties. Inorganic Chemistry, 45(16), 6152–6160. https://doi.org/10.1021/ic052034j
Wang, C. (2017). electronic structure of π-conjugated materials and their effect on organic photovoltaics (Vol. 1893). Linköping University Electronic Press. https://doi.org/10.3384/diss.diva-143025
Duvenhage, M. M., Ntwaeaborwa, M., Visser, H. G., Swarts, P. J., Swarts, J. C., & Swart, H. C. (2015). Determination of the optical band gap of Alq3 and its derivatives for the use in two-layer OLEDs. Optical Materials, 42, 193–198. https://doi.org/10.1016/j.optmat.2015.01.008
Aziz, S. B., Abdullah, O. G., & Rasheed, M. A. (2017). A novel polymer composite with a small optical band gap: New approaches for photonics and optoelectronics. Journal of Applied Polymer Science. https://doi.org/10.1002/app.44847
Lo, K.-W., Lee, P.-K., & Lau, J.-Y. (2008). Synthesis, characterization, and properties of luminescent organoiridium (III) polypyridine complexes appended with an alkyl chain and their interactions with lipid bilayers, surfactants, and living cells. Organometallics, 27(13), 2998–3006. https://doi.org/10.1021/om800212t
Qiu, K., Liu, Y., Huang, H., Liu, C., Hongyi Zhu, Yu., Chen, L. J., & Chao, H. (2016). Biscylometalated iridium (iii) complexes target mitochondria or lysosomes by regulating the lipophilicity of the main ligands. Dalton Transactions, 45, 16144–16147. https://doi.org/10.1039/C6DT03328H
He, L., Cao, J.-J., Zhang, D.-Y., Hao, L., Zhang, M.-F., Tan, C.-P., Ji, L.-N., & Mao, Z.-W. (2018). Lipophilic phosphorescent iridium (III) complexes as one-and two-photon selective bioprobes for lipid droplets imaging in living cells. Sensors and Actuators B Chemical, 262, 313–325. https://doi.org/10.1016/j.snb.2018.02.022
International Organization for Standardization. (2009). ISO 10993-5: 2009-Biological evaluation of medical devices-Part 5: Tests for in vitro cytotoxicity.
Monici, M. (2005). Cell and tissue autofluorescence research and diagnostic applications. Biotechnology Annual Review, 11, 227–256. https://doi.org/10.1016/S1387-2656(05)11007-2
Thorp-Greenwood, F. L., Balasingham, R. G., & Coogan, M. P. (2012). Organometallic complexes of transition metals in luminescent cell imaging applications. Journal of Organometallic Chemistry, 714, 12–21. https://doi.org/10.1016/j.jorganchem.2012.01.020
Mutti, A. M. G., Santos, J. A. O., Cavalcante, D. G. S. M., Gomes, A. S., Job, A. E., Teixeira, G. R., Pires, A. M., & Lima, S. A. M. (2019). Design of a red-emitter hybrid material for bioimaging: Europium complexes grafted on silica particles. Materials Today Chemistry, 14, 100204. https://doi.org/10.1016/j.mtchem.2019.100204
Fan, Y., Zhao, J., Yan, Q., Chen, P. R., & Zhao, D. (2014). Water-soluble triscyclometalated organoiridium complex: Phosphorescent nanoparticle formation, nonlinear optics, and application for cell imaging. ACS Applied Materials and Interfaces, 6(5), 3122–3131. https://doi.org/10.1021/am500549y
Qiu, K., Huang, H., Liu, B., Liu, Y., Huang, Z., Chen, Y., Ji, L., & Chao, H. (2016). Long-term lysosomes tracking with a water-soluble two-photon phosphorescent iridium (III) complex. ACS Applied Materials and Interfaces, 8(20), 12702–12710. https://doi.org/10.1021/acsami.6b03422
Nakagawa, A., Hisamatsu, Y., Moromizato, S., Kohno, M., & Aoki, S. (2014). Synthesis and photochemical properties of pH responsive tris-cyclometalated iridium (III) complexes that contain a pyridine ring on the 2-phenylpyridine ligand. Inorganic Chemistry, 53(1), 409–422. https://doi.org/10.1021/ic402387b
Liu, Y., Zhang, P., Fang, X., Wu, G., Chen, S., Zhang, Z., Chao, H., Tan, W., & Xu, L. (2017). Near-infrared emitting iridium (III) complexes for mitochondrial imaging in living cells. Dalton Transactions, 46(14), 4777–4785. https://doi.org/10.1039/C7DT00255F
Nam, J. S., Kang, M. G., Kang, J., Park, S. Y., Lee, S. J. C., Kim, H. T., Seo, J. K., Kwon, O. H., Lim, M. H., Rhee, H. W., & Kwon, T. H. (2016). Endoplasmic reticulum-localized iridium (III) complexes as efficient photodynamic therapy agents via protein modifications. Journal of the American Chemical Society, 138(34), 10968–10977. https://doi.org/10.1021/jacs.6b05302
Zhang, K. Y., Liu, H. W., Fong, T. T. H., Chen, X. G., & Lo, K. K. W. (2010). Luminescent dendritic cyclometalated iridium (III) polypyridine complexes: Synthesis, emission behavior, and biological properties. Inorganic Chemistry, 49(12), 5432–5443. https://doi.org/10.1021/ic902443e
Shaikh, S., Wang, Y., Rehman, F., Jiang, H., & Wang, X. (2020). Phosphorescent Ir (III) complexes as cellular staining agents for biomedical molecular imaging. Coordination Chemistry Reviews, 416, 213344. https://doi.org/10.1016/j.ccr.2020.213344
Steunenberg, P., Ruggi, A., van den Berg, N. S., Buckle, T., Kuil, J., van Leeuwen, F. W., & Velders, A. H. (2012). Phosphorescence imaging of living cells with amino acid-functionalized tris (2-phenylpyridine) iridium (III) complexes. Inorganic Chemistry, 51(4), 2105–2114. https://doi.org/10.1021/ic201860s
Caporale, C., & Massi, M. (2018). Cyclometalated iridium (III) complexes for life science. Coordination Chemistry Reviews, 363, 71–91. https://doi.org/10.1016/j.ccr.2018.02.006
Acknowledgements
The authors are thankful to the Brazilian agencies FAPESP (Grant No.2019/26103-7), CNPq (Grant No.304003/2018-1) and CAPES for the financial research support. Felipe S. M. Canisares is particularly grateful to the São Paulo research Foundation (FAPESP Grant No.2015/03400-5) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES Grant No.88887.341772/2019-00) for the awarded scholarships. Laboratório de Difração de Raios X (FCT/UNESP), Núcleo de Inovação Tecnológica em Borracha Natural (FCT/UNESP), and Laboratório de Catálise Organometálica e Materiais (FCT/UNESP) for the cyclic voltammetry and FTIR measurements.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Canisares, F.S.M., Mutti, A.M.G., Santana, E.F. et al. Red-emitting heteroleptic iridium(III) complexes: photophysical and cell labeling study. Photochem Photobiol Sci 21, 1077–1090 (2022). https://doi.org/10.1007/s43630-022-00200-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43630-022-00200-8