Abstract
Introduction
Riluzole, a benzothiazole sodium channel blocker is acknowledged as a neuroprotective agent in spinal cord injury (SCI). Most of this evidence is based on pre-clinical studies and its effectiveness in clinical setting is undetermined, heretofore.
Methods
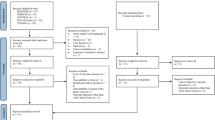
A prospective, randomised-controlled study was conducted between April 2019 and March 2020 at a tertiary-level centre. Patients aged 18–65 years with sub-axial cervical spine injury, who presented within 72 h of injury with incomplete neuro-deficit, were included. They were randomised into groups A (riluzole was administered) and B (no adjuvants). All patients were followed up at 6 weeks/3/6/12 months, and clinical [ASIA motor/sensory scores/grade, SCIM3, and NRS (neuropathic pain)] and radiological evaluation was performed.
Results
Twenty-three and 20 patients were included in groups A and B. Two in group A were females, while others were males (p = 0.49). Mean age in groups A and B was 47.7 ± 14.8 and 51.2 ± 14.1 years (p = 0.44). Five patients died prior to 6th-week follow-up. Among the others, there was significant improvement in all neurological parameters in both groups (post-injury vs 1-year; motor score: p < 0.001, sensory score: p < 0.001, SCIM3: p < 0.001, NRS: p < 0.001). In both groups, initial significant improvement was noticed even at the 6th-week follow-up, which further continued until the end of 1 year. There was no statistically significant difference between groups A and B with respect to these neurological parameters (motor: p = 0.15, sensory: p = 0.39, SCIM3: p = 0.68, NRS: p = 0.06).
Conclusion
Administration of riluzole did not significantly improve neurological outcome/neuropathic pain in our cohort. Nevertheless, both our groups demonstrated an overall improvement in neurological outcome at 1 year, as compared with immediate post-injury status.


Similar content being viewed by others
References
Wilson, J. R., & Fehlings, M. G. (2014). Riluzole for acute traumatic spinal cord injury: A promising neuroprotective treatment strategy. World Neurosurgery, 81, 825–829. https://doi.org/10.1016/j.wneu.2013.01.001
Bracken, M. B., Shepard, M. J., Collins, W. F., et al. (1990). A randomized, controlled trial of methylprednisolone or naloxone in the treatment of acute spinal-cord injury. Results of the Second National Acute Spinal Cord Injury Study. New England Journal of Medicine, 322, 1405–1411. https://doi.org/10.1056/NEJM199005173222001
Agrawal, S. K., & Fehlings, M. G. (1996). Mechanisms of secondary injury to spinal cord axons in vitro: role of Na+, Na(+)-K(+)-ATPase, the Na(+)-H+ exchanger, and the Na(+)-Ca2+ exchanger. Journal of Neuroscience: the Official Journal of the Society for Neuroscience, 16, 545–552.
Amar, A. P., & Levy, M. L. (1999). Pathogenesis and pharmacological strategies for mitigating secondary damage in acute spinal cord injury. Neurosurgery, 44, 1027–1039. https://doi.org/10.1097/00006123-199905000-00052 discussion 1039-1040.
Schwartz, G., & Fehlings, M. G. (2002). Secondary injury mechanisms of spinal cord trauma: a novel therapeutic approach for the management of secondary pathophysiology with the sodium channel blocker Riluzole. Progress Brain Research, 137, 177–190. https://doi.org/10.1016/s0079-6123(02)37016-x
Zhang, Y., Al Mamun, A., Yuan, Y., et al. (2021). Acute spinal cord injury: Pathophysiology and pharmacological intervention (Review). Molecular Medicine Reports. https://doi.org/10.3892/mmr.2021.12056
Fehlings, M. G., Wilson, J. R., Frankowski, R. F., et al. (2012). Riluzole for the treatment of acute traumatic spinal cord injury: rationale for and design of the NACTN Phase I clinical trial. Journal of Neurosurgery. Spine, 17, 151–156. https://doi.org/10.3171/2012.4.AOSPINE1259
Bensimon, G., Lacomblez, L., Delumeau, J. C., et al. (2002). A study of riluzole in the treatment of advanced stage or elderly patients with amyotrophic lateral sclerosis. Journal of Neurology, 249, 609–615. https://doi.org/10.1007/s004150200071
Bensimon, G., Ludolph, A., Agid, Y., et al. (2009). Riluzole treatment, survival and diagnostic criteria in Parkinson plus disorders: the NNIPPS study. Brain: a Journal of Neurology, 132, 156–171. https://doi.org/10.1093/brain/awn291
Vallée, A., Vallée, J.-N., Guillevin, R., & Lecarpentier, Y. (2020). Riluzole: A therapeutic strategy in Alzheimer’s disease by targeting the WNT/β-catenin pathway. Aging, 12, 3095–3113. https://doi.org/10.18632/aging.102830
Lesuis, S. L., Kaplick, P. M., Lucassen, P. J., & Krugers, H. J. (2019). Treatment with the glutamate modulator riluzole prevents early life stress-induced cognitive deficits and impairments in synaptic plasticity in APPswe/PS1dE9 mice. Neuropharmacology, 150, 175–183. https://doi.org/10.1016/j.neuropharm.2019.02.023
Fehlings, M. G., Badhiwala, J. H., Ahn, H., et al. (2021). Safety and efficacy of riluzole in patients undergoing decompressive surgery for degenerative cervical myelopathy (CSM-Protect): A multicentre, double-blind, placebo-controlled, randomised, phase 3 trial. Lancet Neurology, 20, 98–106. https://doi.org/10.1016/S1474-4422(20)30407-5
Meshkini, A., Salehpour, F., Aghazadeh, J., et al. (2018). Riluzole can improve sensory and motor function in patients with acute spinal cord injury. Asian Journal of Neurosurgery, 13, 656–659. https://doi.org/10.4103/ajns.AJNS_259_16
Fehlings, M. G., Nakashima, H., Nagoshi, N., et al. (2016). Rationale, design and critical end points for the Riluzole in Acute Spinal Cord Injury Study (RISCIS): A randomized, double-blinded, placebo-controlled parallel multi-center trial. Spinal Cord, 54, 8–15. https://doi.org/10.1038/sc.2015.95
Grossman, R. G., Fehlings, M. G., Frankowski, R. F., et al. (2014). A prospective, multicenter, phase I matched-comparison group trial of safety, pharmacokinetics, and preliminary efficacy of riluzole in patients with traumatic spinal cord injury. Journal of Neurotrauma, 31, 239–255. https://doi.org/10.1089/neu.2013.2969
Rouanet, C., Reges, D., Rocha, E., et al. (2017). Traumatic spinal cord injury: Current concepts and treatment update. Arquivos de Neuro-Psiquiatria, 75, 387–393. https://doi.org/10.1590/0004-282X20170048
Fehlings, M. G., Vaccaro, A., Wilson, J. R., et al. (2012). Early versus delayed decompression for traumatic cervical spinal cord injury: Results of the Surgical Timing in Acute Spinal Cord Injury Study (STASCIS). PLoS One, 7, e32037. https://doi.org/10.1371/journal.pone.0032037
Liu, J.-M., Long, X.-H., Zhou, Y., et al. (2016). Is urgent decompression superior to delayed surgery for traumatic spinal cord injury? A meta-analysis. World Neurosurgery, 87, 124–131. https://doi.org/10.1016/j.wneu.2015.11.098
Jug, M., Kejžar, N., Vesel, M., et al. (2015). Neurological recovery after traumatic cervical spinal cord injury is superior if surgical decompression and instrumented fusion are performed within 8 hours versus 8 to 24 hours after injury: A single center experience. Journal of Neurotrauma, 32, 1385–1392. https://doi.org/10.1089/neu.2014.3767
Ahuja, C. S., & Fehlings, M. (2016). Concise review: bridging the gap: Novel neuroregenerative and neuroprotective strategies in spinal cord injury. Stem Cells Translational Medicine, 5, 914–924. https://doi.org/10.5966/sctm.2015-0381
Ahuja, C. S., Nori, S., Tetreault, L., et al. (2017). Traumatic spinal cord injury-repair and regeneration. Neurosurgery, 80, S9–S22. https://doi.org/10.1093/neuros/nyw080
Wu, Q., Zhang, Y., Zhang, Y., et al. (2020). Riluzole improves functional recovery after acute spinal cord injury in rats and may be associated with changes in spinal microglia/macrophages polarization. Neuroscience Letters, 723, 134829. https://doi.org/10.1016/j.neulet.2020.134829
Karadimas, S. K., Laliberte, A. M., Tetreault, L., et al. (2015). Riluzole blocks perioperative ischemia-reperfusion injury and enhances postdecompression outcomes in cervical spondylotic myelopathy. Science Translational Medicine, 7, 316ra194. https://doi.org/10.1126/scitranslmed.aac6524
Moon, E. S., Karadimas, S. K., Yu, W.-R., et al. (2014). Riluzole attenuates neuropathic pain and enhances functional recovery in a rodent model of cervical spondylotic myelopathy. Neurobiology of Diseases, 62, 394–406. https://doi.org/10.1016/j.nbd.2013.10.020
Wu, Y., Satkunendrarajah, K., Teng, Y., et al. (2013). Delayed post-injury administration of riluzole is neuroprotective in a preclinical rodent model of cervical spinal cord injury. Journal of Neurotrauma, 30, 441–452. https://doi.org/10.1089/neu.2012.2622
Rajasekaran, S., Aiyer, S. N., Shetty, A. P., et al. (2016). Effectiveness of Riluzole as a pharmacotherapeutic treatment option for early cervical myelopathy: A double-blinded, placebo-controlled randomised controlled trial. European Spine Journal: Official Publication of the European Spine Society, the European Spinal Deformity Society, and the European Section of the Cervical Spine Research Society, 25, 1830–1835. https://doi.org/10.1007/s00586-015-4323-1
Gloviczki, B., Török, D. G., Márton, G., et al. (2017). Delayed spinal cord-brachial plexus reconnection after C7 ventral root avulsion: The effect of reinnervating motoneurons rescued by Riluzole treatment. Journal of Neurotrauma, 34, 2364–2374. https://doi.org/10.1089/neu.2016.4754
Nicholson, K. J., Zhang, S., Gilliland, T. M., & Winkelstein, B. A. (2014). Riluzole effects on behavioral sensitivity and the development of axonal damage and spinal modifications that occur after painful nerve root compression. Journal of Neurosurgery. Spine, 20, 751–762. https://doi.org/10.3171/2014.2.SPINE13672
Tetreault, L. A., Zhu, M. P., Wilson, J. R., et al. (2020). The impact of Riluzole on neurobehavioral outcomes in preclinical models of traumatic and nontraumatic spinal cord injury: Results from a systematic review of the literature. Global Spine Journal, 10, 216–229. https://doi.org/10.1177/2192568219835516
Zhou, L.-Y., Tian, Z.-R., Yao, M., et al. (2019). Riluzole promotes neurological function recovery and inhibits damage extension in rats following spinal cord injury: A meta-analysis and systematic review. Journal of Neurochemistry, 150, 6–27. https://doi.org/10.1111/jnc.14686
Funding
IASA-ASSI (Indo American Spine Alliance—Association of Spine Surgeons of India) grant.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors have no conflicts of interest to disclose.
Ethical Standard Statement
This article does not contain any studies with human or animal subjects performed by the any of the authors.
Informed Consent
For this type of study informed consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kumarasamy, D., Viswanathan, V.K., Shetty, A.P. et al. The Role of Riluzole in Acute Traumatic Cervical Spinal Cord Injury with Incomplete Neurological Deficit: A Prospective, Randomised Controlled Study. JOIO 56, 2160–2168 (2022). https://doi.org/10.1007/s43465-022-00758-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43465-022-00758-6