Abstract
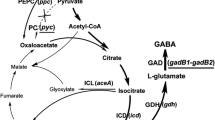
Gamma-aminobutyric acid is an important nonprotein amino acid and has been extensively applied in pharmaceuticals, livestock, food additives, and so on. It is important to develop Corynebacterium glutamicum strains that can efficiently produce gamma-aminobutyric acid from glucose. In this study, production of gamma-aminobutyric acid in C. glutamicum CGY700 was improved by construction of CO2 anaplerotic reaction and overexpression of citrate synthase. The co-expression of ppc encoding phosphoenolpyruvate carboxylase and gltA encoding citrate synthase was constructed and optimized in the chromosome to compensate carbon loss and conquer metabolic bottleneck. The expression of ppc and gltA were controlled by promoters Ptac and PtacM, and the optimal mode of PtacM-ppc-Ptac-gltA was determined. Simultaneously, the genes pknG encoding serine/threonine protein kinase G and ldh encoding l-lactate dehydrogenase were deleted, and glnA2 encoding glutamine synthase was overexpressed in the chromosome. The final strain CGY-PG-304 constructed in this study could produce 41.17 g/L gamma-aminobutyric acid in shake flask cultivation and 58.33 g/L gamma-aminobutyric acid via Fed-Batch fermentation with a yield of 0.30 g/g glucose. CGY-PG-304 was constructed by genome editing; therefore, it is stable and not necessary to add any antibiotics and inducer during fermentation.








Similar content being viewed by others
Availability of data and material
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
References
Nikmaram N, Dar BN, Roohinejad S, Koubaa M, Barba FJ, Greiner R, Johnson SK. Recent advances in gamma-aminobutyric acid (GABA) properties in pulses: an overview. J Sci Food Agric. 2017;97(9):2681–9.
Yang J, Sun C, Zhang Y, Fu D, Zheng X, Yu T. Induced resistance in tomato fruit by γ-aminobutyric acid for the control of alternaria rot caused by Alternaria alternata. Food Chem. 2017;221:1014–20.
Seitanidou M, Franco-Gonzalez JF, Sjostrom TA, Zozoulenko I, Berggren M, Simon DT. pH Dependence of gamma-aminobutyric acid iontronic transport. J Phys Chem B. 2017;121(30):7284–9.
Jacobson LH, Vlachou S, Slattery DA, Li X, Cryan JF. The gamma-aminobutyric acid B receptor in depression and reward. Biol Psychiatry. 2018;83(11):963–76.
Wong CG, Bottiglieri T, Snead OC 3rd. GABA, gamma-hydroxybutyric acid, and neurological disease. Ann Neurol. 2003;54(Suppl 6):S3-12.
Tang J, Chen Z. The protective effect of γ-aminobutyric acid on the development of immune function in chickens under heat stress. J Anim Physiol Anim Nutr. 2016;100(4):768–77.
Cheng JB, Bu DP, Wang JQ, Sun XZ, Pan L, Zhou LY, Liu W. Effects of rumen-protected γ-aminobutyric acid on performance and nutrient digestibility in heat-stressed dairy cows. J Dairy Sci. 2014;97(9):5599–607.
Kim JY, Lee MY, Ji GE, Lee YS, Hwang KT. Production of γ-aminobutyric acid in black raspberry juice during fermentation by Lactobacillus brevis GABA100. Int J Food Microbiol. 2009;130(1):12–6.
Nakamura H, Takishima T, Kometani T, Yokogoshi H. Psychological stress-reducing effect of chocolate enriched with γ-aminobutyric acid (GABA) in humans: assessment of stress using heart rate variability and salivary chromogranin A. Int J Food Sci Nutr. 2009;60(sup5):106–13.
Inoue K, Shirai T, Ochiai H, Kasao M, Hayakawa K, Kimura M, Sansawa H. Blood-pressure-lowering effect of a novel fermented milk containing γ-aminobutyric acid (GABA) in mild hypertensives. Eur J Clin Nutr. 2003;57:490–5.
Leonte A, Colzato LS, Steenbergen L, Hommel B, Akyurek EG. Supplementation of gamma-aminobutyric acid (GABA) affects temporal, but not spatial visual attention. Brain Cogn. 2018;120:8–16.
Park SJ, Kim EY, Noh W, Oh YH, Kim HY, Song BK, Cho KM, Hong SH, Lee SH, Jegal J. Synthesis of nylon 4 from gamma-aminobutyrate (GABA) produced by recombinant Escherichia coli. Bioprocess Biosyst Eng. 2013;36(7):885–92.
Lammens TM, Franssen MCR, Scott EL, Sanders JPM. Synthesis of biobased N-methylpyrrolidone by one-pot cyclization and methylation of γ-aminobutyric acid. Green Chem. 2010;12(8):1430.
Boonstra E, de Kleijn R, Colzato LS, Alkemade A, Forstmann BU, Nieuwenhuis S. Neurotransmitters as food supplements: the effects of GABA on brain and behavior. Front Psychol. 2015;6:1520.
Ke C, Yang X, Rao H, Zeng W, Hu M, Tao Y, Huang J. Whole-cell conversion of l-glutamic acid into gamma-aminobutyric acid by metabolically engineered Escherichia coli. Springerplus. 2016;5(1):1–8.
Luo H, Liu Z, Xie F, Bilal M, Liu L, Yang R, Wang Z. Microbial production of gamma-aminobutyric acid: applications, state-of-the-art achievements, and future perspectives. Crit Rev Biotechnol. 2021;41:491–512.
Shan Y, Man CX, Han X, Li L, Guo Y, Deng Y, Li T, Zhang LW, Jiang YJ. Evaluation of improved γ-aminobutyric acid production in yogurt using Lactobacillus plantarum NDC75017. J Dairy Sci. 2015;98(4):2138–49.
Wang H, Huang J, Sun L, Xu F, Zhang W, Zhan J. An efficient process for co-production of γ-aminobutyric acid and probiotic Bacillus subtilis cells. Food Sci Biotechnol. 2018;28(1):155–63.
Zhao A, Hu X, Wang X. Metabolic engineering of Escherichia coli to produce gamma-aminobutyric acid using xylose. Appl Microbiol Biotechnol. 2017;101(9):3587–603.
Jorge JMP, Leggewie C, Wendisch VF. A new metabolic route for the production of gamma-aminobutyric acid by Corynebacterium glutamicum from glucose. Amino Acids. 2016;48(11):2519–31.
Jorge JMP, Nguyen AQD, Perez-Garc F, Kind S, Wendisch VF. Improved fermentative production of gamma-aminobutyric acid via the putrescine route: systems metabolic engineering for production from glucose, amino sugars, and xylose. Biotechnol Bioeng. 2017;114(4):862–73.
Thu Ho NA, Hou CY, Kim WH, Kang TJ. Expanding the active pH range of Escherichia coli glutamate decarboxylase by breaking the cooperativeness. J Biosci Bioeng. 2013;115(2):154–8.
Yu K, Lin L, Hu S, Huang J, Mei L. C-terminal truncation of glutamate decarboxylase from Lactobacillus brevis CGMCC 1306 extends its activity toward near-neutral pH. Enzyme Microb Technol. 2012;50(4–5):263–9.
Shi F, Xie Y, Jiang J, Wang N, Li Y, Wang X. Directed evolution and mutagenesis of glutamate decarboxylase from Lactobacillus brevis Lb85 to broaden the range of its activity toward a near-neutral pH. Enzyme Microb Technol. 2014;61–62:35–43.
Park S-H, Sohn YJ, Park SJ, Choi J-I. Effect of DR1558, a Deinococcus radiodurans response regulator, on the production of GABA in the recombinant Escherichia coli under low pH conditions. Microb Cell Fact. 2020;19(1):1–12.
Shi F, Luan M, Li Y. Ribosomal binding site sequences and promoters for expressing glutamate decarboxylase and producing gamma-aminobutyrate in Corynebacterium glutamicum. AMB Expr. 2018;8(1):61.
Takahashi C, Shirakawa J, Tsuchidate T, Okai N, Hatada K, Nakayama H, Tateno T, Ogino C, Kondo A. Robust production of gamma-amino butyric acid using recombinant Corynebacterium glutamicum expressing glutamate decarboxylase from Escherichia coli. Enzyme Microb Technol. 2012;51(3):171–6.
Baritugo K-A, Kim HT, David Y, Khang TU, Hyun SM, Kang KH, Yu JH, Choi JH, Song JJ, Joo JC, Park SJ. Enhanced production of gamma-aminobutyrate (GABA) in recombinant Corynebacterium glutamicum strains from empty fruit bunch biosugar solution. Microb Cell Fact. 2018;17(1):1–12.
Zhang R, Yang T, Rao Z, Sun H, Xu M, Zhang X, Xu Z, Yang S. Efficient one-step preparation of γ-aminobutyric acid from glucose without an exogenous cofactor by the designed Corynebacterium glutamicum. Green Chem. 2014;16(9):4190.
Choi JW, Yim SS, Lee SH, Kang TJ, Park SJ, Jeong KJ. Enhanced production of gamma-aminobutyrate (GABA) in recombinant Corynebacterium glutamicum by expressing glutamate decarboxylase active in expanded pH range. Microb Cell Fact. 2015;14(1):1-11.
Yao C, Hu X, Wang X. Construction and application of a CRISPR/Cas9-assisted genomic editing system for Corynebacterium glutamicum. AMB Expr. 2021;11(1):1–15.
Schultz C, Niebisch A, Gebel L, Bott M. Glutamate production by Corynebacterium glutamicum: dependence on the oxoglutarate dehydrogenase inhibitor protein OdhI and protein kinase PknG. Appl Microbiol Biotechnol. 2007;76(3):691–700.
Okai N, Takahashi C, Hatada K, Ogino C, Kondo A. Disruption of pknG enhances production of gamma-aminobutyric acid by Corynebacterium glutamicum expressing glutamate decarboxylase. AMB Expr. 2014;4(1):1–8.
Wang N, Ni Y, Shi F. Deletion of odhA or pyc improves production of gamma-aminobutyric acid and its precursor l-glutamate in recombinant Corynebacterium glutamicum. Biotechnol Lett. 2015;37(7):1473–81.
Riedel C, Rittmann D, Dangel P, Möckel B, Petersen S, Sahm H, Eikmanns BJ. Characterization of the phosphoenolpyruvate carboxykinase gene from Corynebacterium glutamicum and significance of the enzyme for growth and amino acid production. J Mol Microbiol Biotechnol. 2001;3(4):573–83.
Peters-Wendisch PG, Eikmanns BJ, Thierbach G, Bachmann B, Sahm H. Phosphoenolpyruvate carboxylase in Corynebacterium glutamicum is dispensable for growth and lysine production. FEMS Microbiol Lett. 1993;112:269–74.
Xu D, Zhao J, Cao G, Wang J, Li Q, Zheng P, Zhao S, Sun J. Removal of feedback inhibition of Corynebacterium glutamicum phosphoenolpyruvate carboxylase by addition of a short terminal peptide. Biotechnol Bioproc E. 2018;23(1):72–8.
Mao Y, Li G, Chang Z, Tao R, Cui Z, Wang Z, Tang Y-J, Chen T, Zhao X. Metabolic engineering of Corynebacterium glutamicum for efficient production of succinate from lignocellulosic hydrolysate. Biotechnol Biofuels. 2018;11(1):1–7.
Wang Z, Moslehi-Jenabian S, Solem C, Jensen PR. Increased expression of pyruvate carboxylase and biotin protein ligase increases lysine production in a biotin prototrophic Corynebacterium glutamicum strain. Eng Life Sci. 2015;15(1):73–82.
Hu J, Li Y, Zhang H, Tan Y, Wang X. Construction of a novel expression system for use in Corynebacterium glutamicum. Plasmid. 2014;75:18–26.
Zhu L, Mack C, Wirtz A, Kranz A, Polen T, Baumgart M, Bott M. Regulation of γ-aminobutyrate (GABA) utilization in Corynebacterium glutamicum by the PucR-type transcriptional regulator GabR and by alternative nitrogen and carbon sources. Front Microbiol. 2020;11:1–20.
Cho JS, Choi KR, Prabowo CPS, Shin JH, Yang D, Jang J, Lee SY. CRISPR/Cas9-coupled recombineering for metabolic engineering of Corynebacterium glutamicum. Metab Eng. 2017;42:157–67.
Soma Y, Fujiwara Y, Nakagawa T, Tsuruno K, Hanai T. Reconstruction of a metabolic regulatory network in Escherichia coli for purposeful switching from cell growth mode to production mode in direct GABA fermentation from glucose. Metab Eng. 2017;43:54–63.
Im DK, Hong J, Gu B, Sung C, Oh MK. 13C metabolic flux analysis of Escherichia coli engineered for gamma-aminobutyrate production. Biotechnol J. 2020;15(6):1900346.
Acknowledgements
This work is supported by the National Key Research and Development Program of China (2021YFC2100900) and the Key Technology Project of Inner Mongolia Autonomous Region, China (2019GG302).
Funding
Publication costs are funded by the National Key Research and Development Program of China (2021YFC2100900); the Key Technology Project of Inner Mongolia Autonomous Region, China (2019GG302).
Author information
Authors and Affiliations
Contributions
XW and CY conceived and designed research. XW, CY, YL, and XH conducted experiments. XW contributed new reagents or analytical tools. XW and CY analyzed data. CY wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Consent to participate
Not applicable.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Consent for publication
Written informed consent for publication was obtained from all participants.
Rights and permissions
About this article
Cite this article
Yao, C., Liu, Y., Hu, X. et al. Improve gamma-aminobutyric acid production in Corynebacterium glutamicum by optimizing the metabolic flux. Syst Microbiol and Biomanuf 2, 305–316 (2022). https://doi.org/10.1007/s43393-021-00062-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43393-021-00062-8