Abstract
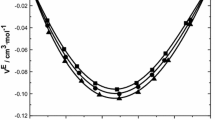
Experimental data of density have been measured for binary liquid mixtures containing {amines (n-butylamine, or s-butylamine, or t-butylamine, or diethylamine, or propylamine) + water}, over the entire range of composition at temperatures between 283.15 and 303.15 K, and atmospheric pressure. The density values enabled the determination of the thermal expansion coefficients. The excess molar volume, \({\text{V}}_{\text{m}}^{\text{E}}\), was calculated using the experimental data, from which a Redlich–Kister type polynomial was fit, enabling the determination of the partial molar volumes, the excess partial molar volumes, the apparent molar volumes and the excess partial molar volumes at infinite dilution. The \({\text{V}}_{\text{m}}^{\text{E}}\) values were also used to test the applicability of the Extended Real Associated Solution Model (ERAS Model). The results for the studied systems suggest that structural effects and chemical interactions must predominate over other possible effects. The magnitude of \({\text{V}}_{\text{m}}^{\text{E}}\) for the studies systems led to the following order: n-butylamine > propylamine > s-butylamine > diethylamine > t-butylamine.



Similar content being viewed by others
References
Acevedo IL, Katz M (1989) Molar excess volumes and partial molar excess volumes of n-butylamine with chloroalkanes at different temperatures. Thermochim Acta 156:199–209. https://doi.org/10.1016/0040-6031(89)87187-4
Almasi M, Nasim H (2015) Thermodynamic and transport properties of binary mixtures; Friction theory coupled with PC-SAFT model. J Chem Thermodyn 89:1–6. https://doi.org/10.1016/j.jct.2015.04.017
Bittencourt SS, Torres RB (2016) Volumetric properties of binary mixtures of (acetonitrile + amines) at several temperatures with application of the ERAS model. J Chem Thermodyn 93:222–241. https://doi.org/10.1016/j.jct.2015.09.002
Bondi A (1964) Van der waals volumes and radii. J Phys Chem 68:441–451. https://doi.org/10.1021/j100785a001
Bonenfant D, Mimeault M, Hausler R (2003) Determination of the structural features of distinct amines important for the absorption of CO2 and regeneration in aqueous solution. Ind Eng Chem Res 42:3179–3184. https://doi.org/10.1021/ie020738k
Chakraborty AK, Astarita G, Bischoff KB (1986) CO2 absorption in aqueous solutions of hindered amines. Chem Eng Sci 41:997–1003. https://doi.org/10.1016/0009-2509(86)87185-8
Chand A, Handa YP, Fenby DV (1975) Excess volumes of triethylamine + chloroform at 298.15 and 308.15 K. J Chem Thermodyn 7:401–402. https://doi.org/10.1016/0021-9614(75)90179-2
Checoni RF, Francesconi AZ (2005) Measurement and correlation of excess molar enthalpy at various temperatures acetonitrile + diethylamine or s-butylamine mixtures. J Therm Anal Calorim 80:295–301. https://doi.org/10.1007/s10973-005-0650-5
Concepción EI, Gómez-Hernández Á, Martín MC, Segovia JJ (2017) Density and viscosity measurements of aqueous amines at high pressures: DEA-water, DMAE-water and TEA-water mixtures. J Chem Thermodyn 112:227–239. https://doi.org/10.1016/j.jct.2017.05.001
Coulier Y, Ballerat-Busserolles K, Mesones J et al (2015) Excess Molar Enthalpies and Heat Capacities of 2-Methylpiperidine-Water and N-Methylpiperidine-Water Systems of Low to Moderate Amine Compositions. J Chem Eng Data 60:1563–1571. https://doi.org/10.1021/je5008444
Dashti A, Raji M, Alivand MS, Mohammadi AH (2020) Estimation of CO2 equilibrium absorption in aqueous solutions of commonly used amines using different computational schemes. Fuel 264:1–20. https://doi.org/10.1016/j.fuel.2019.116616
del Campos V, V., Gómez Marigliano AC, (2017) Thermodynamic study of Butylamine + Propanone, + Methyl Isobutyl Ketone (MIK) at (288.15 to 318.15) K temperature range. Chem Data Collect 11–12:96–107. https://doi.org/10.1016/j.cdc.2017.08.006
Domínguez M, Gascón I, Valén A et al (2000) Densities of (2-butanol + n-hexane + 1-butylamine) at T = 298.15 and T = 313.15 K: Excess and partial excess molar volumes and application of the ERAS model. J Chem Thermodyn 32:1551–1568. https://doi.org/10.1006/jcht.2000.0698
Duttachoudhury MK, Mathur HB (1974) Heats of mixing of butyl amine-water and butyl amine-alcohol systems. J Chem Eng Data 19:145–147. https://doi.org/10.1021/je60061a011
Egorov GI, Makarov DM (2019) Volumetric properties of the water + tetramethylurea mixture over the temperature range from 274.15 to 333.15 K at atmospheric pressure. J Mol Liq 278:279–289. https://doi.org/10.1016/j.molliq.2018.12.132
Handa YP (1977) Molar excess volumes of acetonitrile + chloroform and of acetonitrile + deuterochloroform. J Chem Thermodyn 9:117–120. https://doi.org/10.1016/0021-9614(77)90076-3
Heintz A, Dolch E, Lichtenthaler RN (1986) New experimental VLE-data for alkanol/alkane mixtures and their description by an extended real association (ERAS) model. Fluid Phase Equilib 27:61–79. https://doi.org/10.1016/0378-3812(86)87041-8
Heintz A (1985) New Theoretical Approach for Predicting Excess Properties of Alkanol/Alkane Mixtures. Berichte der Bunsengesellschaft/Physical Chem Chem Phys 89:172–181. https://doi.org/10.1002/bbpc.19850890217
Iloukhani H, Soleimani M (2017) Measurement and modeling the excess molar volumes and refractive index deviations of binary mixtures of 2-propanol, 2-butanol and 2-pentanol with N-propylamine. J Solution Chem 46:2135–2158. https://doi.org/10.1007/s10953-017-0683-y
Joint Committee for Guides in Metrology (2018) Evaluation of the measurement data – Guide to the expression of uncertainty in measurement 2008. https://www.bipm.org/en/publications/guides/gum.html. Accessed 28 Mar 2018
Kinart CM, Kinart WJ, Chȩcińska-Majak D (2002) Density, relative permittivity, and viscosity at various temperatures for 2-methoxyethanol + propylamine mixtures. J Chem Eng Data 47:1537–1539. https://doi.org/10.1021/je0201065
Kipkemboi PK, Easteal AJ (1994) Densities and viscosities of binary aqueous mixtures of nonelectrolytes: tert-butyl alcohol and tert-butylamine. Can J Chem 72:1937–1945. https://doi.org/10.1139/v94-247
Kipkemboi PK, Woolf LA (1995) PVTx Property Measurements for 2-Methyl-2-propanamine + Water from 278 to 313 K. J Chem Eng Data 40:943–947. https://doi.org/10.1021/je00020a046
Lampreia IMS, Dias FA, Mendonça AFSS (2004) Volumetric study of (diethylamine + water) mixtures between (278.15 and 308.15) K. J Chem Thermodyn 36:993–999. https://doi.org/10.1016/j.jct.2004.07.008
Lee MJ, Hwang SM, Kuo YC (1993) Densities and Viscosities of Binary Solutions Containing Butylamine, Benzylamine, and Water. J Chem Eng Data 38:577–579. https://doi.org/10.1021/je00012a026
Letcher TM, Goldon A (1996) The excess molar volumes of (n-butylamine + an ether) at the temperature 298.15 K and the application of the ERAS model. Fluid Phase Equilib 114:147–159. https://doi.org/10.1016/0378-3812(95)02817-x
Mehra R, Gaur AK (2008) Study of a binary liquid mixture of diethylamine and 1-decanol and validation of theoretical approaches of sound speed at different temperatures. J Chem Eng Data 53:863–866. https://doi.org/10.1021/je700619q
Moriyoshi T, Tsubota T, Hamaguchi K (1991) Compressions and densities of (water + a C1 to C4 n-alkylamine) at the temperature 298.15 K. J Chem Thermodyn 23:155–162. https://doi.org/10.1016/S0021-9614(05)80292-7
Muche DNF, Olivieri GV, Torres RB (2019) Density and Derived Properties of Binary Mixtures Containing {2-(Dimethylamino)ethyl Methacrylate + Alcohols} at Temperatures from T = (293.15 to 313.15) K and Pressures of up to 35 MPa. J Chem Eng Data 64:. https://doi.org/10.1021/acs.jced.8b00975
National Institute of Standards and Technology (2019) Isobaric properties for water. https://webbook.nist.gov/cgi/fluid.cgi?P=0.1&TLow=283.15&THigh=303.15&TInc=5&Applet=on&Digits=5&ID=C7732185&Action=Load&Type=IsoBar&TUnit=K&PUnit=MPa&DUnit=g%2Fml&HUnit=kJ%2Fmol&WUnit=m%2Fs&VisUnit=uPa*s&STUnit=N%2Fm&RefState=DEF. Accessed 24 May 2019
Olivieri GV, Torres RB (2019) Thermodynamic and spectroscopic study of binary mixtures containing dimethyl carbonate (DMC) + alcohols at T = (288.15–308.15) K and p = (0.1–40) MPa: Experimental study and modelling. J Chem Thermodyn. https://doi.org/10.1016/j.jct.2019.02.011
Olivieri GV, da Cunha CS, dos Santos ML et al (2018) Thermodynamic and spectroscopic study of binary mixtures of n-butylammonium oleate ionic liquid plus alcohol at T = 288.15–308.15 K. J Therm Anal Calorim. https://doi.org/10.1007/s10973-017-6801-7
Oswal SL (2005) Studies of viscosity and excess molar volume of binary mixtures: 5. Characterization of excess molar volume of 1-alkanol with alkylamines, dialkylamines and trialkylamines in terms of the ERAS model. Thermochim Acta 425:59–68. https://doi.org/10.1016/j.tca.2004.06.002
Oswal SL, Desai HS (1998) Studies of viscosity and excess molar volume of binary mixtures. 1. Propylamine + 1-alkanol mixtures at 303.15 and 313.15 K. Fluid Phase Equilib 149:359–376. https://doi.org/10.1016/S0378-3812(98)00318-5
Oswal SL, Sindhe RG, Patel AT et al (1992) Study of Viscosity of Mono-, Di-, and Trialkylamines. Int J Thermophys 13:617–628. https://doi.org/10.1007/BF00501944
Oswal SL, Oswal P, Gardas RL et al (2004) Acoustic, volumetric, compressibility and refractivity properties and reduction parameters for the ERAS and Flory models of some homologous series of amines from 298.15 to 328.15 K. Fluid Phase Equilib 216:33–45. https://doi.org/10.1016/j.fluid.2003.09.007
Pal A, Bhardwaj RK (2001) Excess molar volumes and viscosities for binary mixtures of 2-propoxyethanol and of 2-Isopropoxyethanol with propylamine and dipropylamine at (298.15, 308.15, and 318.15) K. J Chem Eng Data 46:933–938. https://doi.org/10.1021/je0100443
Pal A, Gaba R (2009) Volumetric and acoustic properties for binary mixtures of dipropylene glycol monopropyl ether with alkylamines at temperatures between 288.15 K and 308.15 K. Int J Thermophys 30:862–882. https://doi.org/10.1007/s10765-009-0593-3
Pal A, Kumar H, Sharma S et al (2010) Mixing properties for binary liquid mixtures of methyl tert-butyl ether with propylamine and dipropylamine at temperatures from (288.15 to 308.15) K. J Chem Eng Data 55:1424–1429. https://doi.org/10.1021/je900597n
Rahmatmand B, Keshavarz P, Ayatollahi S (2016) Study of Absorption Enhancement of CO2 by SiO2, Al2O3, CNT, and Fe3O4 Nanoparticles in Water and Amine Solutions. J Chem Eng Data 61:1378–1387. https://doi.org/10.1021/acs.jced.5b00442
Redlich O, Kister AT (1948) Algebraic Representation of Thermodynamic Properties and the Classification of Solutions. Ind Eng Chem 40:345–348. https://doi.org/10.1021/ie50458a036
Saleh MA, Akhtar S, Khan AR (2000) Excess molar volumes of aqueous solutions of butylamine isomers. Phys Chem Liq 38:137–149. https://doi.org/10.1080/00319100008045303
SavithaJyostna T, Satheesh B, Sreenu D et al (2020) The study of thermo-physical properties of binary liquid mixtures of isoamyl alcohol with amines at 298.15–308.15 K. Phys Chem Liq 58:349–363. https://doi.org/10.1080/00319104.2019.1594226
Scharlin P, Steinby K, Domańska U (2002) Volumetric properties of binary mixtures of N, N-dimethylformamide with water or water-d2 at temperatures from 277.13 K to 318.15 K. J Chem Thermodyn 34:927–957. https://doi.org/10.1006/jcht.2002.0946
Shakila A, Ravikumar S, Pandiyan V, Gaba R (2019) Thermodynamic properties of binary liquid mixtures containing aromatic alcohol and aliphatic amines at different temperatures. J Mol Liq 285:279–287. https://doi.org/10.1016/j.molliq.2019.04.064
Taqul Khan MM, Halligudi SB, Shukla S (1989) Solubility of carbon monoxide in water—n-butylamine, ethanol—cyclohexene, and water—dimethylformamide mixtures. J Chem Eng Data 34:353–355. https://doi.org/10.1021/je00057a026
Tôrres RB, Hoga HE (2008) Volumetric properties of binary mixtures of dichloromethane and amines at several temperatures and p = 0.1 MPa. J Mol Liq 143:17–22. https://doi.org/10.1016/j.molliq.2008.04.007
Tôrres RB, Hoga HE, Magalhães JG, Volpe PLO (2009) Volumetric properties of chloroalkanes + amines mixtures: Theoretical analysis using theERAS-Model. Int J Thermophys 30:1202–1212. https://doi.org/10.1007/s10765-009-0632-0
Tôrres RB, Francesconi AZ (2003) Modeling of excess molar volume of binary mixtures of acetonitrile with amines using the Prigogine-Flory-Patterson theory. In: Journal of Molecular Liquids. Elsevier, pp 99–110
Ullmann’s (2011) Encyclopedia of industrial chemistry, 7th edn. VCH, Weinheim
Vaid Z, More UU, Gardas RL, Malek NI, Ijardar SP (2015) Composition and temperature dependence of excess properties of binary mixtures of imidazolium based ionic liquids: II ([C n mim][PF 6 ]) + propylamine. J Solut Chem 44:718–741. https://doi.org/10.1007/s10953-015-0325-1
Zhang R, Chen J, Mi J (2015) Excess molar enthalpies for binary mixtures of different amines with water. J Chem Thermodyn 89:16–21. https://doi.org/10.1016/j.jct.2015.04.030
Funding
This work has been supported by Fundação Educacional Inaciana Padre Sabóia de Medeiros (FEI) and Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP), Grant #2004/11855–8 and Grant #2016/02652–3.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Munoz, P.A.R., Olivieri, G.V., dos Santos, R.G. et al. Experimental study and correlation of the excess molar volume of binary liquid solutions of (amines + water) at different temperatures and atmospheric pressure. Braz. J. Chem. Eng. 39, 301–318 (2022). https://doi.org/10.1007/s43153-021-00148-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43153-021-00148-9