Abstract
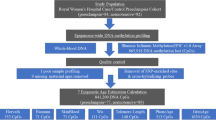
Preeclampsia is a leading cause of maternal and neonatal morbidity and mortality. Chronological age and race are associated with preeclampsia, but the role of these factors is not entirely understood. We hypothesized that DNA methylation age, a measure of biological age, would be higher in individuals with preeclampsia than in individuals with normotensive pregnancy and that DNA methylation age would differ by race across pregnancy. This was a longitudinal, exploratory study of 56 pregnant individuals (n = 28 preeclampsia cases and n = 28 normotensive controls). Genome-wide DNA methylation data were generated from trimester-specific peripheral blood samples. DNA methylation age was estimated using the “Improved Precision” clock, and ∆age, the difference between DNA methylation age and chronological age, was computed. DNA methylation age was compared with chronological age using Pearson correlations. The relationships between ∆age and preeclampsia status, self-reported race, and covariates were tested using multiple linear regression and performed both with and without consideration of cell-type heterogeneity. We observed strong correlation between chronological age and DNA methylation age across pregnancy, with significantly stronger correlation observed in White participants than in Black participants. We observed no association between ∆age and preeclampsia status. However, ∆age was higher in participants with higher pre-pregnancy body mass index in trimester 1 and lower in Black participants than in White participants in trimesters 2 and 3. Observations were largely consistent when controlling for cell-type heterogeneity. Our findings in a small sample support the need for additional studies to investigate the relationship between race and biological age, which could provide further insight into racial disparities across pregnancy. However, this study does not support an association between ∆age and preeclampsia status.


Similar content being viewed by others
Data Availability
dbGAP, accession number: phs001937.v1.p1
Code Availability
The “Improved Precision” epigenetic clock is available through publicly available source code as cited in the paper and located in GitHub at https://github.com/qzhang314/DNAm-based-age-predictor.
References
Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Screening for preeclampsia US preventive services task force recommendation statement. JAMA - J Am Med Assoc. 2017;317:1661–7.
Ananth C V, Keyes KM, Wapner RJ. Pre-eclampsia rates in the United States, 1980-2010: Age-period-cohort analysis. BMJ; 347. Epub ahead of print 7 November 2013. DOI: https://doi.org/10.1136/bmj.f6564.
Valdés G. Preeclampsia and cardiovascular disease: interconnected paths that enable detection of the subclinical stages of obstetric and cardiovascular diseases. Integrated Blood Pressure Control. 2017;10:17–23.
Hauspurg A, Countouris ME, Jeyabalan A, Hubel CA, Roberts JM, Schwarz EB, et al. Risk of hypertension and abnormal biomarkers in the first year postpartum associated with hypertensive disorders of pregnancy among overweight and obese women. Pregnancy Hypertens. 2019;15:1–6.
Tucker MJ, Berg CJ, Callaghan WM, Hsia J. The black-white disparity in pregnancy-related mortality from 5 conditions: differences in prevalence and case-fatality rates. Am J Public Health. 2007;97:247–51.
Shahul S, Tung A, Minhaj M, Nizamuddin J, Wenger J, Mahmood E, et al. Racial disparities in comorbidities, complications, and maternal and fetal outcomes in women with preeclampsia/eclampsia. Hypertens Pregnancy. 2015;34:506–15.
Tanaka M, Jaamaa G, Kaiser M, Hills E, Soim A, Zhu M, et al. Racial disparity in hypertensive disorders of pregnancy in New York state: a 10-year longitudinal population-based study. Am J Public Health. 2007;97:163–70.
Grobman WA, Parker CB, Willinger M, Wing DA, Silver RM, Wapner RJ, et al. Racial disparities in adverse pregnancy outcomes and psychosocial stress. Obstet Gynecol. 2018;131:328–35.
Tanya Nagahawatte N, Goldenberg RL. Poverty, maternal health, and adverse pregnancy outcomes. Ann N Y Acad Sci. 2008;1136:80–5.
Crear-Perry J, Correa-de-Araujo R, Lewis Johnson T, et al. Social and structural determinants of health inequities in maternal health. J Womens Health (Larchmt). Epub ahead of print November 2020. DOI: 10.1089/jwh.2020.8882.
Barcelona de Mendoza V, Huang Y, Crusto CA, et al. Perceived racial discrimination and DNA methylation among African American women in the InterGEN Study. Biol Res Nurs. 2018;20:145–52.
O’Brien R, Neman T, Seltzer N, et al. Structural racism, economic opportunity and racial health disparities: Evidence from U.S. counties. SSM - Popul Heal. 2020;11:100564.
Chambers BD, Arega HA, Arabia SE, Taylor B, Barron RG, Gates B, et al. Black women’s perspectives on structural racism across the reproductive lifespan: a conceptual framework for measurement development. Matern Child Health J. 2021;25:402–13. https://doi.org/10.1007/s10995-020-03074-3.
Sheen J-J, Huang Y, Wright JD, Goffman D, D'Alton ME, Friedman AM. 318: Maternal age and preeclampsia outcomes. Am J Obstet Gynecol. 2019;220:S222–3.
Horvath S, Raj K. DNA methylation-based biomarkers and the epigenetic clock theory of ageing. Nat Rev Genet. 2018;19:371–84.
Horvath S. DNA methylation age of human tissues and cell types. Genome Biol. 2013;14:R115.
Hannum G, Guinney J, Zhao L, Zhang L, Hughes G, Sadda SV, et al. Genome-wide methylation profiles reveal quantitative views of human aging rates. Mol Cell. 2013;49:359–67.
Zhang Q, Vallerga CL, Walker RM, Lin T, Henders AK, Montgomery GW, et al. Improved precision of epigenetic clock estimates across tissues and its implication for biological ageing. Genome Med. 2019;11:54.
Levine ME, Lu AT, Quach A, Chen BH, Assimes TL, Bandinelli S, et al. An epigenetic biomarker of aging for lifespan and healthspan. Aging (Albany NY). 2018;10:573–91.
Mayne BT, Leemaqz SY, Smith AK, Breen J, Roberts CT, Bianco-Miotto T. Accelerated placental aging in early onset preeclampsia pregnancies identified by DNA methylation. Epigenomics. 2017;9:279–89.
Wang X, Zhu H, Snieder H, Su S, Munn D, Harshfield G, et al. Obesity related methylation changes in DNA of peripheral blood leukocytes. BMC Med. 2010;8:87.
McGregor K, Bernatsky S, Colmegna I, Hudson M, Pastinen T, Labbe A, et al. An evaluation of methods correcting for cell-type heterogeneity in DNA methylation studies. Genome Biol. 2016;17:84.
Houseman EA, Accomando WP, Koestler DC, Christensen BC, Marsit CJ, Nelson HH, et al. DNA methylation arrays as surrogate measures of cell mixture distribution. BMC Bioinformatics. 2012;13:86.
Ray M, Heinsberg LW, Conley YP, et al. Utilization of epigenome-wide DNA methylation for longitudinal comparison of white blood cell proportions across preeclamptic and normotensive pregnancy by self-reported race. medRxiv. Epub ahead of print 2020. DOI: https://doi.org/10.1101/2020.09.18.20197491.
Zhang Q. DNA methylation based chronological age predictor. Epub ahead of print 2019. DOI: https://doi.org/10.5281/zenodo.3369456.
Team RC. R: A language and environment for statistical computing. https://www.r-project.org/ (2018).
Jollife IT, Cadima J. Principal component analysis: a review and recent developments. Philos Trans R Soc A Math Phys Eng Sci. 2016;374:20150202.
El Khoury LY, Gorrie-Stone T, Smart M, et al. Systematic underestimation of the epigenetic clock and age acceleration in older subjects. Genome Biol. 2019;20:283.
Hughes A, Smart M, Gorrie-Stone T, Hannon E, Mill J, Bao Y, et al. Socioeconomic position and DNA methylation age acceleration across the life course. Am J Epidemiol. 2018;187:2346–54.
Kresovich JK, Taylor JA. Re: ‘socioeconomic position and DNA methylation age acceleration across the life course’. Am J Epidemiol. 2019;188:487–8.
Hughes A, Bao Y, Smart M, Gorrie-Stone T, Hannon E, Mill J, et al. The authors reply. Am J Epidemiol. 2019;188:488–9.
Ross KM, Carroll J, Horvath S, et al. Immune epigenetic age in pregnancy and 1 year after birth: associations with weight change. Am J Reprod Immunol; 83. Epub ahead of print 12 May 2020. DOI: 10.1111/aji.13229.
Li C, Wang Z, Hardy T, et al. Association of obesity with DNA methylation age acceleration in African American mothers from the interGEN study. Int J Mol Sci; 20. Epub ahead of print 31 August 2019. DOI: 10.3390/ijms20174273.
De Toro-Martín J, Guénard F, Tchernof A, et al. Body mass index is associated with epigenetic age acceleration in the visceral adipose tissue of subjects with severe obesity. Clin Epigenetics. 2019;11:172.
Hunt SC, Chen W, Gardner JP, Kimura M, Srinivasan SR, Eckfeldt JH, et al. Leukocyte telomeres are longer in African Americans than in whites: the National Heart, Lung, and Blood Institute Family Heart Study and the Bogalusa Heart Study. Aging Cell. 2008;7:451–8.
Rewak M, Buka S, Prescott J, de Vivo I, Loucks EB, Kawachi I, et al. Race-related health disparities and biological aging: does rate of telomere shortening differ across blacks and whites? Biol Psychol. 2014;99:92–9.
Horvath S, Gurven M, Levine ME, Trumble BC, Kaplan H, Allayee H, et al. An epigenetic clock analysis of race/ethnicity, sex, and coronary heart disease. Genome Biol. 2016;17:171.
Benjamins MR, Silva A, Saiyed NS, de Maio FG. Comparison of all-cause mortality rates and inequities between Black and White populations across the 30 most populous US cities. JAMA Netw Open. 2021;4:e2032086.
Chambers BD, Arabia SE, Arega HA, Altman MR, Berkowitz R, Feuer SK, et al. Exposures to structural racism and racial discrimination among pregnant and early post-partum Black women living in Oakland, California. Stress Heal J Int Soc Investig Stress. 2020;36:213–9.
McLemore MR, Altman MR, Cooper N, et al. Health care experiences of pregnant, birthing and postnatal women of color at risk for preterm birth. Soc Sci Med. 2018;201:127–35.
Konkel L. Racial and ethnic disparities in research studies: the challenge of creating more diverse cohorts. Environ Health Perspect. 2015;123:A297–302.
Lim EM, Cembrowski G, Cembrowski M, et al. Race-specific WBC and neutrophil count reference intervals. Int J Lab Hematol. 2010;32:590–7.
Harmon AC, Cornelius DC, Amaral LM, Faulkner JL, Cunningham MW Jr, Wallace K, et al. The role of inflammation in the pathology of preeclampsia. Clin Sci (Lond). 2016;130:409–19.
Fiorito G, Polidoro S, Dugué P-A, Kivimaki M, Ponzi E, Matullo G, et al. Social adversity and epigenetic aging: a multi-cohort study on socioeconomic differences in peripheral blood DNA methylation. Sci Rep. 2017;7:16266.
Acknowledgements
We would like to thank the participants for their involvement in this research and Sandra Deslouches, laboratory manager at the University of Pittsburgh School of Nursing, for her expertise and assistance in sample preparation. We would also like to thank the anonymous reviewers who took the time to critically evaluate this paper as their feedback improved the clarity of this work.
Funding
Research reported in this publication was supported by the National Institute of Child Health and Human Development (R21HD092770 and P01HD303067), the National Center for Advancing Translational Sciences (TL1TR001858), and the National Institute of Nursing Research (T32NR009759) of the National Institutes of Health. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
Lacey W. Heinsberg contributed to the study conception and design and analysis and interpretation of the data and drafted, critically revised, and gave final approval for the manuscript. Mitali Ray contributed to the study conception and design and interpretation of the data and critically revised and gave final approval for the manuscript. Yvette P. Conley contributed to the interpretation of the data and critically revised and gave final approval for the manuscript. James M. Roberts contributed to the acquisition and interpretation of the data and critically revised and gave final approval for the manuscript. Arun Jeyabalan contributed to the acquisition and interpretation of the data and critically revised and gave final approval for the manuscript. Carl A. Hubel contributed to the acquisition and interpretation of the data and critically revised and gave final approval for the manuscript. Daniel E. Weeks contributed to the study conception and design and analysis and interpretation of the data and critically revised and gave final approval for the manuscript. Mandy J. Schmella contributed to the study conception and design and acquisition and interpretation of the data and critically revised and gave final approval for the manuscript.
Corresponding author
Ethics declarations
Ethical Approval
This study has Institutional Review Board Approval from the University of Pittsburgh (Study number 19110285, most recent renewal approved 11-25-2020).
Consent to Participate
Written informed consent was obtained from all participants at enrollment.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Heinsberg, L.W., Ray, M., Conley, Y.P. et al. An Exploratory Study of Epigenetic Age in Preeclamptic and Normotensive Pregnancy Reveals Differences by Self-Reported Race but Not Pregnancy Outcome. Reprod. Sci. 28, 3519–3528 (2021). https://doi.org/10.1007/s43032-021-00575-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-021-00575-6