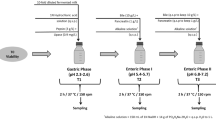
Abstract
This work covers soymilk fermentation by starter and probiotic cultures and explores the influence of cooling protocol on cell viability, organic acid production, sugar consumption, fatty acid profile, and cell survival to in vitro gastrointestinal stress. After fermentation at 37 °C by mono- or co-cultures of Streptococcus thermophilus (St), Lactobacillus bulgaricus (Lb), and Lactobacillus paracasei (Lp), fermented soymilk was cooled directly at 4 °C for 28 days or cooled in two phases (TPC), i.e., by preceding that step by another at 25 °C for 8 h. Soybean milk fermentation by Lb alone lasted longer (15 h) than by StLb or StLbLp (9 h). In ternary culture, TPC increased Lp viability, linoleic, and lactic acid concentrations by 3.8, 22.6, and 96.2%, respectively, whereas the cooling protocol did not influence Lp and St counts after in vitro gastrointestinal stress.

Graphical abstract



Similar content being viewed by others
References
Santos DC, Oliveira Filho JG, Santana ACA, Freitas BSM, Silva FG, Takeuchi KP, Egea MB (2019) Optimization of soymilk fermentation with kefir and the addition of inulin: physicochemical, sensory and technological characteristics. LWT – Food Sci and Technol 104:30–37. https://doi.org/10.1016/j.lwt.2019.01.030
Wijewardana C, Reddy RK, Bellaloui N (2019) Soybean seed physiology, quality, and chemical composition under soil moisture stress. Food Chem 278:92–100. https://doi.org/10.1016/j.foodchem.2018.11.035
Marazza JA, Le Blanc JG, de Giori GS, Garro MS (2013) Soymilk fermented with Lactobacillus rhamnosus CRL981 ameliorates hyperglycemia, lipid profiles and increases antioxidant enzyme activities in diabetic mice. J Funct Foods 5:1848–1853. https://doi.org/10.1016/j.jff.2013.09.005
Bedani R, Rossi EA, Saad SMI (2013) Impact of inulin and okara on Lactobacillis acidophilus LA-5 and Bifidobacterium animalis Bb-12 viability in fermented soy product and probiotic survival under in vitro simulated gastrointestinal conditions. Food Microbiol 34:382–389. https://doi.org/10.1016/j.fm.2013.01.012
Kuda T, Kataoka M, Nemoto M, Kawahara M, Takahashi H, Kimura B (2016) Isolation of lactic acid bacteria from plants of the coastal Satoumi regions for use as starter cultures in fermented milk and soymilk production. LWT - Food Sci Technol 68:202–207. https://doi.org/10.1016/j.lwt.2015.12.023
Aljewicz M, Siemianowska E, Cichosz G, Tonska E (2014) The effect of probiotics (Lactobacillus rhamnosus HN001, Lactobacillus paracasei LPC-37, and Lactobacillus acidophilus NCFM) on the availability of minerals from Dutch-type cheese. J Dairy Sci 97:4824–4831. https://doi.org/10.3168/jds.2014-8240
Forssten SD, Röytiö H, Hibberd AA, Ouwehand AC (2015) The effect of polydextrose and probiotic lactobacilli in a Clostridium difficile infected human colonic model. Microb Ecol Health Dis 26:27988. https://doi.org/10.3402/mehd.v26.27988
Tamime AY, Robinson RK (1999) Yoghurt: science and technology, 2nd edn. CRC Press, Cambridge
Wang W, De Mejia EG (2005) A new frontier in soy bioactive peptides that may prevent age-related chronic diseases. Compr Rev Food Sci Food Saf 4:63–78. https://doi.org/10.1111/j.1541-4337.2005.tb00075.x
Wouters JA, Mailhes M, Rombouts FM, de Vos WM, Kuipers OP, Abee T (2000) Physiological and regulatory effects of controlled overproduction of five cold shock proteins of Lactococcus lactis MG1363. Appl Environ Microbiol 66:3756–3763. https://doi.org/10.1128/aem.66.9.3756-3763.2000
Oliveira RPS, Torres BR, Perego P, Oliveira MN, Converti A (2012) Co-metabolic models of Streptococcus thermophilus in co-culture with Lactobacillus bulgaricus or Lactobacillus acidophilus. Biochem Eng J 62:62–69. https://doi.org/10.1016/j.bej.2012.01.004
Ong L, Henriksson A, Shah NP (2006) Development of probiotic Cheddar cheese containing Lactobacillus acidophilus, Lb. casei, Lb. paracasei and Bifidobacterium spp. and the influence of these bacteria on proteolytic patterns and production of organic acid. Int Dairy J 16:446–456. https://doi.org/10.1016/j.idairyj.2005.05.008
Bleigh EG, Dyer WJ (1959) A rapid method for total lipid extraction and purification. Can J Biochem Physiol 37:911–917. https://doi.org/10.1139/o59-099
ISO (2002) Milk fat-preparation of fatty acid methyl esters (Standard ISO 15884/IDF 182). International Organization for Standardization, Geneva
Florence ACR, Béal C, da Silva RC, Oliveira MN (2014) Survival of three Bifidobacterium animalis subsp. lactis strains is related to trans-vaccenic and α-linolenic acids contents in organic fermented milks. LWT - Food Sci Technol 56:290–295. https://doi.org/10.1016/j.lwt.2013.11.036
AOCS (1997) Official method Ce 1-62: fatty acid composition by gas chromatography. Official Methods and Recommended Practices of the AOCS. Champaign, IL, USA: American Oil Chemist’s Society. Accessed 13 March 2020
Donkor ON, Henriksson A, Vasiljevic T, Shah NP (2007) α-Galactosidase and proteolytic activities of selected probiotic and dairy cultures in fermented soymilk. Food Chem 104:10–20. https://doi.org/10.1016/j.foodchem.2006.10.065
Lalou S, El Kadri H, Gkatzionis K (2017) Incorporation of water-in-oil-in-water (W1/O/W2) double emulsion in a set type yogurt model. Food Res Int 100:122–131. https://doi.org/10.1016/j.foodres.2017.08.027
Lazaridou A, Serafeimidou A, Biliaderis CG, Moschakis T, Tzanetakis N (2014) Structure development and acidification kinetics in fermented milk containing oat β-glucan, a yogurt culture and a probiotic strain. Food Hydrocoll 39:204–214. https://doi.org/10.1016/j.foodhyd.2014.01.015
Florence ACR, Béal C, da Silva RC, Bogsan CSB, Pilleggi ALOS, Gioielli LA, Oliveira MN (2012) Fatty acid profile, trans-octadecenoic, a-linolenic and conjugated linoleic acid contents differing in certified organic and conventional probiotic fermented milks. Food Chem 135:2207–2214. https://doi.org/10.1016/j.foodchem.2012.07.026
El Kadri H, Lalou S, Mantzouridou F, Gkatzionis K (2018) Utilisation of water-in-oil-water (W1/O/W2) double emulsion in a set-typeyogurt model for the delivery of probiotic Lactobacillus paracasei. Food Res Int 107:325–336. https://doi.org/10.1016/j.foodres.2018.02.049
Zavaglia AG, Disalvo EA, De Antoni GL (2000) Fatty acid composition and freeze–thaw resistance in lactobacilli. J Dairy Res 67:241–247. https://doi.org/10.1017/s0022029900004179
Reale A, Di Renzo T, Rossi F, Zotta T, Iacumin L, Preziuso M, Parente E, Sorrentino E, Coppola R (2015) Tolerance of Lactobacillus casei, Lactobacillus paracasei and Lactobacillus rhamnosus strains to stress factors encountered in food processing and in the gastro-intestinal tract. LWT - Food Sci Technol 60:721–728. https://doi.org/10.1016/j.lwt.2014.10.022
Donkor ON, Henriksson A, Vasiljevic T, Shah NP (2005) Probiotic strains as starter cultures improve angiotensin-converting enzyme inhibitory activity in soy yogurt. J Food Sci 70:M375–M381. https://doi.org/10.1111/j.1365-2621.2005.tb11522.x
Wang Y-C, Yu R-C, Yang H-Y, Chou C-C (2003) Sugar and acid contents in soymilk fermented with lactic acid bacteria alone or simultaneously with bifidobacteria. Food Microbiol 20:333–338. https://doi.org/10.1016/S0740-0020(02)00125-9
Peñalvo JL, Castilho MC, Silveira MIN, Matallana MC, Torija ME (2004) Fatty acid profile of traditional soymilk. Eur Food Res Technol 219:251–253. https://doi.org/10.1007/s00217-004-0945-y
Lee JH, Kim B, Hwang CE, Haque MA, Kim SC, Lee CS, Kang SS, Cho KM, Lee DH (2018) Changes in conjugated linoleic acid and isoflavone contents from fermented soymilks using Lactobacillus plantarum P1201 and screening for their digestive enzyme inhibition and antioxidant properties. J Funct Foods 43:17–28. https://doi.org/10.1016/j.jff.2018.01.022
Bengoa AA, Llamas MG, Iraporda C, Dueñas MT, Abraham AG, Garrote GL (2018) Impact of growth temperature on exopolysaccharide production and probiotic properties of Lactobacillus paracasei strains isolated from kefir grains. Food Microbiol 69:212–218. https://doi.org/10.1016/j.fm.2017.08.012
Uriot O, Galia W, Awussi AA, Perrin C, Denis S, Chalancon S, Lorson E, Poirson C, Junjua M, Le Roux Y, Alric M, Dary A, Blanquet-Diot S, Roussel Y (2016) Use of the dynamic gastro-intestinal model TIM to explore the survival of the yogurt bacterium Streptococcus thermophilus and the metabolic activities induced in the simulated human gut. Food Microbiol 53:18–29. https://doi.org/10.1016/j.fm.2015.05.007
Hou RCW, Lin MY, Wang MMC, Tzen JTC (2003) Increase of viability of entrapped cells of Lactobacillus delbrueckii ssp. bulgaricus in artificial sesame oil emulsions. J Dairy Sci 86:424–428. https://doi.org/10.3168/jds.S0022-0302(03)73620-0
Acknowledgments
The authors thank Danisco-DuPont for the bacterial cultures.
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001, the National Council for Scientific and Technological Development (CNPq), and the São Paulo Research Foundation (FAPESP process no. 2019/19054-0).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: Rosane Freitas Schwan
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Piazentin, A.C.M., da Silva, T.M.S., Florence-Franco, A.C. et al. Soymilk fermentation: effect of cooling protocol on cell viability during storage and in vitro gastrointestinal stress. Braz J Microbiol 51, 1645–1654 (2020). https://doi.org/10.1007/s42770-020-00369-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42770-020-00369-z