Abstract
Background and Objective
Until 2009, only reusable bronchoscopes were marketed, but the introduction and widespread adoption of single-use flexible bronchoscopes (SFBs) as an emerging technology has since accelerated. Several studies have described the costs of reusable flexible bronchoscopes (RFBs) and SFBs. This meta-analysis aimed to compile the current published evidence to analyse the cost of different scenarios using RFBs and SFBs.
Methods
All published literature describing the cost of RFBs or SFBs was identified by searching PubMed, Embase and Google Scholar, limited to those between 1 January, 2009 and 6 November, 2020. Included studies should report the total cost of RFBs. Continuous data were extracted for relevant outcomes and analysed using RStudio® 4.0.3 as the standardised mean difference and standard error of the mean in a mixed-effects model. Risk of bias was assessed based on the reporting quality.
Results
In the systematic literature review, 342 studies were initially identified, and 11 were included in the final analysis. The mean RFB procedure cost was $266 (standard error of the mean: 34), including capital investments, repairs and reprocessing costs of $91, $92 and $83, respectively. The mean SFB procedure cost was $289 (standard error of the mean: 10). The incremental cost was $23 (standard error of the mean: 33) and was not significant (p = 0.46). Because of the economy of scale, RFB is more likely to be cost minimising compared with SFB when performing 306 or 39 procedures per site or RFB, respectively.
Conclusions
In this study, we found no significant difference in the cost of use between RFBs and SFBs and a high risk of bias.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The included studies yielded a mean reusable flexible bronchoscope procedure cost of $266. The mean single-use flexible bronchoscope procedure cost was $289. No significant cost difference between single-use flexible bronchoscopes and reusable flexible bronchoscopes was found. |
Depending on the procedure volume, the break-even point was 306 procedures per site annually and 39 annually per reusable flexible bronchoscope. If a site performs less than 306 procedures annually and 39 procedures per reusable flexible bronchoscope, a single-use flexible bronchoscope is more likely to be cost minimising. |
1 Introduction
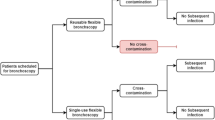
In the past 5 decades, bronchoscopy has been commonly used to examine the lungs in the intensive care unit, operating room, emergency room and the bronchoscopy suite. A reusable flexible bronchoscope (RFB) is the most commonly used device but has a complex design composition with internal channels that have proven to be challenging to decontaminate after use [1, 2]. Difficulties in decontamination have led to multiple outbreaks where post-procedure cross-infections have occurred [2, 3]. The risk of infection does not only affect patient safety but becomes more costly because of the treatment of the infection [4].
Clinical societies and health authorities such as the European Society of Gastrointestinal Endoscopy and the Centers for Disease Control and Prevention have been dedicated to inform pulmonologists and chest physicians about the importance of detailed and correct reprocessing and the societies recommended adding an additional sterilisation step to reduce the risk of cross-infection [5,6,7]. The additional sterilisation step is costly and time consuming and requires additional capital investments for hospitals [4]. Multiple cost evaluations have been conducted to create an understanding of the cost associated with using bronchoscopes, where the cost of RFBs is typically divided into the categories of capital, repair and reprocessing [4, 8, 9]. In 2009, the first single-use flexible bronchoscope (SFB) was introduced as an alternative technology to the RFB while simultaneously making reprocessing and repairs redundant and potentially offering a clinical advantage by eliminating cross-infection caused by the RFB [4].
Based on published evidence related to the performance of SFBs and RFBs, the two technologies are equivalent [10, 11]. Therefore, it is useful to assess the cost differential between SFBs and RFBs considering the similarities in the technology, performance and settings. The aim of this study is to estimate the average cost of RFBs and SFBs by compiling data from published evidence.
2 Methods
2.1 Study Selection
A systematic literature search was conducted to identify evidence investigating the cost associated with RFBs and SFBs. The literature search and subsequent reporting were based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [12]. Studies were identified, in MEDLINE, Embase® and Google Scholar databases, from 1 January, 2009 until 6 November, 2020. The literature search was limited to studies published after 2009 because the first SFB was introduced only in 2009 [13].
The search included the following medical subject headings (MeSH): (Bronchoscop* [All Fields] AND analysis, cost [MeSH Terms]). In addition, truncation was deployed after “bronchoscop” to cover different variations of the term. The full search string from PubMed can be found in Online Appendix 1.
2.2 Inclusion and Exclusion Criteria
The literature search was conducted to identify all relevant case reports, randomised controlled trials and cohort studies assessing the total incremental cost associated with RFBs and SFBs. The studies considered for inclusion needed to state either (1) the total cost of RFBs, including capital investments, repair costs and costs related to reprocessing or (2) the total cost with a description of the included cost. If nothing else was stated in the studies, the capital investment costs were considered to include the bronchoscopes and stack systems; the repair costs included repair and maintenance costs of the capital equipment; and the reprocessing costs included the reprocessing equipment, personal protective equipments, detergents and time tracking in accordance with standard reprocessing guidelines [14, 15]. Studies did not have to state the cost of SFBs to be included. Two authors independently reviewed the titles and abstracts. Studies that did not fulfil the above-mentioned criteria were excluded from the analysis. Full-text reviews were performed by all authors. Any disagreements were resolved by consensus.
2.3 Data Extraction
None of the authors was blinded to any information related to the studies. For each study, the following information was extracted where applicable: author, year of publication, price date, country, the total cost in the stated valuta, number of RFBs, RFB procedures per year, procedure setting, reprocessing set-up and SFB cost, if any.
2.4 Outcomes
The primary outcome is shown as the total incremental direct procedure cost of RFBs compared to SFBs shown in Quarter 1-2022 US Dollars (USD). The RFB direct cost of use is segmented into capital, repair and reprocessing costs. The SFB cost is based on the stated cost in the included publications. All calculations from the local currency and earlier price dates are projected using the consumer price indices database from the Organisation for Economic Co-operation and Development Statistics and then converted to Quarter 1-2022 USD. Conversion rates used were 1.11, 1.25 and 0.74 from Euro, GBP and Singapore Dollars to USD, respectively [16].
2.5 Data Analysis and Statistical Methods
To assess the reporting quality and risk of bias of the costing studies, each study was categorised depending on whether they followed the Consolidated Health Economic Evaluation Reporting Standards (CHEERS) items relevant for costing studies, see Online Appendix 2. The relevant CHEERS items were selected by the authors via consensus. All statistical analyses were performed in RStudio® 4.0.3 [17]. Missing data were handled via imputation of mean values.
The significance level was set as p < 0.05. All results are expressed as the mean and standard error of the mean (SEM) where applicable. The incremental cost between SFBs and RFBs was investigated via a mixed-effect model with the bronchoscope type as the fixed effect and the study ID as the random effect. General linear regression was utilised to investigate the effect of country, setting, reprocessing set-up or procedure volume on the incremental cost.
Monte Carlo simulations with 10,000 iterations were utilised to investigate the probability of SFBs being cost minimising compared to RFBs in the base-case analysis dependent on the procedure volume per site and procedure volume per RFB. Furthermore, the linear function of these simulations was utilised to calculate the break-even point dependent on procedure volume via simple linear algebra. A sensitivity analysis was conducted including both the full-text studies from the base-case analysis and abstracts with the same eligibility criteria.
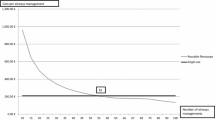
The incremental cost of use and break-even point dependent on procedure volume was graphically illustrated along with iterations and 95% confidence interval, respectively. All extracted data from the included studies were summarised for the base-case and sensitivity analysis in a table along with the mean cost of RFBs and SFBs.
3 Results
A total of 342 studies were identified, of which 28 duplicates were removed. The remaining 314 studies were screened based on the title and abstract and narrowed down to 36 studies. Of these, 11 full-text studies were included in the base-case analysis based on the inclusion criteria. Eight studies also included a cost for SFBs and none of the included studies complied with the CHEERS items. A PRISMA flowchart showing the selection process is shown in Fig. 1. The included studies yielded a base-case mean RFB procedure cost of $266 (SEM: 34), consisting of $91 (SEM: 19), $92 (SEM: 15) and $83 (SEM: 14) for capital investments, repair costs and reprocessing costs, respectively. The base-case means SFB procedure cost was $289 (SEM: 10), given a mean incremental cost of $23 (SEM: 33) per procedure with SFB. The sensitivity analysis included 25 studies of which 14 were non-peer reviewed studies. The sensitivity analysis resulted in a mean RFB procedure cost of $306 (SEM: 35), consisting of $105 (SEM: 15), $90 (SEM: 11) and $111 (SEM: 20) for capital investments, repair costs and reprocessing costs, respectively. The sensitivity analysis mean SFB procedure cost was $278 (SEM: 7) giving a mean incremental cost of −$28 (SEM: 34) per procedure with SFB, see Table 1.
There was no significant cost difference between SFBs and RFBs found in the base-case [p = 0.46, BIC = 250.91, α = 265 (SEM: 25)], β = 23 (SEM: 33)] or sensitivity analysis [p = 0.41, BIC = 622.39, α = 306 (SEM: 25), β = −28 (SEM: 34)]. The base-case mean procedure volume was 515 (SEM: 159) and 51 (SEM: 11) per site and RFB, respectively. The base-case incremental cost was significantly dependent on annual procedure volumes per site [p = 0.01, BIC = 267.67, α = −35 (SEM: 28), β = 0.11 (SEM: 0.04)], and procedure volume per RFB (p < 0.01, BIC = 262.65, α = −78 (SEM: 31), β = 2 (SEM: 0.5)] (Figs. 2, 3).
The incremental cost per use was not significantly dependent on reprocessing set-up, clinical setting or country (p > 0.05). The probability of SFBs being cost minimising compared to RFBs was 36% in the base case and 51% in the sensitivity analysis.
The base-case break-even point depending on procedure volume was found at 306 procedures per site annually and 39 annually per RFB (Figs. 4, 5). The incremental cost in the sensitivity analysis was also significantly dependent on the annual procedure volume and procedure volume per RFB, where the break-even points were 713 procedures per site annually and 61 annual procedures per RFB.
4 Discussion
Recently, studies have been published to elucidate the incremental cost of RFBs, costs related to the change from RFBs to SFBs and the organisational benefits of changing to SFBs [4, 8, 19]. In this study, the incremental cost was estimated based on the best available evidence via a systematic literature review and meta-analysis. To the authors’ knowledge, this is the first meta-analysis of per-procedure costs, including all studies investigating the cost difference between SFBs and RFBs. This analysis demonstrated that the mean procedure cost of SFBs was $23 higher than RFBs in the base case, whereas the mean procedure cost was $28 lower per procedure with SFBs in the sensitivity analysis. The incremental cost per procedure between SFBs and RFBs in the base-case and sensitivity analysis, at various settings, countries and reprocessing set-up was not significant. The incremental cost of SFBs and RFBs was significantly dependent on procedure volume per RFB and the annual procedure volume per site, with break-even points at 39 annual procedures per RFB and 306 annual procedures per department owing to the economy of scale [41]. However, the break-even points for both annual procedures per RFB and the annual procedure volume were approximately twice as high in the sensitivity analysis. This estimation enables individual hospitals to determine whether RFBs or SFBs are advantageous from a cost perspective, depending on the size, procedure volume and procedures per RFB.
The cost of reprocessing accounted for approximately 31% of the total per procedure cost of RFBs. A large SEM between the studies indicates uncertainty surrounding the cost of reprocessing that may be a result of (1) studies reporting on reprocessing costs based on different reprocessing guidelines and (2) the complexity of guidelines, including the many steps that may be difficult to comply with, resulting in varying reprocessing methods [9]. The reprocessing cost also had the most prominent variation between the base-case and sensitivity analyses at $29 per procedure. The higher reprocessing cost is likely because four of the abstracts in the sensitivity analysis had high reprocessing costs compared with the full texts, driving the overall reprocessing cost further. The highest reprocessing cost in the sensitivity analysis was $470, while it was $169 in the base case. Given the lack of information in the abstracts, all the included resources and costs are unknown, decreasing the transparency and making the comparison between the studies more difficult. However, higher reprocessing costs in the abstracts might be because of more resources being included, or the costs of the resources being higher than in the full-text studies.
Capital costs and repair costs both accounted for ~34% of the total per procedure costs of RFB. The variation in capital costs between studies may result from different annuitisation and discounting methods of capital equipment, amongst others. In contrast, repair costs may differ depending on the type of bronchoscope as fiberoptic bronchoscopes are reported to have a higher breakage ratio than video bronchoscopes [42], and the different experiences of the clinicians may affect the number of repairs owing to more breaks by junior clinicians [19, 25]. As repairs and reprocessing are avoided with SFBs, the SFB cost was not expected to vary remarkably between studies.
The overall variation across cost categories may result from a difference in micro-costing assessments and most of the included studies failed to report on all the relevant CHEERS items for costing studies [43]. Lack of transparency in the included studies made it difficult to assess the quality and the applied methodology. However, including non-peer-reviewed evidence in the sensitivity analysis did not alter the results significantly.
Even though this is the first meta-analysis investigating the costs of RFBs and SFBs, a systematic review conducted in 2019 has also analysed and compared the costs of RFBs and SFBs. In that review, neither included the repair cost of RFBs nor performed a statistical analysis as performed in this meta-analysis. The authors assessed the repair cost via a survey and found that the percutaneous dilatory tracheostomy procedure cost was higher than the procedure cost for intubation [44]. It makes it impossible to know whether the cost difference between RFBs and SFBs was statistically significant. The outcome of the systematic review from 2019 revealed a cost per procedure with RFBs at $135 and $123 for capital and reprocessing costs, respectively. Hence, the cost per procedure with RFBs was $258 without repair costs, while the cost per procedure with SFBs was $249 [44]. The costs of RFBs and SFBs in the systematic review from 2019 were then more aligned with the sensitivity analysis in this meta-analysis. This alignment was expected as the review contained the same studies as this meta-analysis, including four non-full-text studies. However, the number of included studies differed between the review and this meta-analysis, where this meta-analysis included 25 studies and the review included 11 studies. The difference in the number of studies might be attributed to the publication years included in the literature search, where the review limited the search from 2012 to 2017 and the current meta-analysis from 2009 to 2020, as the first SFB was launched in 2009 [44].
4.1 Limitations
The quality and the lack of method transparency within the included studies are major limitations of this meta-analysis. Varying reporting on relevant CHEERS items makes it difficult to assess the costing quality and consistency in methodology. The included studies may therefore be of varying quality, and utilise heterogenous costing methodology. However, the CHEERS items are intended for assessing reporting quality of full economic evaluations and not intended to assess methodological quality for a costing analysis [45].
On the contrary, this meta-analysis included all studies investigating the costs of RFBs and SFBs, considering that a systematic literature search in various databases and Google Scholar was performed, potentially decreasing the risk of publication bias. Additionally, the included studies did not describe whether costs were collected in a private or public setting, which might affect the used resources and costs.
Second, only capital, repair and reprocessing costs of RFBs, and capital costs and item costs of SFBs were included. It remains uncertain if costs of storage, waste and implementation differ between RFBs and SFBs, as these costs were not assessed in the included studies. Other parameters such as adverse events, infection risk, quality of the bronchoscopes and workflow were also not included even though they may have influenced the choice and use of bronchoscopes [2,3,4, 8, 46].
Third, we used a mixed-effects model, as a mix of fixed-effect and random-effect methods indicates that the studies are a mix of homogenous and heterogenous studies [47]. However, considering the risk of methodological heterogeneity between the included studies in the various quality, different resources, and costs included and distributed into the cost categories, it can be questioned whether such different studies can be combined in a mixed-effect model [48]. Nonetheless, the heterogeneity was assessed through a meta-regression analysis. Considering the similarity in the purpose of the included studies as an estimate and the comparison of the capital, repair and reprocessing cost of RFBs and SFBs, the mixed-effect model was used with the study ID as the random effect.
Overall, the included studies lacked transparency, which decreased the quality of the studies and the credibility of their outcomes. Therefore, better quality studies or more systematic reporting should be conducted to make an informed decision regarding the cost of RFBs and SFBs to investigate whether SFBs are cheaper or more expensive per procedure. Furthermore, when it comes to the choice of bronchoscopes, more aspects need to be investigated, such as organisational impact and the inclusion of an effect measure as in a cost-effectiveness analysis or cost-utility analysis, as it affects the choice of the bronchoscope.
5 Conclusions
The outcome of our cost model demonstrates a mean cost difference between RFBs and SFBs of $23. However, the cost difference was non-significant in the base-case and sensitivity analysis, between settings, and between counties, even though the cost varied considerably across the included studies.
References
Botana-Rial M, et al. A pseudo-outbreak of Pseudomonas putida and Stenotrophomonas maltophilia in a bronchoscopy unit. Respiration. 2016;92(4):274–8. https://doi.org/10.1159/000449137.
Kovaleva J, et al. Transmission of infection by flexible gastrointestinal endoscopy and bronchoscopy. Clin Microbiol Rev. 2013;26(2):230–53. https://doi.org/10.1128/CMR.00085-12.
Mehta AC, Muscarella LF. Bronchoscope-related ‘superbug’ infections. Chest. 2020;157(2):454–69. https://doi.org/10.1016/j.chest.2019.08.003.
Mouritsen JM, Ehlers LH, Kovaleva J, Ahmad I, El-Boghdadly K, El-Boghdadly K. A systematic review and cost effectiveness analysis of reusable vs. single-use flexible bronchoscopes. Anaesthesia. 2019;75(4):anae.14891. https://doi.org/10.1111/anae.14891.
Society of Gastroenterology Nurses and Associates Inc: Standards of infection control in reprocessing of flexible gastrointestinal endoscopes. Gastroenterol Nurs. 2013;36:293–303. https://doi.org/10.1097/SGA.0b013e31829c6d5b.
Rutala WA, Weber DJ. Gastrointestinal endoscopes a need to shift from disinfection to sterilization? JAMA. 2014;312:1405–1406. https://doi.org/10.1001/jama.2014.12559.
Mughal MM, Minai OA, Culver DA, Mehta AC. Reprocessing the bronchoscope: the challenges. Semin Respir Crit Care Med. 2004;25(4):443–9. https://doi.org/10.1055/s-2004-832717.
Châteauvieux C, Farah L, Guérot E, Wermert D, Pineau J, Prognon P, Borget I, Martelli N. Single-use flexible bronchoscopes compared with reusable bronchoscopes: positive organizational impact but a costly solution. J Eval Clin Pract. 2018;24(3):528–35. https://doi.org/10.1111/jep.12904.
Ofstead CL, et al. A glimpse at the true cost of reprocessing endoscopes: results of a pilot project. Int Assoc Healthc Cent Serv Mater Manag. 2017:62–78.
Kriege M, et al. Evaluation of intubation and intensive care use of the new Ambu® aScope™ 4 broncho and Ambu® aView™ compared to a customary flexible endoscope a multicentre prospective, non-interventional study. Trends Anaesth Crit Care. 2020;31:35–41. https://doi.org/10.1016/j.tacc.2020.02.001.
Flandes J, et al. Bronchoscopist’s perception of the quality of the single-use bronchoscope (Ambu aScope4™) in selected bronchoscopies: a multicenter study in 21 Spanish pulmonology services. Respir Res. 2020;21(1):320. https://doi.org/10.1186/s12931-020-01576-w.
Page M, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372: n71. https://doi.org/10.1136/bmj.n71.
Travis HS, Russell RV, Adamsen S, Larsen NB, Larsen S. Early cost-utility analysis comparing the sterile single-use Ambu® aScopeTM Duodeno to reusable duodenoscopes. SSRN Electron J. 2020. https://doi.org/10.2139/ssrn.3732784.
ANSI AAMI ST91 | Flexible and semi-rigid endoscope processing | AAMI. https://www.aami.org/st91;Stand:10.08.2022.
Ulrike Beilenhoff A, et al. Reprocessing of flexible endoscopes and endoscopic accessories used in gastrointestinal endoscopy: position statement of the European Society of Gastrointestinal Endoscopy (ESGE) and European Society of Gastroenterology Nurses and Associates (ESGENA)-Upda. Endoscopy. 2018;50:1205–34. https://doi.org/10.1055/a-0759-1629.
OECD Data. Inflation (CPI). https://data.oecd.org/price/inflation-cpi.htm. Accessed 31 Jan 2022.
RStudio. RStudio 4.0.3. 2020.
Gupta D, Wang H. Cost-effectiveness analysis of flexible optical scopes for tracheal intubation: a descriptive comparative study of reusable and single-use scopes. J Clin Anesth. 2011;23(8):632–5. https://doi.org/10.1016/j.jclinane.2011.04.007.
Tvede MF, Kristensen MS, Nyhus-Andreasen M. A cost analysis of reusable and disposable flexible optical scopes for intubation. Acta Anaesthesiol Scand. 2012;56(5):577–84. https://doi.org/10.1111/j.1399-6576.2012.02653.x.
Liu SS, Brodsky JB, Macario A. Cost identification analysis of anesthesia fiberscope use for tracheal intubation. J Anesth Clin Res. 2012;3(5):3–6. https://doi.org/10.4172/2155-6148.1000215.
Marshall DC, et al. Experience with the use of single-use disposable bronchoscope in the ICU in a tertiary referral center of Singapore. J Bronchol Interv Pulmonol. 2017;24(2):136–43. https://doi.org/10.1097/LBR.0000000000000335.
Perbet S, Constantin J-M, Bazin J-E. The single-use endoscope ascope™ for fibreoptical monitoring in percutaneous dilatational tracheostomy: a feasibility study. Intensive Care Med. 2011;37:S225.
Videau M, Rghioui K, Mottet B, Sainfort A, Lefort I. Analyse comparative de coût entre les fibroscopes bronchiques à usage unique et réutilisables: le fibroscope à usage unique, est-ce que ça vaut le coût ? Ann Pharm. 2017;75(6):473–9. https://doi.org/10.1016/j.pharma.2017.07.004.
Aïssou M, et al. Analyse de coût comparant les fibroscopes à usage unique (Ambu®aScope™) et les fibroscopes réutilisables pour l’intubation difficile. Ann Fr Anesth Reanim. 2013;32(5):291–5. https://doi.org/10.1016/j.annfar.2013.01.014.
McCahon RA, Whynes DK. Cost comparison of re-usable and single-use fibrescopes in a large English teaching hospital. Anaesthesia. 2015;70(6):699–706. https://doi.org/10.1111/anae.13011.
Larsen S, Holm JH, Sauer TN, Andersen C. A cost-effectiveness analysis comparing the VivaSight double-lumen tube and a conventional double-lumen tube in adult patients undergoing thoracic surgery involving one-lung ventilation. Pharmacoeconomics Open. 2020;4(1):159–69. https://doi.org/10.1007/s41669-019-0163-y.
Bertrand A, Lefrançois A, Saurel N. Etude de cout en faveur du fibroscope á usage unique. In: Europharmat. 2013.
Debraine C, Foy G, Touratier S, Faure P, Levert H. Fibroscope réutilisable versus usage unique : analyse de coûts en réanimation. 2016. http://www.google.dk/url?sa=t&rct=j&q=&esrc=s&source=web&cd=1&cad=rja&uact=8&ved=0ahUKEwiw0cnN4v3ZAhUFJ1AKHTE8BpYQFggoMAA&url=http%3A%2F%2Fvisualisation.ambu.com%2FAdmin%2FPublic%2FDWSDownload.aspx%3FFile%3D%252FFiles%252FFiler%252FFR%252FClinical%2Bstudi. Accessed 19 Jul 2022.
Barth E, Zujalovic B, Schilling M. Kostenevaluation und sensitivitätsanalyse zum einsatz von einweg und mehrweg. Bronchoskopen am Beispiel einer Intensivstation. 2015;17:2515591.
Sorli SC, et al. Etude de coût des fibroscopes réutilisables vs jetables en réanimation. In: Europharmat. 2015.
Wojcik A, et al. Medico-economic benefit from replacing reusable bronchoscopes with single-use versions: A microcosting evaluation. In: 16th World Sterilization Congress ans Annual conference of AFS. 2015.
Dhonneur G, Bazin J-E, Haouache H, Diemunsch P, Koffel C, Meistelman C. Étude comparative prospective et multicentrique des performances de aScopeTM 3 d’Ambu® en réanimation: une analyse intermédiaire. Anesth Réanim. 2015;1:A268–9. https://doi.org/10.1016/j.anrea.2015.07.411.
Ofstead CL, et al. Managing Bronchoscope quality and cost. Results of a real-world study. Hot Top. 2019.
ISPOR. Introducing budget impact analysis comparing reusable to single-use bronchoscopes within a large UK university hospital. https://www.ispor.org/heor-resources/presentations-database/presentation/euro2019-3122/97252. Accessed 17 Oct 2019.
Robert J, et al. Etude de minimasation des couts des video endoscopes réutilisables et à usage unique évolution des dépenses selon le nombred ’ examens? In: Pharmat. 2017.
Bertucat V, et al. Quelle place pour les fibroscopes à usage unique en anesthésie ? In: Pharmat. 2017.
Fournier C, Amrani A, Laigneau C, Clapeau G. Étude médico-économique des fibroscopes à usage multiple versus usage unique dans un service de réanimation. Le Pharm Hosp Clin. 2016;51(4):348. https://doi.org/10.1016/j.phclin.2016.10.021.
Bazin J-E, et al. Reusable versus single use fiberscope in the ICU: a medico-economical evaluation in the ICU. Ann Intensiv Care. 2016;7(1):91.
Valentine D. Thèse: „POUR L’OBTENTION DU DIPLÔME D’ÉTAT DE DOCTEUR EN PHARMACIE“. 2018.
Yassin MH, Hariri R, Hamad Y, Ferrelli J, McKibben L, Doi Y. Disposable bronchoscope model for simulating endoscopic reprocessing and surveillance cultures. Infect Control Hosp Epidemiol. 2017;38(2):136–42. https://doi.org/10.1017/ice.2016.264.
Drummond MF, et al. Methods for the Economic Evaluation of Health Care Programmes. Oxford University Press. 2015.
Choure AJ, Manali ED, Krizmanich G, Gildea TR, Mehta AC. High price of bronchoscopy: cost of maintenance and repair of flexible bronchoscopes. Can Nurse. 2005;101(6):6–7.
Husereau D, Drummond M, Petrou S, Carswell C, Moher D, Greenberg D, et al. CHEERS: consolidated health economic evaluation reporting\standards. Eur J Health Econ. 2013;14(3):367–72.
Sohrt A, Ehlers L, Udsen FW, Mærkedahl A, McGrath BA. Cost comparison of single-use versus reusable bronchoscopes used for percutaneous dilatational tracheostomy. Pharmacoeconomics Open. 2019;3(2):189–95. https://doi.org/10.1007/s41669-018-0091-2.
Husereau D, et al. Consolidated Health Economic Evaluation Reporting Standards (CHEERS) 2022 explanation and elaboration: a report of the ISPOR CHEERS II Good Practices Task Force. Value Health. 2022;25(1):10–31. https://doi.org/10.1016/j.jval.2021.10.008.
Ofstead CL, et al. Effectiveness of reprocessing for flexible bronchoscopes and endobronchial ultrasound Bbonchoscopes. Chest. 2018;154(5):1024–34. https://doi.org/10.1016/j.chest.2018.04.045.
Haidich A. Meta-analysis in medical research. Br Med J. 1944;1(4346):572. https://doi.org/10.1136/bmj.1.4346.572-a.
Rao G, et al. Methodological standards for meta-analyses and qualitative systematic reviews of cardiac prevention and treatment studies a scientific statement from the American Heart Association. Circulation. 2017;136(10):e172–94. https://doi.org/10.1161/CIR.0000000000000523.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The authors did not receive any specific funding for this research from any funding agency in the public, commercial or not-for-profit sectors.
Conflicts of interest/competing interests
Helena Travis and Rasmus Russell are employed by Ambu A/S, Ballerup Denmark. Carina Østervig Andersen and Emile Dehlholm-Lambertsen are employed by Ambu Ltd., Cambridgeshire, UK. Emmelie Ploug Jørgensen has no conflicts of interest that are directly relevant to the content of this article.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Ethics approval
Not applicable.
Availability of data and material
All data generated or analysed during this study are included in this published article (and its supplementary information files).
Code availability
The statistical coding is available upon request to the corresponding author.
Author contributions
Data collection and data analysis were performed by CØA, HT and ED-L. Manuscript writing was performed by CØA, HT, ED-L and EPJ. Statistical analyses were performed by RR. Revision of the manuscript for important intellectual content was performed by all authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Andersen, C.Ø., Travis, H., Dehlholm-Lambertsen, E. et al. The Cost of Flexible Bronchoscopes: A Systematic Review and Meta-analysis. PharmacoEconomics Open 6, 787–797 (2022). https://doi.org/10.1007/s41669-022-00356-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41669-022-00356-0