Abstract
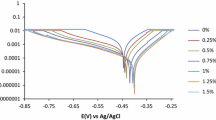
The effect of synergistic corrosion inhibition efficiency of Azadirachta indica gum with halides (KCl, KBr and KI) on mild steel in 1 mol L−1 HCl was studied using conventional weight loss and electrochemical studies. The temperature (303–328 ± 1 K) and time (1, 2, 3, 4, 6 and 24 h) effect on the inhibition of corrosion has also been tested. The inhibition efficiency of gum exudates of Azadirachta indica considerably increased with the presence of halide ions at all temperature ranges studied. Corrosion kinetic parameters and thermodynamic adsorption parameters were calculated and discussed in detail. The mode of adsorption of inhibitor on the mild steel surface was examined by FT-IR spectroscopy.













Similar content being viewed by others
References
Gopi D, Bhauvanerwaran N, Rajewari S (2000) Synergistic effect of thiourea derivatives and non-ionic surfactants on the inhibition of corrosion of carbon steel in acid environments. Corr Meth Mater 47:332–342
Li X, Deng S, Fu H (2009) Inhibition action of tween-80 on the corrosion of cold rolled steel in sulfuric acid. Corros Sci 51:1344–1351
Umoren SA, Solomon MM, Udosoro II, Udoh AP (2010) Synergistic and antagonistic effects between halide ions and carboxymethyl cellulose for the corrosion inhibition of mild steel in sulphuric acid solution. Cellulose 17:635–648
Larabi L, Harek Y, Traisnel M, Mansri A (2004) Synergistic influence of poly(4-vinylpyridine) and potassium iodide on inhibition of corrosion of mild steel in 1 M HCl. J Appl Electrochem 34:833–839
Umoren SA, Ebenso EE (2008) Studies of the anti-corrosive effect of Raphia hookeri exudate gum-halide mixtures for aluminium corrosion in acidic medium. Pigm Resin Technol 37:173–186
Oguzie EE (2006) Gum arabic as a potential corrosion inhibitor for aluminium in alkaline medium and its adsorption characteristics. Mater Chem Phys 99:441–446
Umoren SA, Obot IB, Ebenso EE, Obi-Egbedi N (2008) Synergistic inhibition between naturally occurring exudates gum and halide ions on the corrosion of mild steel in acidic medium. Int J Electrochem Sci 3:1029–1046
Benabdellah M, Aouniti A, Dafali A, Hammouti B, Benkaddaur M, Yahyi A, Ettouhami A (2006) Investigation of the inhibitive effect of triphenyltin 2-thiophene carboxylate on corrosion of steel in 2 M H3PO4 solutions. App Surf Sci 252:8341–8347
Tang L, Mu G, Liu G (2003) The effect of neutral red on the corrosion inhibition of cold rolled steel in 1.0 M hydrochloric acid. Corros Sci 45:2251–2262
Eddy NO, Ebenso EE (2008) Corrosion inhibitive properties and adsorption behavior of ethanol extract of Piper guinensis as a green corrosion inhibitor for mild steel in H2SO4. Afr J Pure Appl Chem 2:46–54
Migahed MA (2005) Effectiveness of some non ionic surfactants as corrosion inhibitors for carbon steel pipelines in oil fields. Mater Chem Phys 93:48–53
Ashassi-Sorkhabi H, Shaabani B, Seifzadeh D (2005) Effect of some pyrimidinic Shciff bases on the corrosion of mild steel in hydrochloric acid solution. Appl Surf Sci 239:154–164
Bockris JOM, Reddy AKN (2000) Modern electrochemistry: 2nd edition, Electrodics in chemistry, engineering, biology and environmental science. Kluwer Academic, New York
Chin RJ, Nobe K (1972) Electrodissolution kinetics of iron in chloride solutions III. Acidic Electrodissolution kinetics of iron in chloride solutions III. J Electrochem Soc 119:1457–1461
Okafor PC, Zheng Y (2009) Gum arabic as a potential corrosion inhibitor for aluminium in alkaline medium and its adsorption characteristics. Corros Sci 51:850–859
Aramaki K, Hackerman N (1969) Inhibition mechanism of medium-sized polymethyleneimine. J Electrochem Soc 116:568–574
Li X, Deng S, Fu H, Li T (2009) Adsorption and inhibition effect of acknowledgment 6-benzylaminopurine on cold rolled steel in 1.0 M HCl. Electrochem Acta 54:4089–4098
Ferreira ES, Giancomelli C, Giacomelli FC, Spinelli A (2004) Spinelli evaluation of the inhibitor effect of L-ascorbic acid on the corrosion of mild steel. Mater Chem Phys 83:129–134
EIS Equivalent Circuits (2010) Electrochemical impedance spectroscopy in PEM fuel cells. Springer, London
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest on publishing the article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Brindha, T., Parimalagandhi, K. & Mallika, J. Thermodynamic and Electrochemical Analysis of Synergistic Corrosion Inhibition Performance of Natural Polysaccharides with Metal Halides on Mild Steel in Hydrochloric Acid Solution. J Bio Tribo Corros 6, 3 (2020). https://doi.org/10.1007/s40735-019-0296-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40735-019-0296-7