Abstract
Objective
To assess overall and by neighborhood risk environments whether multilevel resilience resources were associated with HIV virologic suppression among African American/Black adults in the Southeastern United States.
Setting and Methods
This clinical cohort sub-study included 436 African American/Black participants enrolled in two parent HIV clinical cohorts. Resilience was assessed using the Multilevel Resilience Resource Measure (MRM) for African American/Black adults living with HIV, where endorsement of a MRM statement indicated agreement that a resilience resource helped a participant continue HIV care despite challenges or was present in a participant’s neighborhood. Modified Poisson regression models estimated adjusted prevalence ratios (aPRs) for virologic suppression as a function of categorical MRM scores, controlling for demographic, clinical, and behavioral characteristics at or prior to sub-study enrollment. We assessed for effect measure modification (EMM) by neighborhood risk environments.
Results
Compared to participants with lesser endorsement of multilevel resilience resources, aPRs for virologic suppression among those with greater or moderate endorsement were 1.03 (95% confidence interval: 0.96–1.11) and 1.03 (0.96–1.11), respectively. Regarding multilevel resilience resource endorsement, there was no strong evidence for EMM by levels of neighborhood risk environments.
Conclusions
Modest positive associations between higher multilevel resilience resource endorsement and virologic suppression were at times most compatible with the data. However, null findings were also compatible. There was no strong evidence for EMM concerning multilevel resilience resource endorsement, which could have been due to random error. Prospective studies assessing EMM by levels of the neighborhood risk environment with larger sample sizes are needed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
African American/Black people are the racial and ethnic group most adversely impacted by HIV in the USA. Representing only 13% of the US population, African American/Black people ≥ 13 years of age comprise nearly half of all persons with diagnosed HIV [1, 2] and experience a disproportionate burden of HIV virologic failure, morbidity, and mortality [3,4,5].
Systemic racism contributes to racial disparities in HIV outcomes [6,7,8,9]. For example, disadvantage in predominantly Black neighborhoods is largely due to systemic racism embedded in historical and current housing policies (e.g., residential segregation) [10,11,12] and is a robust correlate of HIV [13, 14]. African American/Black individuals disproportionately reside in high-risk neighborhood environments characterized by intentional disinvestment, socioeconomic deprivation, and/or social and physical disorder [2, 13,14,15]. Yet not all African American/Black people who reside in high-risk neighborhoods experience adverse HIV-related behaviors or outcomes [16, 17]. This suggests that resilience resources (i.e., factors both internal and external to the individual that promote positive behaviors and outcomes despite adversity) [18,19,20] may help reduce racial disparities.
Resilience resources may lessen racial/ethnic HIV health disparities by serving as a mediator on pathways that promote positive health behaviors (e.g., care engagement and antiretroviral therapy (ART) adherence) [21,22,23] and by buffering the effects of adversities on physiological functioning [24, 25]. Yet prior work has overwhelmingly focused on individual-level resilience in relation to HIV outcomes [26] even though resources at the non-individual-level can facilitate resilience among African American/Black adults in the USA [27,28,29]. Thus, there remains a critical evidence gap surrounding the role interpersonal, organizational, and neighborhood resilience resources play in improving HIV-related behaviors and outcomes [26]. Furthermore, most of the available tools for examining HIV-related resilience were psychometrically tested among individuals less likely to be impacted by the epidemic (e.g., White adults who are not living with HIV) [30,31,32,33], thereby limiting the extent to which resilience resources can be understood to affect HIV outcomes among disproportionately impacted populations.
Using a multilevel resilience measure validated specifically for African American/Black people living with HIV (AA/B PLWH) [34], this sub-study assessed overall and levels of the neighborhood risk environment whether endorsement of multilevel resilience resources was associated longitudinally with virologic suppression among AA/B PLWH in the Southeastern US. We hypothesized that greater endorsement of resilience resources would be positively associated with virologic suppression, especially among African American/Black adults living in high-risk neighborhoods.
Methods
Study Population
African American/Black patients who met the following criteria and agreed to participate in the sub-study were included in the study population: ≥ 18 years of age; enrolled in the University of North Carolina (UNC) Center for AIDS Research HIV Clinical Cohort [35] or the University of Alabama Birmingham (UAB) 1917 Clinic Cohort [36, 37] for at least one year; provided accompanying patient-reported outcome data at least once while enrolled in the parent UNC and UAB cohort studies; and had the ability to speak, read, and understand English sufficiently to complete consent procedures and study instruments. Enrollment occurred from September 1, 2019 to August 31, 2020 at UNC and September 1, 2019 to October 31, 2020 at UAB. At enrollment and during follow-up through either August 31, 2021 or a maximum of 18 months post-enrollment, self-reported data on resilience resources and other data (e.g., medical record data and self-reported housing status) were ascertained.
To maximize sample size, the study population described above was expanded to include UNC and UAB clinical cohort participants in a prior sub-study [34]. Enrollment in the prior sub-study was from November 1, 2018 to November 12, 2019 at UNC and November 1, 2018, to October 15, 2019 at UAB. Eligibility criteria for the prior sub-study are detailed elsewhere [34]. Self-reported data on resilience resources and other data were ascertained for these prior sub-study participants at enrollment and during follow-up through a maximum of 18 months post-enrollment.
All human subjects activities for the current and prior sub-study were approved by Institutional Review Boards at UAB, UNC, and Brown University. Participants provided written informed consent to participate in each sub-study.
Measures
Outcome
The outcome was time-updated virologic suppression at enrollment or during follow-up. Consistent with prior work [38,39,40], virologic suppression was considered as a binary measure of achieving an HIV-1 RNA level below 50 copies/mL (cp/mL) at a given HIV-1 RNA assessment. HIV-1 RNA levels below a given assay’s lower detection limit were set equal to half the assay’s lower limit. Levels above an assay’s upper detection limit were set equal to 1 cp/mL above the assay’s upper limit.
Primary and Secondary Exposure
The primary exposure was a self-reported, time-fixed measure of endorsement of multilevel resilience resources at enrollment based on responses to the 38-item Multilevel Resilience Resource Measure-Long Form (MRM-LF) [34, 41]. The MRM-LF was developed and psychometrically tested among African American/Black adults living with HIV in the Southeastern US. The MRM-LF items demonstrated content validity (as assessed by experts) and performed well in cognitive testing. Additionally, the MRM-LF demonstrated strong internal consistency reliability (coefficient alpha = 0.91) and convergent validity with established measures [34]. The MRM-LF asks respondents how much they agree that specific resilience resources at the individual-, interpersonal-, or organizational-level helped them to continue with their HIV care despite life challenges. Example resilience resources include “I have someone who helps me keep a positive attitude about living with HIV” and “The healthcare staff call me with reminders for appointments or medications.” The MRM-LF also asks respondents how much they agree a given statement describes the neighborhood where they currently live. An example statement is “My neighborhood has religious services.” Item response options range from 1 (strongly disagree) to 5 (strongly agree). To generate continuous, tertile (i.e., greater, moderate, and lesser), and binary (i.e., greater and lesser based on median split) summary measures of endorsement of multilevel resilience resources, valid responses were necessary on at least 12/16 individual-level items, 7/9 interpersonal-level items, 5/6 organizational-level items, and all neighborhood-level items of the MRM-LF [34]. The continuous multilevel summary measure was created by averaging the non-missing MRM-LF items, with summary scores ranging from 1 to 5 and higher scores reflecting greater endorsement. Tertile and binary multilevel measures were created from the continuous measure. The secondary exposure was self-reported, time-fixed tertile and binary measures of level-specific resilience resource endorsement (i.e., individual-, interpersonal-, organizational-, and neighborhood-level). Level-specific measures were created by averaging the non-missing items for each resilience level.
Covariates
Time-fixed non-resilience covariate information included age (continuous), gender (male/female), sexual orientation (heterosexual/gay, lesbian, or bisexual), housing status (stably housed/unstably housed/homeless/other), health insurance (uninsured/insured (i.e., private insurance, Medicaid, Medicare, or other public insurance)), at-risk alcohol use (yes/no), drug use (yes/no), depression (yes/no), panic syndrome (yes/no), other mental illness(es) (yes/no), AIDS-defining illnesses (yes/no), years since ART initiation (continuous), CD4 cell count (cells/μL) (continuous), virologic suppression (yes/no), and three indices of neighborhood risk environments (tertiles: high/moderate/low). Covariate information was ascertained at or prior to enrollment based on data availability during relevant time windows. Variables were selected to describe the analytic sample, minimize potential sources of confounding or selection bias, or explore effect measure modification (EMM). Potential sources of confounding or selection bias were discerned from a causal directed acyclic graph [42, 43] (not shown) that was constructed based on the literature, where amount of resilience resources was the exposure [21, 26, 44,45,46,47,48,49,50,51,52,53].
Gender and sexual orientation were captured via self-report on the Lesbian, Gay, Bisexual, and Transgender (LGBT) Identity Measure [54] or from medical records. Current housing status was self-reported [55], and health insurance was obtained from medical records.
Following recommendations [56, 57] and prior work [58], self-reported and medical record data were combined for select covariates. Specifically, at-risk alcohol use was defined as having an alcohol abuse or dependence diagnosis in the medical record [59] or via self-reported alcohol use on the Alcohol Use Disorders Identification Test-Concise (AUDIT-C score ≥ 4 for men and ≥ 3 for women) [60] in the 12 months before enrollment [61, 62]. Drug use was defined as having a drug-related diagnosis in the medical record or via self-reported use of crack/cocaine, amphetamines, or opioids not prescribed by a physician on the Alcohol, Smoking and Substance Involvement Screening Test [63] in the 3 months before enrollment. Marijuana use was excluded following prior work [57, 58]. Medical records and self-reported depressive symptoms on the Patient Health Questionnaire (PHQ-9) [64] were used to create a binary indicator of moderate to severe depression in the 2 weeks before enrollment [65]. Medical records and self-reported anxiety on the Patient Health Questionnaire (PHQ-5) [66] were used to create a binary indicator of the presence/absence of panic syndrome in the 4 weeks before enrollment. Mental health diagnoses other than depression and panic syndrome (e.g., schizophrenia and bipolar disorder) in the 4 weeks before enrollment were captured via medical records [67].
AIDS-defining illnesses at or two years before enrollment were ascertained from medical records. Participants without an alcohol abuse/dependence, drug use, mental health diagnosis, or an AIDS-defining illness in their medical record were considered to not have the diagnosis. Years since ART initiation at enrollment, last CD4 cell count (cells/μL) in the two years before enrollment, and virologic suppression for the last HIV-1 RNA measurement taken in the two years before enrollment were obtained from medical records. When a level-specific resilience resource measure was the exposure, the other level-specific resilience resource measures were considered as covariates.
Three census tract–level indices of the neighborhood risk environment at enrollment included a Neighborhood Disadvantage Index and Esri-derived Assault Rate and Murder Rate indices that were developed based on an established geocoding and data linkage protocol [68, 69]. The Neighborhood Disadvantage Index included three indicators of education, unemployment, and income obtained from 2019 American Community Survey 5-year estimates. The Index was normed to the national level such that a Z-score = 1 indicated one standard deviation greater disadvantage than the national level. Assault and Murder Rate Indices were categorized by tertile where neighborhoods with an index > 100 had an increased assault or murder risk compared to the national risk level.
Statistical Analyses
We excluded enrolled participants with missing values for required items on the MRM-LF at enrollment, without information for at least one of the non-resilience covariates at or before enrollment, or without HIV-1 RNA data at or within the 5 months following enrollment. We constructed two time intervals during 10 months of follow-up per participant, with time interval 1 occurring 0–5 months and time interval 2 occurring > 5–10 months post-enrollment. If a participant had more than one HIV-1 RNA measurement available within a time interval, we used the last measurement. Death during follow-up was considered to be a censored event rather than defining outcomes after death as being undefined [70, 71]. Specifically, if a participant had an available HIV-1 RNA measurement and later died during time interval 1, they were censored at time interval 2 (n = 0). Participants were also censored at time interval 2 due to death (n = 1) (e.g., died during time interval 2 prior to HIV-1 RNA measurement) or another reason that precluded an HIV-1 RNA measurement being taken during the second time interval (n = 153). Data on other reasons precluding a HIV-1 RNA measurement were unavailable but may have reflected missed or canceled clinic appointments.
Chi-squared and Mann–Whitney tests were used to compare characteristics of included and a subset of excluded participants, as well as to compare characteristics of included participants across clinic cohorts. In primary analysis for the overall relationship, we fit time-updated binary virologic suppression as a function of the multilevel resilience resource summary score (tertiles or binary), time intervals, and/or non-resilience covariates. We used modified Poisson regression models fit with generalized estimating equations (GEE) to estimate prevalence ratios (PRs) for virologic suppression, accounting for outcomes clustered within neighborhoods defined by census tract at enrollment [72]. We assumed that repeated outcomes for an individual were nested within the same census tract specified at enrollment, whereby clustering within neighborhoods also accounted for clustering within individuals [73]. The unadjusted model included the time-fixed multilevel resilience resource summary measure and time interval variable. The adjusted model additionally included all covariates to control for potential confounding and selection bias. During additional primary analyses of the overall relationship, we included product terms between multilevel resilience resources and time interval in all models to assess whether PRs differed by time interval. To assess for EMM by neighborhood risk environment during primary analyses, we added product terms between categorical (i.e., binary or tertiles) multilevel resilience and the relevant neighborhood risk index to adjusted models that excluded multilevel resilience-time interval product terms. We tested for EMM by one neighborhood risk index at a time. P values for product terms were obtained from global chi-squared tests.
Our secondary analyses used modified Poisson regression models to estimate the PR for virologic suppression using a tertile or binary measure of one level–specific resilience resource summary measure at a time (i.e., individual-, interpersonal-, organizational-, and neighborhood-level). The same set of covariates in the primary analysis was included in all adjusted models, but no exposure-time interval product terms were included. The approach to handling clustering in the primary analysis was also used during secondary analyses. We also assessed for EMM by neighborhood risk environment during secondary analyses.
We used restricted quadratic splines for continuous independent variables at 4 unequal intervals (i.e., 5th, 35th, 65th, and 95th percentiles) [74], indicators for categorical variables, and an independent working correlation structure in our outcome models. During tertiary analyses, we repeated all primary and secondary analyses but specified an exchangeable correlation. We also repeated primary and secondary analyses using an independent working correlation structure but clustered within individuals and not within census tract at enrollment.
Our interpretation of study findings was based on compatibility with the data using the point estimates, confidence intervals (CIs), and P values [75,76,77,78]. Specifically, we did not assess for evidence of an association based solely on whether the 95% CIs did not include the null or P values were < 0.05. Analyses were performed using SAS 9.4 (SAS Institute, Inc., Cary, NC).
Results
Figure 1 presents the exclusion criteria used to obtain our final analytic sample of 436 participants. Table 1 shows the characteristics of those who were included and excluded from the final analytic sample. Compared to excluded participants, included participants had greater endorsement of multilevel and interpersonal-level resilience resources and the same degree of endorsement of individual-, organizational-, and neighborhood-level resilience resources. Included participants were younger and on ART for a longer time period and had lower CD4 cell counts. The majority of included participants were men and heterosexual and were virally suppressed before enrollment. At time intervals 1 and 2, 89.9% (392/436) and 85.1% (240/282) achieved virologic suppression, respectively (not shown). Online Resource 1 shows the characteristics of included participants stratified by clinical cohort.
Exclusion criteria used to identify 436 participants included in the final analysis sample. *Resilience data were complete if at least 12 of the 16 individual-level items, at least 7 of the 9 interpersonal-level items, at least 5 of the 6 organizational-level items, and all neighborhood-level items were not missing
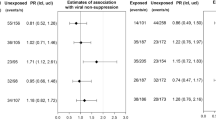
Focusing on adjusted findings henceforth, Table 2 displays the adjusted PRs (aPRs) and corresponding 95% CIs for virologic suppression, comparing greater versus lesser endorsement of multilevel resilience resources based on a binary measure, overall, and levels of neighborhood risk environments. Concerning the overall findings across time intervals that compared greater to lesser resource endorsement, a null finding was most compatible with the data (aPR = 0.99, 95% CI = 0.94–1.05). The corresponding aPRs in Table 2 were not meaningfully different by time interval.
Comparing virologic suppression by tertiles of multilevel resilience resource endorsement (greater or moderate versus lesser) across time intervals (Table 3) indicated that positive associations were most compatible with the data (aPR = 1.03, 95% CI = 0.96–1.11 and aPR = 1.03, 95% CI = 0.96–1.11, respectively). By time interval, aPRs were higher in time interval 2 than in time interval 1.
There was no strong evidence for EMM by neighborhood risk environment for either the binary measure or tertiles of multilevel resilience resource endorsement (Tables 2 and 3).
Online Resources 2–5 show the aPRs overall for binary measures of level-specific resilience resources. At times, modest non-null relationships between endorsement of level-specific resilience resources and virologic suppression were most compatible with the data (e.g., interpersonal-level (aPR = 1.03, 95% CI = 0.97–1.11) and neighborhood level (aPR = 1.04, 95% CI = 0.98–1.10)). There was some evidence of EMM by the neighborhood risk environment for greater versus lesser organizational-level resilience resources. For example, a negative association between greater (versus lesser) endorsement of organizational-level resilience resources and virologic suppression was observed among participants residing in neighborhoods characterized by high (aPR = 0.88, 95% CI = 0.78–1.00) or moderate (aPR = 0.96, 95% CI = 0.87–1.05) disadvantage, while a modest positive association was observed among participants residing in neighborhoods with low disadvantage (aPR = 1.05, 95% CI = 0.96–1.15). There was no strong evidence for EMM for other level-specific resilience resources. The corresponding tertile analyses (Online Resources 6–9) provided evidence that a modest positive overall relationship between greater versus lesser resilience resources and virologic suppression was most compatible with the data at the individual and neighborhood level. EMM findings from the tertiles analyses were similar to those from the binary measures of level-specific resilience resource endorsement. Findings from tertiary analyses did not meaningfully change our inferences (results not shown).
Discussion
This study, which used longitudinal data from African American/Black participants enrolled in two HIV clinical cohorts in the Southeastern US, found modest positive associations between greater endorsement of multilevel resilience resources and virologic suppression. Specifically, the overall adjusted findings based on tertiles indicated that African American/Black adults with greater and moderate levels of endorsement of multilevel resilience resources experienced slightly more virologic suppression compared to participants with lesser resource endorsement. Concerning level-specific resilience resources, greater endorsement of individual- and neighborhood-level resources was modestly positively associated with virologic suppression. Though there was no strong evidence for EMM by the neighborhood risk environment concerning endorsement of multilevel resilience resources, there was at times evidence of EMM for certain level-specific resilience resources. However, given our study’s small sample size, our EMM findings may be in part due to random error. If we assume that African American/Black individuals with greater endorsement have greater multilevel resilience resources and in turn are more resilient and more engaged in behaviors (e.g., sufficient ART adherence) [53, 79] that facilitate virologic suppression, then these findings are consistent with our prior hypotheses and suggest that greater resilience may be modestly positively associated with virologic suppression.
This study’s findings are similar to recent resilience literature in the USA. Cross-sectional data from African American participants in the Women’s Interagency HIV Study showed that individual-level resiliency skills measured via the Brief Resilience Scale [32] were positively associated with a higher likelihood of virologic suppression [80] among women reporting low HIV-related stigma (adjusted odds ratio (aOR) = 1.51, 95% CI = 1.00–2.27) and depressive symptoms (aOR = 1.68, 95% CI = 1.04–2.70); for women reporting high stigma or depression, individual resiliency did not predict virologic improvements. Among patients in the HIV Research Network [81], analyses of individual resilience found that every five-point increase in Patient Activation Measure score correlated with an 8% increase in the odds of virologic suppression (aOR = 1.08, 95% CI = 1.00–1.17), with this relationship mediated by greater ART adherence [53]. Exploratory [79, 82] studies among African Americans at higher risk of adverse HIV outcomes (e.g., men who have sex with men) have also shown positive associations between individual- and community-level resilience resources and virologic suppression. However, these prior studies were cross-sectional, had small sample sizes, and focused largely on individual and interpersonal resilience resources. We were unable to find any literature that assessed the resilience–virologic suppression relationship by neighborhood risk environments, which is a key contribution of the current study.
Regarding results that were most compatible with the data in our study, differences in measure instructions may have contributed to the positive resilience–virologic suppression relationship that was observed for greater versus lesser neighborhood-level resources but not at the interpersonal- or organizational-level overall. By asking how well statements concerning resilience resources described a respondent’s neighborhood (rather than how well resilience resources helped to handle life challenges in order to stick with HIV care), the neighborhood-level instructions may have more directly captured the amount of resilience resources a participant possessed.
Other limitations may have influenced our findings. First, there was minimal variability in resilience resource endorsement and virologic suppression among study participants, which potentially limited our ability to fully characterize the resilience–virologic suppression relationship. Second, our study may have been subject to unmeasured sources of confounding or selection bias due to censoring or exclusions. For example, we could not adjust for stress, discrimination, or trauma due to lack of access to these data or considerable missing data [44,45,46,47, 83]. Third, our study’s overall findings likely also suffer from random error because of the small sample size. Fourth, our analytic sample combined resilience data ascertained both before and after the start of the COVID-19 pandemic, which may have impacted findings. However, we did not stratify analyses by the pandemic start date to avoid further reducing our sample size. Fifth, for outcome clustering within neighborhoods, we did not account for participants moving to different census tracts between outcome assessment in time interval 1 (which included enrollment) and in time interval 2. However, 95.4% (62/65) of participants with census tract information during time interval 2 resided in the same census tract as those during time interval 1. Last, our findings may not be generalizable to other populations with a different distribution of effect measure modifiers. These effect measure modifiers may include gender (e.g., transgender versus cisgender) and sexual orientation (e.g., heterosexual versus gay, lesbian, or bisexual). Given that the majority of the analytic sample in this study was male and heterosexual and was virally suppressed prior to enrollment, additional research using the MRM is needed to examine the resilience–virologic suppression relationship by level of the neighborhood risk environment among populations who historically have been more disproportionately affected by the HIV epidemic, such as African American/Black women (transgender and cisgender) living with HIV [2, 84], African American/Black men who have sex with men [2], and African American/Black adults with lower ART adherence [85].
In summary, to the best of our knowledge, this study is the first to rigorously examine the longitudinal relationship between multilevel resilience resources and HIV virologic suppression among African American/Black adults and to explore this relationship by levels of the neighborhood risk environment. Our findings provide some evidence of a modest positive relationship between endorsement of multilevel resilience resources and virologic suppression and perhaps multilevel resilience and virologic suppression. Therefore, African American/Black adults living with HIV may benefit from resilience-building interventions that support virologic suppression (e.g., flexible healthcare services such as mobile clinics and establishments such as safe injection sites that help reduce public drug use [86]). Given the high levels of endorsement of multilevel resilience resources and virologic suppression, future research should explore the resilience–virologic suppression relationship for a longer time period (> 10 months) among a community sample who may not be actively engaged in HIV care. In addition, future studies with a larger sample size are needed to better assess EMM by levels of the neighborhood risk environment.
Data Availability
The datasets generated and/or analyzed during the current study are not publicly available, but may be available from the corresponding author if relevant data have not been discarded and with approval from relevant IRBs and the University of North Carolina Center for AIDS Research HIV Clinical Cohort and the University of Alabama Birmingham 1917 Clinic Cohort.
References
Centers for Disease Control and Prevention. Diagnoses of HIV infection in the United States and dependent areas 2019: national profile. 2021. https://www.cdc.gov/hiv/library/reports/hiv-surveillance/vol-32/content/national-profile.html#Diagnoses. Accessed 26 Apr 2022.
Dailey AF, Gant Z, Hu X, Johnson Lyons S, Okello A, Satcher Johnson A. Association between social vulnerability and rates of HIV diagnoses among black adults, by selected characteristics and region of residence — United States, 2018. MMWR Morb Mortal Wkly Rep. 2022;71:167–70. https://doi.org/10.15585/mmwr.mm7105a2
The Henry J. Kasier Family Foundation. Black Americans and HIV/AIDS: The Basics. 2020. https://www.kff.org/hivaids/fact-sheet/black-americans-and-hivaids-the-basics/. Accessed 26 Apr 2022.
Centers for Disease Control and Prevention. HIV Among African American People. 2022. https://www.cdc.gov/hiv/pdf/group/racialethnic/africanamericans/cdc-hiv-africanamericans.pdf. Accessed 26 Apr 2022.
Centers for Disease Control and Prevention. HIV Surveillance Report, 2019; vol. 32. 2021. https://www.cdc.gov/hiv/pdf/library/reports/surveillance/cdc-hiv-surveillance-report-2018-updated-vol-32.pdf. Accessed 26 Apr 2022.
Ahonkhai AA, Rebeiro PF, Jenkins CA, Rickles M, Cook M, Conserve DF, et al. Individual, community, and structural factors associated with linkage to HIV care among people diagnosed with HIV in Tennessee. Shook NJ editor. PLoS One. 2022;17:e0264508. https://doi.org/10.1371/journal.pone.0264508.
Bonacci RA, Smith DK, Ojikutu BO. Toward greater pre-exposure prophylaxis equity: increasing provision and uptake for Black and Hispanic/Latino individuals in the U.S. Am J Prev Med. 2021;61(5 Suppl 1):S60-S72. https://doi.org/10.1016/j.amepre.2021.05.027
English D, Carter JA, Boone CA, Forbes N, Bowleg L, Malebranche DJ, et al. Intersecting structural oppression and black sexual minority men’s health. Am J Prev Med. 2021;60:781–91. https://doi.org/10.1016/j.amepre.2020.12.022.
Bowleg L, Malekzadeh AN, Mbaba M, Boone CA. Ending the HIV epidemic for all, not just some: structural racism as a fundamental but overlooked social-structural determinant of the US HIV epidemic. Curr Opin HIV AIDS. 2022;17:40–5. https://doi.org/10.1097/COH.0000000000000724.
Riley AR. Neighborhood disadvantage, residential segregation, and beyond—lessons for studying structural racism and health. J Racial Ethn Heal Disparities. 2018;5:357–65. https://doi.org/10.1007/s40615-017-0378-5.
Mujahid MS, Gao X, Tabb LP, Morris C, Lewis TT. Historical redlining and cardiovascular health: the multi-ethnic study of atherosclerosis. Proc Natl Acad Sci U S A. 2021;118(51):e2110986118. https://doi.org/10.1073/pnas.2110986118.
Diaz A, O’Reggio R, Norman M, Thumma JR, Dimick JB, Ibrahim AM. Association of historic housing policy, modern day neighborhood deprivation and outcomes after inpatient hospitalization. Ann Surg. 2021;274:985–91. https://doi.org/10.1097/SLA.0000000000005195.
Ibragimov U, Beane S, Adimora AA, Friedman SR, Williams L, Tempalski B, et al. Relationship of racial residential segregation to newly diagnosed cases of HIV among Black heterosexuals in US metropolitan Areas, 2008–2015. J Urban Heal. 2019;96:856–67. https://doi.org/10.1007/s11524-018-0303-1.
Dawit R, Trepka MJ, Duncan DT, et al. Neighborhood factors associated with racial/ethnic disparities in achieving sustained HIV viral suppression among Miami-Dade County Ryan White Program Clients. AIDS Patient Care STDS. 2021;35:401–10. https://doi.org/10.1089/apc.2021.0067.
Fennie KP, Lutfi K, Maddox LM, Lieb S, Trepka MJ. Influence of residential segregation on survival after AIDS diagnosis among non-Hispanic blacks. Ann Epidemiol. 2015;25:113-119.e1. https://doi.org/10.1016/j.annepidem.2014.11.023.
Rebeiro PF, Howe CJ, Rogers WB, Bebawy SS, Turner M, Kheshti A, et al. The relationship between adverse neighborhood socioeconomic context and HIV continuum of care outcomes in a diverse HIV clinic cohort in the Southern United States. AIDS Care. 2018;30:1426–34. https://doi.org/10.1080/09540121.2018.1465526.
Bauermeister JA, Eaton L, Andrzejewski J, Loveluck J, VanHemert W, Pingel ES. Where you live matters: structural correlates of HIV risk behavior among young men who have sex with men in Metro Detroit. AIDS Behav. 2015;19(12):2358–69. https://doi.org/10.1007/s10461-015-1180-1.
Aburn G, Gott M, Hoare K. What is resilience? An integrative review of the empirical literature. J Adv Nurs. 2016;72(5):980–1000. https://doi.org/10.1111/jan.12888.
Wilson DK, Bamishigbin ON, Guardino C, Schetter CD. Resilience resources in low-income Black, Latino, and White fathers. Soc Sci Med. 2021;282:114139. https://doi.org/10.1016/j.socscimed.2021.114139.
Schetter CD, Dolbier C. Resilience in the context of chronic stress and health in adults. Soc personal psychol compass. 2011;5(9):634–52. https://doi.org/10.1111/j.1751-9004.2011.00379.x.
Dale S, Cohen M, Weber K, Cruise R, Kelso G, Brody L. Abuse and resilience in relation to haart medication adherence and HIV viral load among women with HIV in the United States. AIDS Patient Care STDS. 2014;28(3):136–43. https://doi.org/10.1089/apc.2013.0329.
Hudelson C, Cluver L. Factors associated with adherence to antiretroviral therapy among adolescents living with HIV/AIDS in low- and middle-income countries: a systematic review. AIDS Care - psychol socio-medical Asp AIDS/HIV. 2015;27(7):805–16. https://doi.org/10.1080/09540121.2015.1011073.
Palmer RC, Ismond D, Rodriquez EJ, Kaufman JS. Social determinants of health: future directions for health disparities research. Am J Public Health. 2019;5(9):634–52. https://doi.org/10.1111/j.1751-9004.2011.00379.x.
Spies G, Seedat S. Depression and resilience in women with HIV and early life stress: does trauma play a mediating role? A cross-sectional study BMJ Open. 2014;4:e004200. https://doi.org/10.1136/bmjopen-2013-004200.
Sauceda JA, Wiebe JS, Simoni JM. Childhood sexual abuse and depression in Latino men who have sex with men: does resilience protect against nonadherence to antiretroviral therapy? J Health Psychol. 2016;21(6):1096–106. https://doi.org/10.1177/1359105314546341.
Dulin AJ, Dale SK, Earnshaw VA, Fava JL, Mugavero MJ, Napravnik S, et al. Resilience and HIV: a review of the definition and study of resilience. AIDS Care - Psychol Socio-Medical Asp AIDS/HIV. 2018;30(sup5):S6–17. https://doi.org/10.1080/09540121.2018.1515470.
Lipira L, Williams EC, Nevin PE, Kemp CG, Cohn SE, Turan JM, et al. Religiosity, social support, and ethnic identity: exploring “resilience resources” for African-American women experiencing HIV-related stigma. J Acquir Immune Defic Syndr. 2019;81(2):175–83. https://doi.org/10.1097/QAI.0000000000002006.
Chen Y-T, Duncan DT, Issema R, Goedel WC, Callander D, Bernard-Herman B, et al. Social-environmental resilience, PrEP uptake, and viral suppression among young Black men who have sex with men and young Black transgender women: the neighborhoods and networks (N2) study in Chicago. J Urban Heal. 2020;97:728–38. https://doi.org/10.1007/s11524-020-00425-x.
Qiao S, Ingram L, Deal ML, Li X, Weissman SB. Resilience resources among African American women living with HIV in Southern United States. AIDS. 2019;33:S35-44. https://doi.org/10.1097/QAD.0000000000002179.
Windle G, Bennett KM, Noyes J. A methodological review of resilience measurement scales. Health Qual Life Outcomes. 2011;9:8. https://doi.org/10.1186/1477-7525-9-8.
Connor KM, Davidson JRT. Development of a new resilience scale: the Connor-Davidson Resilience scale (CD-RISC). Depress Anxiety. 2003;18(2):76–82. https://doi.org/10.1002/da.10113.
Smith BW, Dalen J, Wiggins K, Tooley E, Christopher P, Bernard J. The brief resilience scale: assessing the ability to bounce back. Int J Behav Med. 2008;15(3):194–200. https://doi.org/10.1080/10705500802222972.
Friborg O, Hjemdal O, Rosenvinge JH, Martinussen M. A new rating scale for adult resilience: what are the central protective resources behind healthy adjustment? Int J Methods Psychiatr Res. 2003;12:65–76. https://doi.org/10.1002/mpr.143.
Dulin AJ, Fava JL, Earnshaw VA, Dale SK, Carey MP, Wilson-Barthes M, et al. Development of long and short forms of the multilevel resilience resource measure for African American/Black adults living with HIV. AIDS Behav. 2022;26:2469–84. Available from: https://link.springer.com/10.1007/s10461-022-03579-8
Howe CJ, Cole SR, Napravnik S, Eron JJ. Enrollment, retention, and visit attendance in the University of North Carolina Center for AIDS Research HIV Clinical Cohort, 2001–2007. AIDS Res Hum Retroviruses. 2010;26:875–81. https://doi.org/10.1089/aid.2009.0282.
Mugavero MJ, Lin H-Y, Allison JJ, Willig JH, Chang P-W, Marler M, et al. Failure to establish HIV care: characterizing the “No Show” phenomenon. Clin Infect Dis. 2007;45:127–30. https://doi.org/10.1086/518587.
Kitahata MM, Rodriguez B, Haubrich R, Boswell S, Mathews WC, Lederman MM, et al. Cohort profile: the centers for AIDS Research Network of Integrated Clinical Systems. Int J Epidemiol. 2008;37:948–55. https://doi.org/10.1093/ije/dym231.
Mugavero MJ, Amico KR, Westfall AO, Crane HM, Zinski A, Willig JH, et al. Early retention in HIV care and viral load suppression. JAIDS J Acquir Immune Defic Syndr. 2012;59:86–93. https://doi.org/10.1097/QAI.0b013e318236f7d2.
Mugavero MJ, Lin H-Y, Allison JJ, Giordano TP, Willig JH, Raper JL, et al. Racial disparities in HIV virologic failure: do missed visits matter? JAIDS J Acquir Immune Defic Syndr. 2009;50:100–8. https://doi.org/10.1097/QAI.0b013e31818d5c37.
Edwards JK, Cole SR, Hall HI, Mathews WC, Moore RD, Mugavero MJ, et al. Virologic suppression and CD4+ cell count recovery after initiation of raltegravir or efavirenz-containing HIV treatment regimens. AIDS. 2018;32:261–6. https://doi.org/10.1097/QAD.0000000000001668.
Dulin AJ, Earnshaw VA, Dale SK, Carey MP, Fava JL, Wilson-Barthes M, et al. A concept mapping study to understand multilevel resilience resources among African American/Black adults living with HIV in the Southern United States. AIDS Behav. 2020;25(3):773–86. https://doi.org/10.1007/s10461-020-03042-6.
Greenland S, Pearl J, Robins JM. Causal diagrams for epidemiologic research. Epidemiology. 1999;10:37–48.
Hernán MA, Robbins JM. Causal Inference: What If. Boca Raton: Chapman & Hall/CRC; 2020. https://cdn1.sph.harvard.edu/wp-content/uploads/sites/1268/2021/03/ciwhatif_hernanrobins_30mar21.pdf
Kirby JB, Kaneda T. Neighborhood socioeconomic disadvantage and access to health care. J Health Soc Behav. 2005;46:15–31 (http://journals.sagepub.com/doi/10.1177/002214650504600103).
Karriker-Jaffe KJ, Zemore SE, Mulia N, J, et al. Neighborhood disadvantage and adult alcohol outcomes: differential risk by race and gender. J Stud Alcohol Drugs. 2012;73(6):865–73.
Wu L-TT, Kouzis AC, Schlenger WE. Substance use, dependence, and service utilization among the US uninsured nonelderly population. Am J Public Health. 2003;93(12):2079–85. https://doi.org/10.2105/ajph.93.12.2079.
Bernhardt C, King C. Neighborhood disadvantage and prescription drug misuse in low-income urban mothers. Drug alcohol depend. 2022;231:109245. https://doi.org/10.1016/j.drugalcdep.2021.109245.
Ironson G, Stuetzle R, Fletcher MA. An increase in religiousness/spirituality occurs after HIV diagnosis and predicts slower disease progression over 4 years in people with HIV. J Gen Intern Med. 2006;21(S5):S62–8. https://doi.org/10.1111/j.1525-1497.2006.00648.x.
Turan B, Fazeli PL, Raper JL, et al. Social support and moment-to-moment changes in treatment self-efficacy in men living with HIV: psychosocial moderators and clinical outcomes. Heal Psychol. 2016;35(10):1126–1134. https://doi.org/10.1037/hea0000356
Weaver KE, Llabre MM, Durán RE, et al. A stress and coping model of medication adherence and viral load in HIV-positive men and women on Highly Active Antiretroviral Therapy (HAART). Heal Psychol. 2005;24(4):385–92. https://doi.org/10.1037/0278-6133.24.4.385.
Friedman MR, Coulter RWS, Silvestre AJ, et al. Someone to count on: social support as an effect modifier of viral load suppression in a prospective cohort study. AIDS Care. 2017;29(4):469–80. https://doi.org/10.1080/09540121.2016.1211614.
Ironson G, Balbin E, Stuetzle R, et al. Dispositional optimism and the mechanisms by which it predicts slower disease progression in HIV: proactive behavior, avoidant coping, and depression. Int J Behav Med. 2005;12(2):86–97. https://doi.org/10.1207/s15327558ijbm1202_6.
Marshall R, Beach MC, Saha S, et al. Patient activation and improved outcomes in HIV-infected patients. J Gen Intern Med. 2013;28(5):668–74. https://doi.org/10.1007/s11606-012-2307-y.
The GenIUSS Group. Best practices for asking questions to identify transgender and other gender minority respondents on population-based surveys. J.L. Herman (Ed.). Los Angeles, CA: The Williams Institute; 2014. https://williamsinstitute.law.ucla.edu/wp-content/uploads/Survey-Measures-Trans-GenIUSS-Sep-2014.pdf. Accessed 26 Apr 2022.
Marshall BDL, Kerr T, Shoveller JA, Patterson TL, Buxton JA, Wood E. Homelessness and unstable housing associated with an increased risk of HIV and STI transmission among street-involved youth. Health Place. 2009;15:783–90. https://doi.org/10.1016/j.healthplace.2008.12.005.
Lesko CR, Keil AP, Moore RD, Chander G, Fojo AT, Lau B. Measurement of current substance use in a cohort of HIV-infected persons in continuity HIV care, 2007–2015. Am J Epidemiol. 2018;187:1970–9. https://academic.oup.com/aje/article/187/9/1970/4986741
Kozak MS, Mugavero MJ, Ye J, Aban I, Lawrence ST, Nevin CR, et al. Patient reported outcomes in routine care: advancing data capture for HIV cohort research. Clin Infect Dis. 2012;54:141–7. https://doi.org/10.1093/cid/cir727.
Howe CJ, Cole SR, Napravnik S, Kaufman JS, Adimora AA, Elston B, et al. The role of at-risk alcohol/drug use and treatment in appointment attendance and virologic suppression among HIV + African Americans. AIDS Res Hum Retroviruses. 2014;30:233–40. https://doi.org/10.1089/AID.2013.0163.
Armed Forces Health Surveillance Division. Alcohol Related Disorders. 2022. https://www.health.mil/Reference-Center/Publications/2019/03/01/Alcohol-Related-Disorders. Accessed 26 Apr 2022.
Bush K, Kivlahan DR, McDonell MB, Fihn SD, Bradley KA. The AUDIT alcohol consumption questions (AUDIT-C): an effective brief screening test for problem drinking. Arch Intern Med. 1998;158(16):1789–95. https://doi.org/10.1001/archinte.158.16.1789.
Parrish C, Whitney BM, Nance RM, Puttkammer N, Fishman P, Christopoulos K, et al. Substance use and HIV stage at entry into care among people with HIV. Arch Public Heal. 2021;79:153. https://doi.org/10.1186/s13690-021-00677-2.
Pettit AC, Bian A, Schember CO, Rebeiro PF, Keruly JC, Mayer KH, et al. Development and validation of a multivariable prediction model for missed HIV health care provider visits in a large US clinical cohort. Open Forum Infect Dis. 2021;8(7). https://doi.org/10.1093/ofid/ofab130
WHO Assist Working Group. The alcohol, smoking and substance involvement screening test (ASSIST): development, reliability and feasibility. Addiction. 2002;97:1183–94. https://doi.org/10.1046/j.1360-0443.2002.00185.x.
Kroenke K, Spitzer RL, Williams JBW. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16(9):606–13. https://doi.org/10.1046/j.1525-1497.2001.016009606.x.
Fojo AT, Lesko CR, Calkins KL, Moore RD, McCaul ME, Hutton HE, et al. Do symptoms of depression interact with substance use to affect HIV continuum of care outcomes? AIDS Behav. 2019;23:580–91. https://doi.org/10.1007/s10461-018-2269-0.
Spitzer RL, Kroenke K, Williams JBW. Validation and utility of a self-report version of PRIME-MD: the PHQ Primary Care Study. J Am Med Assoc. 1999;282(18):1737–44. https://doi.org/10.1001/jama.282.18.1737.
Pence BW, Mills JC, Bengtson AM, Gaynes BN, Breger TL, Cook RL, et al. Association of increased chronicity of depression with HIV appointment attendance, treatment failure, and mortality among HIV-infected adults in the United States. JAMA Psychiat. 2018;75:379. https://doi.org/10.1001/jamapsychiatry.2017.4726.
Nassel A, Wilson-Barthes M, Howe CJ, Napravnik S, Agil D, Mugavero MJ, et al. Characterizing the neighborhood risk environment in multisite clinic-based cohort studies: a practical geocoding and data linkages protocol for protected health information. PLoS One. 2022;17(12):e0278672. https://doi.org/10.1371/journal.pone.0278672.
ESRI. Crime summary sample report. 2021. https://doc.arcgis.com/en/esri-demographics/data/crime-indexes.htm. Accessed 26 Apr 2022.
Chiba Y, Vanderweele TJ. A simple method for principal strata effects when the outcome has been truncated due to death. Am J Epidemiol. 2011;173(7):745–51. https://doi.org/10.1093/aje/kwq418.
VanderWeele TJ. Principal stratification - uses and limitations. Int J Biostat. 2011;7(1):28. https://doi.org/10.2202/1557-4679.1329.
Zou G, Donner A. Extension of the modified Poisson regression model to prospective studies with correlated binary data. Stat Methods Med Res. 2013;22:661–70. https://doi.org/10.1177/0962280211427759.
Miglioretti DL, Heagerty PJ. Marginal modeling of nonnested multilevel data using standard software. Am J Epidemiol. 2006;165:453–63. https://doi.org/10.1093/aje/kwk020.
Howe CJ, Cole SR, Westreich DJ, Greenland S, Napravnik S, Eron JJ. Splines for trend analysis and continuous confounder control. Epidemiology. 2011;22(6):874–5. https://doi.org/10.1097/EDE.0b013e31823029dd.
Wasserstein RL, Schirm AL, Lazar NA. Moving to a world beyond “ p < 0.05.” Am Stat. 2019;73:1–19. https://www.tandfonline.com/doi/full/10.1080/00031305.2019.1583913
Amrhein V, Greenland S, McShane BB. Statistical significance gives bias a free pass. Eur J Clin Invest. 2019;49:e13176. https://doi.org/10.1111/eci.13176
Greenland S, Senn SJ, Rothman KJ, Carlin JB, Poole C, Goodman SN, et al. Statistical tests, P values, confidence intervals, and power: a guide to misinterpretations. Eur J Epidemiol. 2016;31:337–50. https://doi.org/10.1007/s10654-016-0149-3.
Amrhein V, Greenland S, McShane B. Scientists rise up against statistical significance. Nature. 2019;567:305–7. https://doi.org/10.1038/d41586-019-00857-9.
Brewer R, Hood KB, Hotton A, Moore M, Spieldenner A, Daunis C, et al. Associations between experienced HIV stigma, resulting consequences, and the HIV care continuum: moderating effects of two resilience characteristics among persons living with HIV (PLWH) in Louisiana. J Racial Ethn Heal Disparities. 2022;9:9–22. https://doi.org/10.1007/s40615-020-00925-1.
Fletcher FE, Sherwood NR, Rice WS, Yigit I, Ross SN, Wilson TE, et al. Resilience and HIV treatment outcomes among women living with HIV in the United States: a mixed-methods analysis. AIDS patient care STDS. 2020;34(8):356–66. https://doi.org/10.1089/apc.2019.0309.
Gebo KA, Moore RD, Fleishman JA. The HIV research network: a unique opportunity for real time clinical utilization analysis in HIV. Hopkins HIV Rep. 2003;15:5–6.
Brewer R, Hood KB, Moore M, Spieldenner A, Daunis C, Mukherjee S, et al. An exploratory study of resilience, HIV-related stigma, and HIV care outcomes among men who have sex with men (MSM) living with HIV in Louisiana. AIDS Behav. 2020;24(7):2119–29. https://doi.org/10.1007/s10461-020-02778-5.
Dantzer C, Swendsen J, Maurice-Tison S, et al. Anxiety and depression in juvenile diabetes: a critical review. Clin Psychol Rev. 2003;23(6):787–800. https://doi.org/10.1016/s0272-7358(03)00069-2.
Wright IA, Reid R, Shahid N, et al. Neighborhood characteristics, intersectional discrimination, mental health, and HIV outcomes among Black women living with HIV, southeastern United States, 2019–2020. Am J Public Health. 2022;112(S4):S433–43. https://doi.org/10.2105/AJPH.2021.306675.
Simoni JM, Huh D, Wilson IB, et al. Racial/ethnic disparities in ART adherence in the United States: findings from the MACH14 study. J Acquir Immune Defic Syndr. 2012;60(5):466–72. https://doi.org/10.1097/QAI.0b013e31825db0bd.
Levengood TW, Yoon GH, Davoust MJ, Ogden SN, Marshall BDL, Cahill SR, et al. Supervised injection facilities as harm reduction: a systematic review. Am J Prev Med. 2021;61:738–49. https://doi.org/10.1016/j.amepre.2021.04.017.
Acknowledgements
The authors gratefully acknowledge the UNC and UAB clinic staff, the GIS data analyst at UAB, and study participants for their time and valuable contributions to this study.
Funding
Research reported in this publication was supported by the National Institute of Mental Health of the National Institutes of Health under Award Number R01MH112386 and by the National Institute of Allergy and Infectious Diseases of the National Institutes of Health under Award Number P30AI042853. One hundred percent of the total project costs are financed with Federal money. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
CJH and AJD obtained the funding for the project leading to this publication. CJH, AJD, JH, MM, SN, MC, JF, SD, and VE made substantial contributions to the study’s conception and design. Data acquisition was performed by DA, SD-S, BJ, MW-B, and CJH. Data analysis was performed by JWP with input from CJH and JH. The first draft of the manuscript was written by MW-B and JWP, and all authors revised the manuscript critically for important intellectual content. All authors gave the final approval of the version to be published, and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Ethics Approval
This study was performed in line with the principles of the Declaration of Helsinki. All human subjects activities were approved by Institutional Review Boards (IRB) at UAB (Protocol #: IRB-300001171), UNC at Chapel Hill (Protocol #: 17–2584), and Brown University (Protocol #: 1707001833) as relevant.
Consent to Participate
Participants provided written informed consent to participate in each sub-study.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Park, J.W., Wilson-Barthes, M.G., Dulin, A.J. et al. Multilevel Resilience and HIV Virologic Suppression Among African American/Black Adults in the Southeastern United States. J. Racial and Ethnic Health Disparities 11, 313–325 (2024). https://doi.org/10.1007/s40615-023-01520-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40615-023-01520-w