Abstract
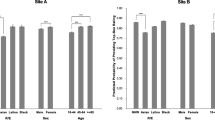
Our study investigates whether levels of motivation and barriers to participation in clinical trials vary with patients’ education and income. A self-administered survey asked outpatients to rank potential influential factors on a “0” to “4” significance scale for their motivation to participate in clinical trials. Principal component analysis (PCA), analysis of variance (ANOVA), Kruskal-Wallis, and Mann-Whitney U tests analyzed the impact of race, education, and income on their motivation to participate. Analysis included 1841 surveys; most respondents had a high school education or some college, and listed annual income < $30,000. There was a significant interaction between race and income on our motivation scale 1 scores (p = .0261). Compared with their counterparts, subjects with less education/lower income ranked monetary compensation (p = .0420 and p < .0001, respectively) as a higher motivator. Minorities and patients with less education and lower income appear to be more influenced by their desire to please the doctor, the race and sex of the doctor, and the language spoken by the doctor being the same as theirs. For all races, education appeared to have a direct relationship with motivation to participate, except for African-Americans, whose motivation appeared to decline with more education. Income appeared to have an inverse relationship with motivation to participate for all races.


Similar content being viewed by others
References
Colby SL, Ortman JM. Projections of the size and composition of the U.S. population: 2014 to 2060, Population Estimates and Projections, Current Population Reports. In: United States Census Bureau, 2014 National Population Projections: Publications. 2015. http://www.census.gov/population/projections/data/national/2014/publications.html. Accessed 14 Sept 2016.
National Institutes of Health. AHRQ policy on the inclusion of priority populations in research. In: Notice #NOT-HS-03-010. Department of Health and Human Services, National Institutes of Health. 2003. http://grants.nih.gov/grants/guide/notice-files/NOT-HS-03-010.html. Accessed 14 Sept 2016.
National Institutes of Health. NIH policy and guidelines on the inclusion of women and minorities as subjects in clinical research (amendment to the 1994 Federal Register). In: Notice #NOT-OD-02-001. 2001. http://grants.nih.gov/grants/guide/notice-files/NOT-OD-02-001.html. Accessed 14 Sept 2016.
Newman PA, Duan N, Roberts KJ, Seiden D, Rudy ET, Swendeman D, et al. HIV vaccine trial participation among ethnic minority communities: barriers, motivators, and implications for recruitment. J Acquir Immune Defic Syndr. 2006;41(2):210–7. https://doi.org/10.1097/01.qai.0000179454.93443.60.
Chen MS Jr, Lara PN, Dang JH, Paterniti DA, Kelly K. Twenty years post-NIH Revitalization Act: enhancing minority participation in clinical trials (EMPaCT): laying the groundwork for improving minority clinical trial accrual: renewing the case for enhancing minority participation in cancer clinical trials. Cancer. 2014;120(Suppl 7):1091–6. https://doi.org/10.1002/cncr.28575.
Eshera N, Itana H, Zhang L, Soon G, Fadiran EO. Demographics of clinical trials participants in pivotal clinical trials for new molecular entity drugs and biologics approved by FDA from 2010 to 2012. Am J Ther. 2015;22(6):435–55. https://doi.org/10.1097/MJT.0000000000000177.
Fisher JA, Kalbaugh CA. Challenging assumptions about minority participation in US clinical research. Am J Public Health. 2011;101(12):2217–22. https://doi.org/10.2105/AJPH.2011.300279.
Corbie-Smith G, Thomas SB, St George DM. Distrust, race, and research. Arch Intern Med. 2002;162(21):2458–63. https://doi.org/10.1001/archinte.162.21.2458.
Giuliano AR, Mokuau N, Hughes C, Tortolero-Luna G, Risendal B, Ho RCS, et al. Participation of minorities in cancer research: the influence of structural, cultural, and linguistic factors. Ann Epidemiol. 2000;10(8 Suppl):S22–34. https://doi.org/10.1016/S1047-2797(00)00195-2.
Hornbulm A. Acres of skin: human experiments at Holmesburg Prison. A true story of abuse and exploitation in the name of medical science. 1st ed. Medical History: Routledge; 1998.
Stones M, McMillan J. Payment for participation in research: a pursuit for the poor? J Med Ethics 2010;36(1):34–6, DOI: https://doi.org/10.1136/jme.2009.030965.
Grann VR, Jacobson JS, Troxel AB, Hershman D, Karp J, Myers C, et al. Barriers to minority participation in breast carcinoma prevention trials. Cancer. 2005;104(2):374–9. https://doi.org/10.1002/cncr.21164.
Ford JG, Howerton MW, Lai GY, Gary TL, Bolen S, Gibbons MC, et al. Barriers to recruiting underrepresented populations to cancer clinical trials: a systematic review. Cancer. 2008;112(2):228–42. https://doi.org/10.1002/cncr.23157.
Langford A, Resnicow K, An L. Clinical trial awareness among racial/ethnic minorities in HINTS 2007: sociodemographic, attitudinal, and knowledge correlates. J Healthc Commun. 2010;15(Suppl 3):92–101. https://doi.org/10.1080/10810730.2010.525296.
Braunstein JB, Sherber NS, Schulman SP, Ding EL, Powe NR. Race, medical researcher distrust, perceived harm, and willingness to participate in cardiovascular prevention trials. Medicine (Baltimore). 2008;87(1):1–9. https://doi.org/10.1097/MD.0b013e3181625d78.
Gross CP, Filardo G, Mayne ST, Krumholz H. The impact of socioeconomic status and race on trial participation for older women with breast cancer. Cancer. 2005;103(3):483–91. https://doi.org/10.1002/cncr.20792.
Svensson K, Ramírez OF, Peres F, Barnett M, Claudio L. Socioeconomic determinants associated with willingness to participate in medical research among a diverse population. Contemp Clin Trials. 2012;33(6):1197–205. https://doi.org/10.1016/j.cct.2012.07.014.
Hoberman A, Shaikh N, Bhatnagar S, Haralam MA, Kearney D, Colborn DK, et al. Factors that influence parental decisions to participate in clinical research: consenters vs nonconsenters. JAMA Pediatr. 2013;167(6):561–6. https://doi.org/10.1001/jamapediatrics.2013.1050.
Jiminez R, Zhang B, Joffe S, Nilsson M, Rivera L, Mutchler J, et al. Clinical trial participation among ethnic/racial minority and majority patients with advanced cancer: what factors most influence enrollment? J Palliat Med. 2013;16(3):256–62. https://doi.org/10.1089/jpm.2012.0413.
Bernstein SL, Feldman J. Incentive to participate in clinical trials: practical and ethical considerations. Am J Emerg Med. 2015;33(9):1197–200. https://doi.org/10.1016/j.ajem.2015.05.020.
Walter JK, Burke JF, Davis MM. Research participation by low-income and racial/ethnic minority groups: how payment may change the balance. Clin Transl Sci. 2013;6(5):363–71. https://doi.org/10.1111/cts.12084.
Kurt A, Semler L, Jacoby JL, Johnson MB, Careyva BA, Stello B, et al. Racial differences among factors associated with participation in clinical research trials. J Racial Ethn Health Disparities. 2016;104(2):1–10.
Carifio J, Perla RJ. Ten common misunderstandings, misconceptions, persistent myths and urban legends about Likert scales and Likert response formats and their antidotes. J Social Sci. 2007;3(3):106–16.
Dinno A. Nonparametric pairwise multiple comparisons in independent groups using Dunn’s test. Stata J. 2015;15(1):292–300.
Kurt A, Kincaid HM, Curtis C, Semler L, Meyers M, Johnson M, et al. Factors influencing participation in clinical trials: emergency medicine vs. other specialties. West JEM. 2017;18(5):846–55.
Jagsi R, Motomura AR, Amarnath S, Jankovic A, Sheets N, Ubel PA. Under-representation of women in high-impact published clinical cancer research. Cancer. 2009;115(14):3293–301. https://doi.org/10.1002/cncr.24366.
Murthy VH, Krumholz HM, Gross CP. Participation in cancer clinical trials: race-, sex, and age-based disparities. JAMA. 2004;291(22):2720–6. https://doi.org/10.1001/jama.291.22.2720.
George S, Duran N, Norris K. A systematic review of barriers and facilitators to minority research participation among African American, Latinos, Asian Americans and Pacific Islanders. Am J Public Health. 2014;104(2):e16–31. https://doi.org/10.2105/AJPH.2013.301706.
Street RL, O’Malley KJ, Cooper LA, Haidet P. Understanding concordance in patient-physician relationships: personal and ethnic dimensions of shared identity. Ann Fam Med. 2008;6(3):198–205. https://doi.org/10.1370/afm.821.
Albrecht TL, Eggly SS, Gleason ME, Harper FW, Foster TS, Peterson AM, et al. Influence of clinical communication on patients’ decision making on participation in clinical trials. J Clin Oncol. 2008;26(16):2666–73. https://doi.org/10.1200/JCO.2007.14.8114.
Hooghe M, Marien S, de Vroome T. The cognitive basis of trust. The relation between education, cognitive ability, and generalized and political trust. Intelligence. 2012;40(6):604–13. https://doi.org/10.1016/j.intell.2012.08.006.
Katz RV, Green BL, Kressin NR, Green BL, Wang MQ, James SA, et al. Exploring the “legacy” of the Tuskegee Syphilis study: a follow-up study from the Tuskegee legacy project. J Natl Med Assoc. 2009;101(2):179–83. https://doi.org/10.1016/S0027-9684(15)30833-6.
Emmons WR, Noeth BJ, Federal Reserve Bank of St. Louis. Why didn’t higher education protect Hispanic and Black wealth? In the Balance. 2015;12:1–3. https://www.stlouisfed.org/publications/in-the-balance/issue12-2015/why-didnt-highereducation-protect-hispanic-and-black-wealth. Accessed 14 Sept 2016.
Patten E, Fact tank news in the numbers. Racial, gender wage gaps persist in U.S. despite some progress. Pew Research Center. 2016. http://www.pewresearch.org/fact-tank/2016/07/01/racial-gender-wage-gaps-persist-in-u-s-despite-some-progress/
Kurt A, Semler L, Meyers M, Glenn-Porter B, Jacoby JL, Stello B. Research professionals’ perspectives, barriers and recommendations regarding minority participation in clinical trials. J Racial Ethn Health Disp. 2016;4(6):1–9.
Powell JH. Clinical trial diversity, drug development, and health disparities: A perspective from Project IMPACT. Monitor (newsletter). 2008;12:29–33. http://www.africanamericanhealthmatters.com/uploads/JPowell-Clinicaltrialdiversity-drugdevelopment-healthdisparities.pdf. Accessed April 25, 2016
Valcarcel M, Diaz C, Santiago-Borrero PJ. Training and retaining of underrepresented minority physician scientists—a Hispanic perspective: NICHD-AAP workshop on research in neonatology. J Perinatol. 2006;26(Suppl 2):S49–52. https://doi.org/10.1038/sj.jp.7211526.
Jeste DV, Twamley EW, Cardenas V, Lebowitz B, Reynolds CF III. A call for training the trainers: focus on mentoring to enhance diversity in mental health research. Am J Public Health. 2009;99(Suppl 1):S31–7. https://doi.org/10.2105/AJPH.2008.154633.
Getz K, Faden L. Racial disparities among clinical research investigators. Am J Ther. 2008;15(1):3–11. https://doi.org/10.1097/MJT.0b013e31815fa75a.
National Institutes of Health FAQ, ClinicalTrials.gov - Clinical Trial Phases. Department of Health and Human Services, U.S. National Library of Medicine. 2008. http://www.nlm.nih.gov/services/ctphases.html. Accessed 2 Jun 2017.
Adeyemi OF, Evans A, Bahk M. HIV-infected adults from minority ethnic groups are willing to participate in research if asked. AIDS Patient Care STDs. 2009;23(10):859–65. https://doi.org/10.1089/apc.2009.0008.
Gollin L, Harrigan R, Perez J, Easa D. Improving Hawaiian and Filipino involvement in clinical research opportunities qualitative findings from Hawai’i. Ethn Dis. 2005;15(4 Suppl 5):S5-111. 9
Farmer DF, Jackson SA, Camacho F, Hall M. Attitudes of African American and low socioeconomic status white women toward medical research. J Health Care Poor Underserved. 2007;18(1):85–99. https://doi.org/10.1353/hpu.2007.0008.
Acknowledgements
We acknowledge Tyler Adams, BS for figure development as well as Micaela Wilson, BA and Manuel Colón, BS for editing and formatting material for this manuscript. We also thank Matthew Meyers, BS for his help with data entry and maintenance of the database.
Funding
This study was supported by an unrestricted research grant funding from a non-philanthropic trust, the Anne and Carl Anderson Trust.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All persons listed as authors meet the criteria for authorship established by the ICMJE and take responsibility for the article’s content.
Ethical Approval
All procedures performed in this study involving human subjects were in accordance with the ethical standards of the IRB and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
This anonymous and voluntary survey study was IRB-approved as “exempt;” therefore, an alteration of informed consent was granted.
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Kurt, A., Kincaid, H., Semler, L. et al. Impact of Race Versus Education and Race Versus Income on Patients’ Motivation to Participate in Clinical Trials. J. Racial and Ethnic Health Disparities 5, 1042–1051 (2018). https://doi.org/10.1007/s40615-017-0452-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40615-017-0452-z