Abstract
Purpose of Review
Cancer is a major public health problem worldwide. In aggressive cancers, which are heterogeneous in nature, there exists a paucity of targetable molecules that can be used to predict outcome and response to therapy in patients, especially those in the high-risk category with a propensity to relapse following chemotherapy. This review addresses the challenges pertinent to treating aggressive cancer cells with inherent stem cell properties, with a special focus on triple-negative breast cancer (TNBC).
Recent Findings
Plasticity underlies the cancer stem cell (CSC) phenotype in aggressive cancers like TNBC. Progenitors and CSCs implement similar signaling pathways to sustain growth, and the convergence of embryonic and tumorigenic signaling pathways has led to the discovery of novel oncofetal targets, rigorously regulated during normal development, but aberrantly reactivated in aggressive forms of cancer.
Summary
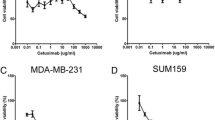
Translational studies have shown that Nodal, an embryonic morphogen, is reactivated in aggressive cancers, but not in normal tissues, and underlies tumor growth, invasion, metastasis, and drug resistance. Front-line therapies do not inhibit Nodal, but when a combinatorial approach is used with an agent such as doxorubicin followed by anti-Nodal antibody therapy, significant decreases in cell growth and viability occur. These findings are of special interest in the development of new therapeutic interventions that target the stem cell properties of cancer cells to overcome drug resistance and metastasis.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Lapidot T, Sirard C, Vormoor J, Murdoch B, Hoang T, Caceres-Cortes J, et al. A cell initiating human acute myeloid leukaemia after transplantation into SCID mice. Nature. 1994;367(6464):645–8.
Malhotra GK, Zhao X, Band H, Band V. Histological, molecular and functional subtypes of breast cancers. Cancer Biol Ther. 2010;10:955–60.
Rakha EA, El-Sayed ME, Lee AH, Elston CW, Grainge MJ, Hodi Z, et al. Prognostic significance of Nottingham histologic grade in invasive breast carcinoma. J Clin Oncol. 2008;26:3153–8.
Meijnen P, Peterse JL, Antonini N, Rutgers EJ, van de Vijver MJ. Immunohistochemical categorisation of ductal carcinoma in situ of the breast. Br J Cancer. 2008;98:137–42.
Perou CM, Sorlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA, et al. Molecular portraits of human breast tumors. Nature. 2000;406:747–52.
Al-Hajj M, Wicha MS, Benito-Hernandez A, Morrison SJ, Clarke MF. Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci U S A. 2003;100:3983–8.
•• Strizzi L, Hardy KM, Margaryan NV, Hillman DW, Seftor EA, Chen B, et al. Potential for the embryonic morphogen Nodal as a prognostic and predictive biomarker in breast cancer. Breast Cancer Res. 2012;14(3):R75. This study demonstrates for the first time the promise of Nodal as a prognostic and predictive biomarker in breast cancer
• Kirsammer G, Strizzi L, Margaryan NV, Gilgur A, Hyser M, Atkinson J, et al. Nodal signaling promotes a tumorigenic phenotype in human breast cancer. Semin Cancer Biol. 2014;29:40–50. This study was the first to directly downregulate Nodal and examine the consequences on multiple signaling pathways
•• Bodenstine TM, Chandler GS, Reed DW, Margaryan NV, Gilgur A, Atkinson J, et al. Nodal expression in triple-negative breast cancer: cellular effects of its inhibition following doxorubicin treatment. Cell Cycle. 2016;15(9):1295–302. This study demonstrated the potential efficacy and rationale for using anti-Nodal therapy in combination with a front-line therapy, doxorubicin, for treating triple-negative breast cancer
•• Bittner M, Meltzer P, Chen Y, Jiang Y, Seftor E, Hendrix M, et al. Molecular classification of cutaneous malignant melanoma by gene expression profiling. Nature. 2000;406(6795):536–40. This original work is an extensive survey that classifies cutaneous, malignant melanoma based on gene expression profiling
Kirschmann DA, Seftor EA, Nieva DR, Mariano EA, Hendrix MJC. Differentially expressed genes associated with the metastatic phenotype in breast cancer. Breast Cancer Res Treat. 1999;55(2):127–36.
• Hendrix MJC, Seftor EA, Kirschmann DA, Seftor REB. Molecular biology of breast cancer metastasis. Molecular expression of vascular markers by aggressive breast cancer cells. Breast Cancer Res. 2000;2(6):417–22. This is one of the earliest studies demonstrating the expression of vascular makers by aggressive breast cancer cells pertinent to vasculogenic mimicry
• Hendrix MJC, Seftor EA, Hess AR, Seftor REB. Vasculogenic mimicry and tumour-cell plasticity: lessons from melanoma. Nat Rev Cancer. 2003;3:411–21. This review summarizes the evidence supporting vasculogenic mimicry by aggressive tumor cells
•• van de Schaft DW, Seftor REB, Seftor EA, Hess AR, Gruman LM, Kirschmann DA, et al. Effects of angiogenesis inhibitors on vascular network formation by human endothelial and melanoma cells. J Natl Cancer Inst. 2004;96(19):1473–7. This was the first study to demonstrate how and why angiogenesis inhibitors may not be effective in inhibiting vasculogenic mimicry in melanoma cells
van der Schaft DW, Hillen F, Pauwels P, Kirschmann DA, Castermans K, Egbrink MG, et al. Tumor cell plasticity in Ewing sarcoma, an alternative circulatory system stimulated by hypoxia. Cancer Res. 2005;65(24):11520–8.
•• Wagenblast E, Soto M, Gutiérrez-Ángel S, Hartl CA, Gable AL, Maceli AR, et al. A model of breast cancer heterogeneity reveals vascular mimicry as a driver of metastasis. Nature. 2015;520(7547):358–62. This key study demonstrated how vascular mimicry drives metastasis with respect to breast cancer heterogeneity
• Charafe-Jauffret E, Ginestier C, Iovino F, Wicinski J, Cervera N, Finetti P, et al. Breast cancer cell lines contain functional cancer stem cells with metastatic capacity and a distinct molecular signature. Cancer Res. 2009;69(4):1302–13. This study correlated breast cancer metastatic capacity with the presence of functional cancer stem cells and a distinct molecular signature
Charafe-Jauffret E, Ginestier C, Iovino F, Tarpin C, Diebel M, Esterni B, et al. Aldehyde dehydrogenase 1-positive cancer stem cells mediate metastasis and poor clinical outcome in inflammatory breast cancer. Clin Cancer Res. 2010;16(1):45–55.
Al-Hajj M, Wicha MS, Benito-Hernandez A, Morrison SJ, Clarke MF. Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci U S A. 2003;100(7):3983–98.
Dontu G, Jackson KW, McNicholas E, Kawamura MJ, Abdallah WM, Wicha MS. Role of Notch signaling in cell-fate determination of human mammary stem/progenitor cells. Breast Cancer Res. 2004;6(6):R605–15.
Korkaya H, Wicha MS. HER-2, notch, and breast cancer stem cells: targeting an axis of evil. Clin Cancer Res. 2009;15(6):1845–7.
D’Angelo RC, Ouzounova M, Davis A, Choi D, Tchuenkam SM, Kim G, et al. Notch reporter activity in breast cancer cell lines identifies a subset of cells with stem cell activity. Mol Cancer Ther. 2015;14(3):779–87.
Arnold SJ, Robertson EJ. Making a commitment: cell lineage allocation and axis patterning in the early mouse embryo. Nat Rev Mol Cell Biol. 2009;10:91–103.
Schier AF. Nodal morphogens. Cold Spring Harb Perspect Biol 2009;1:a003459. Pauklin S, Vallier L. Activin/Nodal signalling in stem cells. Development 2015;142:607–619.
• Postovit LM, Seftor EA, Seftor REB, Hendrix MJC. Targeting Nodal in malignant melanoma cells. Expert Opin Ther Targets. 2007;11(4):497–505. This was one of the first studies to identify Nodal as a potential therapeutic target for melanoma
• Schier AF, Shen MM. Nodal signalling in vertebrate development. Nature. 2000;403(6768):385–9. This is an excellent review of the importance of Nodal in embryogenesis
•• Topczewska JM, Postovit LM, Margaryan NV, Sam A, Hess AR, Wheaton WW, et al. Embryonic and tumorigenic pathways converge via Nodal signaling: role in melanoma aggressiveness. Nat Med. 2006;12(8):925–32. This was the landmark paper introducing Nodal, an embryonic morphogen, being reactivated in aggressive melanoma cells
Postovit LM, Costa FF, Bischof JM, Seftor EA, Wen B, Seftor REB, et al. The commonality of plasticity underlying multipotent tumor cells and embryonic stem cells. J Cellular Bioch. 2007;101:908–17.
•• Postovit LM, Margaryan NV, Seftor EA, Kirschmann DA, Lipavsky A, Wheaton WW, et al. Human embryonic stem cell microenvironment suppresses the tumorigenic phenotype of aggressive cancer cells. Proc Natl Acad Sci U S A. 2008;105(11):4329–34. This study introduced a novel model showing that Nodal signaling in aggressive cancer cells can be regulated by signaling molecules, such as Lefty, in a human embryonic stem cell environment
Costa FF, Seftor EA, Bischof JM, Kirschmann DA, Strizzi L, Arndt K, et al. Epigenetically reprogramming metastatic tumor cells with an embryonic microenvironment. Epigenomics. 2009;1(2):387–98.
Strizzi L, Hardy KM, Seftor EA, Costa FF, Kirschmann DA, Seftor RE, et al. Development and cancer: at the crossroads of Nodal and Notch signaling. Cancer Res. 2009;69:7131–4.
Strizzi L, Postovit LM, Margaryan NV, Lipavsky A, Gadiot J, Blank C, et al. Nodal as a biomarker for melanoma progression and a new therapeutic target for clinical intervention. Expert Rev Dermatol. 2009;4:67–78.
Quail DF, Siegers GM, Jewer M, Postovit LM. Nodal signalling in embryogenesis and tumourigenesis. Int J Biochem Cell Biol. 2013;45:885–98.
Postovit LM, Margaryan NV, Seftor EA, Strizzi L, Seftor REB, Hendrix MJC. Plasticity underlying multipotent tumor stem cells. In: Bagley RG, Teicher BA, editors. Cancer drug discovery and development: stem cells and cancer. USA: Humana Press; 2009. p. 99–112.
• Lawrence MG, Margaryan NV, Loessner D, Collins A, Kerr KM, Turner M, et al. Reactivation of embryonic Nodal signaling is associated with tumor progression and promotes the growth of prostate cancer. Prostate. 2011;71:1198–09. This was the first study showing a role for Nodal signaling in tumor progression and growth in prostate cancer
Strizzi L, Sandomenico A, Margaryan NV, Foca A, Sanguigno L, Bodenstine TM, et al. Effects of a novel Nodal-targeting monoclonal antibody in melanoma. Oncotarget. 2015;6(33):34071–86.
Tomao F, Papa A, Zaccarelli E, Rossi L, Caruso D, Minozzi M, et al. Triple-negative breast cancer: new perspectives for targeted therapies. Onco Targets Ther. 2015;8:177–93.
Mahamodhossen YA, Liu W, Rong-Rong Z. Triple-negative breast cancer: new perspectives for novel therapies. Med Oncol. 2013;30:653.
Anders C, Carey LA. Understanding and treating triple-negative breast cancer. Oncology. 2008;22:1233–43.
• Hardy KM, Strizzi L, Margaryan NV, Gupta K, Murphy GF, Scolyer RA, et al. Targeting Nodal in conjunction with dacarbazine induces synergistic anticancer effects in metastatic melanoma. Mol Cancer Res. 2015;13(4):670–80. This study demonstrated the potential efficacy and rationale for using anti-Nodal therapy in combination with front-line therapies for treating melanoma
•• Hendrix MJC, Kandela I, Mazar AP, Seftor EA, Seftor RE, Margaryan NV, et al. Targeting melanoma with front-line therapy does not abrogate Nodal-expressing tumor cells. Lab Investig. 2017;97(2):176–86. This was the first study that demonstrated that treating melanoma patients with the front line therapy, iBRAF, did not decrease the expression of Nodal in the patients’ tissue.
Lonardo E, Hermann PC, Mueller MT, Huber S, Balic A, Miranda-Lorenzo I, et al. Nodal/Activin signaling drives self-renewal and tumorigenicity of pancreatic cancer stem cells and provides a target for combined drug therapy. Cell Stem Cell. 2011;9:433–46.
Ahrlund-Richter L, Hendrix MJC. Oncofetal signaling as a target for cancer therapy. Semin Cancer Biol. 2014;29:1–2.
Acknowledgements
Supported by the National Institute of General Medical Sciences U54GM104942 (NVM, EAS, REBS) and NIH/NCI R37CA59702 and RO1CA121205 (MJCH).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Naira V. Margaryan declares that she has no conflicts of interest. Elisabeth A. Seftor, Richard E.B. Seftor, and Mary J.C. Hendrix are listed as co-inventors on Nodal-related patents and/or disclosures.
Human and Animal Rights and Informed Consent
This article does not contain any raw data with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Molecular Biotechnology of Adult Stem Cells
Rights and permissions
About this article
Cite this article
Margaryan, N.V., Seftor, E.A., Seftor, R.E. et al. Targeting the Stem Cell Properties of Adult Breast Cancer Cells: Using Combinatorial Strategies to Overcome Drug Resistance. Curr Mol Bio Rep 3, 159–164 (2017). https://doi.org/10.1007/s40610-017-0067-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40610-017-0067-5