Abstract
Purpose of Review
The purpose of this review is to provide a better understanding of anaphylaxis pathophysiology and describe the underlying mechanisms, effector cells, and the potential biomarkers involved depending on the anaphylaxis endotypes.
Recent Findings
New insight into the potential relevance of pathways others than IgE-dependent anaphylaxis has been unraveled, as well as other biomarkers than tryptase, such as the role of platelet activation factor, basogranulin, dipeptidyl peptidase I, CCL-2, and other cytokines.
Summary
Gaining knowledge of all the mediators and cellular activation/communication pathways involved in each endotype of anaphylaxis will allow the application of precision medicine in patients with anaphylactic reactions, providing insights to the most appropriate approach in each case and helping to stratify severity and risk prediction.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Anaphylaxis is a severe, multisystem syndrome that is rapid in onset and potentially lethal, requiring immediate medical intervention. However, the diagnosis of anaphylaxis is still based on clinical symptoms after exposure to a potential allergen or event, given that no biomarker allows an unequivocal diagnosis of anaphylaxis [1, 2•]. Therefore, anaphylaxis remains under-recognized, undertreated, and poorly understood. The current gold standard laboratory test involves the measurement of serum total mast cell tryptase (MCT) during an acute phase followed by a baseline measurement at least 24 h after the reaction. However, even when sampling of tryptase is timely, an increase is not detectable in all cases [1, 2•].
Phenotypes of anaphylaxis are defined by clinical presentation and endotypes are based on cellular and molecular mechanisms involved during an anaphylactic reaction [1, 2•]. The activation of different pathways during an anaphylaxis will depend on which receptors are activated in the underlying effector cells and which mechanism and mediators are involved in the reaction.
This review summarizes the most recent knowledge of how different effector cells and their mediators are involved in anaphylaxis depending on the mechanism providing a better understanding of the pathophysiology of anaphylaxis and explores and discusses the role for new biomarkers of anaphylaxis beyond tryptase.
Endotypes and effector cells involved in anaphylaxis
The type of receptor activated in the different effector cells during an anaphylactic reaction will influence which mediators (tryptase, interleukin [IL]-6, …) are released and, subsequently, which can be potential diagnostic biomarkers [3••]. Depending on these cellular and molecular mechanisms, anaphylaxis is divided in different endotypes, summarized in Table 1.
The major molecular mechanism underlying anaphylaxis is the classic allergic IgE-mediated reaction involving mast cells (MC) and basophils. Both cell types express the high-affinity IgE receptor (FcεRI) on their surface and are considered the main effector cells in this pathologic event [4]. However, a considerable percentage of subjects do not show evidence of IgE-dependent immune activation; therefore, other effector cells and pathways must be involved in anaphylaxis. Nowadays, there is enough evidence to corroborate that IgG antibodies can induce anaphylaxis by binding to their different receptors (FcγR) [5••]. Such receptors are found in MC, basophils, neutrophils, monocytes, and macrophages conforming the major cellular types activated by this alternative pathway [3••]. In fact, some studies have shown that human IgG receptors are capable of activating macrophages and neutrophils to secrete platelet-activating factor (PAF), and then PAF can activate mast cells in vitro, reinforcing the role of this mediator in human anaphylaxis as described below [6]. Moreover, the amount of antibody and antigen seems to be crucial in determining IgG- or IgE-dependent anaphylaxis. Thus, to induce a similar response in IgG-dependent anaphylaxis, a higher dose of antigen is required compared to IgE-dependent [7]. Complement-mediated anaphylaxis has also been described in human anaphylaxis [8]. C3a and C5a, known as anaphylatoxins, are potent inflammatory mediators generated upon activation of the complement cascades. Some substances, such as peanuts, have been described to have the ability to rapidly activate complement, with production of large amounts of anaphylatoxins that stimulates macrophages, basophils, and MC which contribute to the induction of anaphylaxis [9, 10]. In addition, the contact and coagulation systems can be activated in anaphylaxis through immunological and non-immunological mechanisms [11].
Different non-immunologic activating paths have been described in anaphylaxis. Some drugs (e.g., opioids, neuromuscular blockers, quinolones) are also capable of activating MC and basophils, triggering degranulation, through the Mas-related G-protein coupled receptor member X2 (MRGPRX2) [12, 13]. This is a highly relevant pathway independent of specific immunoglobulins. External factors such as physical exercise, exposure to cold, or ultraviolet radiation may also act as elicitors of MC [14, 15••, 16]. Another endotype discovered due to the increasingly common use of monoclonal antibodies and chemotherapeutic agents is cytokine release reaction. These reactions are suspected to be mediated by tumor necrosis factor alpha (TNFα), IL-1β and IL-6 produced by MC, monocytes, and T cells. In some patients, the endotype could be mixed due to co-occurrence of IgE-mediated reactions and cytokine release reactions [4].
Considering all the above, anaphylaxis endotypes can be divided mainly in two different groups, immune-mediated anaphylaxis (IgE-dependent and non-IgE-dependent such as IgG-dependent, complement system, contact system, cytokine release reactions) and non-immunological anaphylaxis.
Mast cell
MC are hematopoietic cells that reside in different tissues (such as connective tissue, blood, skin, among others) and have an important role in inflammation. MC are the key effector cells in anaphylaxis [16, 17]. They are involved in IgE-mediated reactions (immediate hypersensitive reactions), due to cross-linking by allergens of specific IgE (sIgE) attached to its high-affinity receptor (FCeRI) expressed predominantly by MC and basophils [18]. This interaction, allergen-sIgE-MC, results in intracellular signaling that triggers the release of MC mediators. There are two mayor types of MC, MC (T) that are characterized by a large content in tryptase and MC (TC) which produce both tryptase and chymase [19]. Characteristically, MC express an array of different receptors, the activation of which may induce activation through diverse pathways, as explained above. Figure 1 outlines the different MC activating receptors and mediators.
The different MC activation receptors and mediators. FcϵRI, high affinity IgE receptor; FcϵRII (CD23), low affinity IgE receptor; FcγRIIa, IgG receptor; Mrgprs, Mas-related G protein-coupled receptor; Trk, tyrosine kinase receptor; TLR, Toll-like receptor; HR, histamine receptor; PAFR, plasmin activator factor receptor; NK-1R, neurokinin receptor 1; Tryp, tryptase; Hist, histamine; PAF, platelet activator factor; Chy, chymase; CPA3, carboxypeptidase A3; DPPI, dipeptidyl peptidase I/cathepsin C; CAT-G, cathepsin G; LTE4, lipoxygenase product E4; PGD2, prostaglandin D2; TXA2, thromboxane A2; HEP, heparin; FXII, factor XII; FXIIa, activated factor XII; CSF, stem cell factor; HMWK, high molecular weight kininogen; TNF-alpha, necrosis tumoral factor alpha. This image is created with BioRender.com
Basophil
Basophils are the largest and least common granular immune cells, representing about 0.5 to 1% of circulating leukocytes. Unlike MC, they are blood-circulating leukocytes and not tissue-resident cells [20, 21] Recent studies suggest that basophils activate complementary to MC and they play a key role in anaphylaxis, especially in food-induced anaphylaxis [22, 23]. Basophils share many mediators and receptors with MC [24] and are more commonly used to perform laboratory allergen-specific activation tests (BAT), since they are easy to sample from whole blood. BAT has been demonstrated useful to confirm IgE-mediated allergy and also to monitor allergen immunotherapy, in order to differentiate short-term desensitization versus sustained unresponsiveness to the allergen [25, 26].
Neutrophil, eosinophil, monocyte/macrophage, and platelet
The most recent studies on anaphylaxis have highlighted the role of other immune effector cells such as monocytes, macrophages, neutrophils, eosinophils, and platelets. Most of this evidence is based on murine models, so knowledge about their involvement in human anaphylaxis still remains scarce [27].
The implication of both monocytes and macrophages has been demonstrated in passive and active systemic anaphylaxis. They have been more related to IgG-dependent and cytokine release reaction endotypes [27,28,29]. However, especially neutrophils have gained much relevance as key cellular player eliciting non-IgE-dependent anaphylactic reactions [4, 30]. Evidence in patients has shown elevated circulating serum levels of neutrophil elastase and myeloperoxidase, major mediators stored in their granules. These results supported the existence of a neutrophil-associated IgG molecular mechanism associated with drug-induced anaphylaxis [31,32,33]. As represented in Table 1, neutrophils not only express FcγR, but also FcεRI and MRGPRX2.
MC and eosinophils share several receptors and mediators (Tables 1 and 2), so suspecting their involvement in anaphylaxis seems reasonable. Accumulation of eosinophils has been detected in passive cutaneous anaphylactic reactions in guinea pigs, as well as in spleens and coronary arteries from anaphylactic human cadavers [34, 35]. And lastly, but by no means less importantly, there is evidence of platelet contribution in anaphylactic reactions. The release of important mediators (Table 2) has been observed in both IgE- and IgG-dependent pathways [6, 36,37,38].
Taken together, this data indicate that many myeloid cells have a role in the physiopathology of anaphylaxis.
Endothelium
The endothelium consists of a single and extensive layer of endothelial cells (ECs). Molecularly, stabilization of the endothelial barrier depends on a series of connections, such as tight junctions (TJ) and adherent junctions (AJ). A dysfunctional endothelium is the cause of important cardiovascular diseases such as thrombosis, atherosclerosis, or hypertension [39]. A damaged endothelium has been observed in patients with mastocytosis [40] and, in acute inflammatory situations as in COVID-19 disease, ECs contribute as effector cells to the cytokine releases syndrome [41]. Therefore, endothelium is an important organ-cell in anaphylaxis playing a role not only in the control of fluids and the vascular tone but also as an activation surface for the coagulation, contact system, and complement [11, 42, 43]. ECs release relevant anaphylactic mediators such as nitric oxide (NO), together with other molecules (presumably cytokines or interleukins) which also released, contribute to the pool of mediators in the reaction [3••]. Specifically, anaphylactic shock depending on endothelial Gq/G11 has been characterized in mice [43]. However, the exact contribution of these cells during anaphylaxis is still not fully understood.
Mediators release in anaphylaxis
During anaphylaxis there is a rapid release of mediators from different sources, but most important, from MC and basophils. These mediators are proteases, sulfated polysaccharides, cytokines, chemokines, vasoactive agents (histamine, bradykinin) and proinflammatory lipid mediators, among others [44]. They are usually divided into three classes: preformed mediators (that are stored in cytoplasmic granules), newly generated in minutes (proinflammatory lipid mediators), and newly generated over hours (growth factors, cytokines, and chemokines). Table 2 summarizes the main mediators, their major cell sources, best sampling time, and the availability of commercial assays.
Preformed mediators
Preformed mediators are stored in cytoplasmatic granules of the effector cells (mainly MC and basophils) and include proteases (tryptase, chymase), highly sulfated polysaccharides (heparin and other proteoglycans), and histamine [45].
Histamine
Plasma histamine is an amine secreted by MC and basophils with vasoactive and pro-inflammatory functions. It is synthetized from the amino acid histidine and catalyzed by L-histidine decarboxylase. Histamine is stored in cytoplasmic granules and can be released when immunological or non-immunological activation occurs. There is evidence that neutrophils can also synthetize and release histamine under specific circumstances [46]. When MC/basophils are activated, histamine is released and it acts through its g-protein receptors (H1, H2, H3, and H4), causing arteriolar vasodilation, increased capillary permeability, bronchoconstriction, itching, swelling, rhinorrhea, urticaria, and rhinitis, among others [47]. The peak of histamine is 5–10 min from the onset of the reaction and is rapidly metabolized into methyl histamine by the N-methyltransferase or into imidazole acetaldehyde by diamine oxidase, and then secreted in urine [48]. In daily practice, histamine is not a good diagnostic biomarker for anaphylaxis due to its short half-life [4, 49].
Histamine metabolites
Methyl histamine and imidazole acetaldehyde had longer half-life compared to histamine and can be measured in urine or plasma after 30–60 min of the onset of the reaction [48]. Normal levels do not exclude a hypersensitivity reaction. Some false positive results may occur due to ingestion of foods with high concentrations of histamine (in example, in scombroidosis), endogenous production by other type of cells such as neurons or enterochromaffin-like cells in stomach, or by certain bacteria [24].
Tryptase
Tryptase is a neutral serine protease highly specific of MC, stored in large amounts in cytoplasmatic granules. It is currently used as a diagnostic biomarker in anaphylaxis. Also, basophils produce tryptase but in much lower amounts. Tryptase provides information about MC activation, distribution, and number so it is useful for diagnosis and follow-up of mast cell diseases [50, 51].
There are two biologically important forms of tryptase, alpha-protryptase and beta-tryptase. The alpha-protryptase is present in serum as an inactive proenzyme and is secreted constitutively. Beta-tryptase, on the other hand, is the functional tryptase, a mature tetramer stabilized by proteoglycans, stored predominantly in MC secretory granules and released by active MC. Tryptase acts binding to protease activated receptor type 2 (PAR-2, present in gastrointestinal, cardiovascular, neurons, among others). It is responsible of cleaving extracellular substrates, such as calcitonin gene-related peptide, kininogens, fibronectin, and vasoactive intestinal peptide; also, it is a potent growth factor for EC, airway smooth muscle cells, and fibroblasts [45, 52]. It also has a chemotactic effect on neutrophils and eosinophils via IL-8 [53]. During anaphylaxis, tryptase is responsible for vascular vasodilation (activating the contact system to generate bradykinin) and bronchial hyperresponsiveness [49].
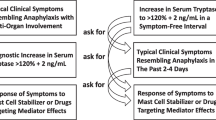
Basal tryptase in healthy individuals ranges from 0 to 11.4 ng/mL. Currently, it is accepted that levels of 120% of the baseline plus 2 ng/mL (1.2 × baseline tryptase + 2 ng/mL) is a significant increase, indicating MC activation (54, 55). In MC disorders, a basal tryptase > 20 ng/mL is considered as a minor criteria of mastocytosis [51].
During active MC degranulation in allergic reactions, β-tryptase is released. Levels of tryptase are increased from 15 min to 3 h after the anaphylaxis onset [54•, 56]. Sequential measurement of serum tryptase during anaphylaxis and basal serum levels increases sensitivity and specific of the diagnostic. Higher peak/baseline tryptase correlates with more severe symptoms [57]. Nevertheless, it is important to take into account that an increase is not detectable in all cases, for example, in food-induced anaphylaxis where tryptase does not increase as in drug-induced reactions [56, 58], but may still be useful for diagnosis. Current commercial methods measure immature alpha and beta tryptase, but as a diagnostic marker of anaphylaxis, it would be optimal to measure beta-tryptase alone, since this is the form released by MC.
Chymase
Chymase is a serine endopeptidase also found in MC (TC). It is usually released during MC/basophil activation, and its concentration in serum has been seen to be stable. There are no commercialized kits to measure chymase, so is not used routinely in anaphylactic reactions [59]. Chymase participates in local arterial pressure regulation due to the hydrolysis of angiotensin I into angiotensin II [60]; it also activates endothelin-1 and 2, leading to the production of metalloproteinases and cytokines [24]. It has been reported alternative methods to evaluate indirectly Chymase via alpha-2-macroglobulin, but is not systematically used [61]. Postmortem chymase concentrations in cases of fatal anaphylaxis have been shown to correlate with levels of tryptase [62].
Carboxypeptidase A3
Carboxypeptidase A3 is another metalloproteinase contained in MC and basophil granules [63]. Zhou et al. [64] measured levels of carboxypeptidase A3 in cases of anaphylaxis, systemic mastocytosis, and control groups. Significantly elevated levels were found 8 h after the onset of an allergic reaction compared to the control cohort. There are no available commercial methods for its quantification in daily practice.
Basogranulin
Basogranulin is a basophil granule protein that is released in parallel with histamine. It shares receptors and pathways with histamine [65]. Nowadays, there are no available methods for its measurement [66].
Heparin
Highly sulfated proteoglycan heparin is preformed in MC (predominantly) and also in basophil granules. It is stored in its active form with a negative charge. It has been reported that heparin has an important role in activating the plasma contact system [11, 67]. Due to its negative charge, it is capable of activating the FXII-dependent pathway and cleaving high molecular weight kininogen into bradykinin, leading to increased vascular permeability [11].
Dipeptidyl peptidase I (DPPI)/cathepsin C
Dipeptidyl peptidase I (DPPI)/cathepsin C is a member of the papain family proteases that is expressed in numerous cell types, including both MC and basophils. Levels of chymase and DDPI have been shown to increase and correlated during anaphylaxis, but do not correlate with tryptase [68, 69]. Its activity outside of cells is limited. There are no available measurement methods for its quantification currently.
Cathepsin G
Cathepsin G is a protease related to chymase and has similar functions (cleavage of angiotensin I to angiotensin II) [69]. It is found in MC granules. There are no available techniques to measure it.
Newly generated over minutes
Newly generated mediators are generated over minutes after the onset of stimuli by the different types of cells involved in the reactions.
Bradykinin (BK)
Is a nonapeptide from the kinin family with vasoactive functions that has been demonstrated to be released during anaphylaxis by activation of the contact system [11, 67]. When the contact system is activated by a negative charge (for instance heparin that is released in anaphylaxis), FXII is activated to FXIIa, which cleaves prekallikrein to kallikrein, which subsequently hydrolyses the high molecular weight kininogen to release BK. BK has a very short half-life, less than 1 min, due to its fast metabolism by the specific enzymes (angiotensin converting enzyme, also known as kininase II), dipeptidyl peptidase IV, carboxypeptidase N also known as kininase I, and neutral endopeptidase [70]. BK acts through its specific receptors, B1 and B2, inducing an increase of vascular permeability and plasma extravasation [71]. Currently, it is not possible to measure BK in daily practice, due to its short half-life, so indirect methods are used to assess it in research setting.
Lipid mediators
Lipid mediators are synthetized de novo in activated MC and basophils. They derive from arachidonic acid and have vasoactive and pro-inflammatory functions.
Lipoxygenase products: leukotriene E4
Leukotriene E4 (LTE4) is produced by activated MC, neutrophils, eosinophils, and macrophages. It is derived from the precursor LTA4, converted to LTD4 and finally LTE4. LTE4 is the most stable metabolite of the lipoxygenase products and binds to three different receptors (CysLTR1, CysLTR2, CysLTR3). Its activation causes airway constriction, smooth muscle contraction, and increased vascular permeability [24]. LTE4 can be measured in urine, but due to their rapid metabolism, LTC4 and LTD4 cannot be measured [72].
Cyclooxygenase products: prostaglandins and thromboxanes
Generation of metabolites of arachidonic acid (thromboxane A2, thromboxane B2, prostaglandin (PG) D2, PGE2, PGF2) in anaphylaxis has been demonstrated in several studies with animals and humans [73, 74]. PGD2 is the main cyclooxygenase product formed in activated MC, also produced by neutrophils, monocytes, macrophages, and platelets, but not by basophils. PGD2 binds to its receptors DP1 and/or DP2 and increases vascular permeability and induces bronchoconstriction. PGD2 has a rapid degradation to more stable metabolites such D-prostaglandin (PGD), F-prostaglandin (PGF), and J-prostaglandin (PGJ), which are excreted in urine [75]. Nassiri et al. [76] suggest that serum levels of 9α,11β-PGF2 could be useful biomarkers of anaphylaxis. The urinary metabolite 2,3-dinor-11β-PGF2α can be measured in urine [75].
Platelet-activating factor (PAF)
PAF is a potent phospholipid derived mediator produced by activated MC, endothelial cells, polymorphonuclear (PMN) leukocytes, eosinophils, macrophages, monocytes, and platelets. It is rapidly metabolized into its inactive metabolite lyso-PAF (by the enzyme PAF acetylhydrolase—PAF-AH). During anaphylaxis, PAF binds to its receptor on monocytes, platelets, macrophages, and neutrophil. It induces a potent bronchoconstriction and vasodilation with edema. Also is involved in platelet aggregation, activation of eosinophils, and PMN–leucocytes. Elevated levels of PAF correlate with the severity of anaphylaxis [77, 78]. Also, there is an inverse correlation between PAF and PAF-AH activity. Vadas et al. (79) showed that PAF correlates better to MC anaphylaxis compared to histamine and tryptase. PAF is common in both, IgE and IgG anaphylaxis, due to that it is released by all myeloid cell subsets. In mouse models, it had been demonstrated a synergistic effect with histamine [27]. One of the challenges of measuring PAF and PAF-AH in a daily practice is its short half-life and the transport precautions it requires.
C3a and C5a
Activation of complement by immune complexes can lead to the generation of anaphylatoxins (AT), such as C3a and C5a, which can then bind to complement receptors on MC. C3a and C5a regulate vasodilation, increase the permeability of small blood vessels, and induce contraction of smooth muscles. Basophils and MC react upon AT stimulation with release of histamine [80]. In macrophages, neutrophils, and eosinophils, C3a and C5a can trigger oxidative burst [81, 84].
Newly generated over hours
Newly generated over hours mediators are mainly growth factors, cytokines, and chemokines. Specific mediators released by MC may have autocrine, paracrine, and endocrine effects of interest to the pathogenesis of anaphylaxis. The cytokines IL-8 and GM-CSF have paracrine effects on other cells of leukocyte lineage, including eosinophils, neutrophils, and macrophages [83]. Similarly, the expression of stem cell factor by MC can have facilitatory autocrine effects on mast cell survival, chemotaxis, growth, and proliferation [84, 85]. Another paracrine loop is mast cell-derived histamine binding to endothelial H1-receptors and leading to the elaboration of NO which contributes to vasodilation [86].
Stone et al. [87] analyzed the peak concentrations of different mediators including histamine, tryptase, cytokines [IL-2, IL-4, IL-5, IL-6, IL-10, IL-13, interferon (IFN)-gamma and tumor necrosis factor (TNF)-alpha and TNFRI] and chemokines in a prospective study in patients who suffered anaphylaxis. All these mediators were higher in anaphylactic patients compared to control group. In this study, only tryptase, histamine, IL-2, IL-6, IL-10, and TNFRI correlated with hypotension, a finding similar to that found by Brown et al. [88]. Moreover, IL-6 and other inflammatory cytokines such as IL-8, TNF-a, interferon gamma (IFN-g), and IL-1b induce the inactivation of cadherin, which mediate cell adhesion, leading to vascular leakage by increased capillary permeability. It has been postulated that IL-1 released from monocytes and macrophages activates endothelial expression of NO leading to this capillary permeability. On the other hand, recently, the role of IL-6 has been described as an excellent biomarker of cytokine release reactions because of its correlation with the severity of the reactions and its longevity in a blood serum. In addition, it has been demonstrated the role of IL-6 in the systemic inflammatory response syndrome (SIRS) [89]. There are promising data regarding major basophil chemotactic factor, chemokine (C–C motif) ligand 2, or CCL-2. Concentration of CCL-2 has been found to be significantly higher in anaphylaxis compared to healthy control cohort, and potentially identified severe anaphylaxis cases [65]. Latest studies, including human and murine models, have detailed several novel mediators that may participate in anaphylaxis reaction [3••, 83, 90], as it has been summarized in Table 2.
Conclusions
Pathophysiology of anaphylaxis involves the activation of multiple pathways beyond IgE, different cell types, and a wide range of mediators. Therefore, measuring only one mediator may not be enough to diagnose anaphylaxis. This could partly explain the current lack of optimal biomarkers to confirm the diagnosis.
Given that anaphylaxis remains a clinical diagnosis, there is still exist a challenge in the laboratory diagnosis of anaphylaxis. Biomarkers are useful to reinforce the diagnosis and distinguish anaphylaxis from similar clinical scenarios. New biomarkers are pointing their interest such as chymase, carboxypeptidaseA3, PAF, PAF-AH, IL-2, IL-6, IL-10, and CCL-2, providing sample requirements, assay platform, process, and costs. However, a consensus on a reference range about sample handling and processing requirements for different biomarkers is needed.
Further studies applying these approaches are needed and they might provide greater insight into factors determining severity, clinical risk stratification, identification of mast cell disorders, and improving our understanding of this relatively complex acute condition.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Cardona V, Ansotegui IJ, Ebisawa M, El-Gamal Y, Fernandez Rivas M, Fineman S, et al. World Allergy Organization anaphylaxis guidance 2020. World Allergy Organization Journal. 2020;13(10).
Muraro A, Worm M, Alviani C, Cardona V, DunnGalvin A, Garvey LH, et al. EAACI guidelines anaphylaxis (2021 update). Allergy Eur J Allergy Clin Immunol. 2022;77(2):357–77. This paper reviews the latest European guidelines on anaphylaxis.
Nuñez-Borque E, Fernandez-Bravo S, Yuste-Montalvo A, Esteban V. Pathophysiological, cellular, and molecular events of the vascular system in anaphylaxis. Front Immunol. 2022;13(March):1–20. This study reviews the role of human vessels in anaphylaxis.
Jimenez-Rodriguez TW, Garcia-Neuer M, Alenazy LA, Castells M. Anaphylaxis in the 21st century: phenotypes, endotypes, and biomarkers. Vol. 11, Journal of Asthma and Allergy. Dove Medical Press Ltd.; 2018. 121–42.
Cianferoni A. Non–IgE-mediated anaphylaxis. J Allergy Clin Immunol [Internet]. 2021;147(4):1123–31. This paper reviews pathways that may lead to anaphylaxis in the absence of specific IgE/allergen activation.
Kajiwara N, Sasaki T, Bradding P, Cruse G, Sagara H, Ohmori K, et al. Activation of human mast cells through the platelet-activating factor receptor. J Allergy Clin Immunol. 2010;125(5):1137-1145.e6.
Finkelman FD, Khodoun MV, Strait R. Rostrum: human IgE-independent systemic anaphylaxis. J Allergy Clin Immunol. 2016;137(6):1674–80.
Schäfer B, Piliponsky AM, Oka T, Song CH, Gerard NP, Gerard C, et al. Mast cell anaphylatoxin receptor expression can enhance IgE-dependent skin inflammation in mice. J Allergy Clin Immunol. 2013;131(2):541-8.e1-9.
Khodoun M, Strait R, Orekov T, Hogan S, Karasuyama H, Herbert BR, et al. Peanuts can contribute to anaphylactic shock by activating complement. J Allergy Clin Immunol. 2009;123(2):342–51.
Kodama T, Sekine H, Takahashi M, Iwaki D, Machida T, Kanno K, et al. Role of complement in a murine model of peanut-induced anaphylaxis. Immunobiology. 2013;218(6):844–50.
Sala-Cunill A, Björkqvist J, Senter R, Guilarte M, Cardona V, Labrador M, et al. Plasma contact system activation drives anaphylaxis in severe mast cell-mediated allergic reactions. J Allergy Clin Immunol. 2015;135(4):1031-1043.e6.
Subramanian H, Gupta K, Ali H. Roles of MAS-related G protein coupled receptor-X2 (MRGPRX2) on mast cell-mediated host defense, pseudoallergic drug reactions and chronic inflammatory diseases. J Allergy Clin Immunol. 2016;138(3):700–10.
Elst J, Maurer M, Sabato V, Faber MA, Bridts CH, Mertens C, et al. Novel insights on MRGPRX2-mediated hypersensitivity to neuromuscular blocking agents and fluoroquinolones. Front Immunol. 2021;27(12):668962.
Muñoz-Cano RM, Bartra J, Picado C, Valero A. Mechanisms of anaphylaxis beyond IgE. J Investig Allergol Clin Immunol. 2016;26(2):73–82.
Kow ASF, Chik A, Soo KM, Khoo LW, Abas F, Tham CL. Identification of soluble mediators in IgG-mediated anaphylaxis via Fcγ receptor: a meta-analysis. Front Immunol. 2019;12(10):190. This paper is a meta-analysis that describes the potential soluble mediators that are involved in an IgG-mediated anaphylaxis reaction.
Reber LL, Hernandez JD, Galli SJ. The pathophysiology of anaphylaxis. J Allergy Clin Immunol. 2017;140(2):335–48.
Maurer M, Magerl M, Betschel S, Aberer W, Ansotegui IJ, Aygören-Pürsün E, et al. The international WAO/EAACI guideline for the management of hereditary angioedema—the 2021 revision and update. Allergy. 2022;77(7):1961–90.
Kabashima K, Nakashima C, Nonomura Y, Otsuka A, Cardamone C, Parente R, et al. Biomarkers for evaluation of mast cell and basophil activation. Vol. 282, Immunological Reviews. 2018. 114–20.
Elieh Ali Komi D, Wöhrl S, Bielory L. Mast cell biology at molecular level: a comprehensive review. Clin Rev Allergy Immunol. 2020;58(3):342–65.
Siracusa MC, Kim BS, Spergel JM, Artis D. Basophils and allergic inflammation. J Allergy Clin Immunol. 2013;132(4):789–801.
Stone KD, Prussin C, Metcalfe DD. IgE, mast cells, basophils, and eosinophils. J Allergy Clin Immunol. 2010;125(2):S73-80.
Parrish CP, Kim H. Food-induced anaphylaxis: an update. Curr Allergy Asthma Rep. 2018;18(8):41.
Dispenza MC, Bochner BS, MacGlashan DW. Targeting the FcεRI pathway as a potential strategy to prevent food-induced anaphylaxis. Front Immunol. 2020;11(December):1–8.
Kabashima K, Nakashima C, Nonomura Y, Otsuka A, Cardamone C, Parente R, et al. Biomarkers for evaluation of mast cell and basophil activation. Immunol Rev. 2018;282(1):114–20.
Savage JH, Courneya JP, Sterba PM, MacGlashan DW, Saini SS, Wood RA. Kinetics of mast cell, basophil, and oral food challenge responses in omalizumab-treated adults with peanut allergy. J Allergy Clin Immunol. 2012;130(5):1123-1129.e2.
Paranjape A, Tsai M, Mukai K, Hoh RA, Joshi SA, Chinthrajah RS, et al. Oral immunotherapy and basophil and mast cell reactivity in food allergy. Front Immunol. 2020;14(11):602660.
Balbino B, Sibilano R, Starkl P, Marichal T, Gaudenzio N, Karasuyama H, et al. Pathways of immediate hypothermia and leukocyte infiltration in an adjuvant-free mouse model of anaphylaxis. J Allergy Clin Immunol. 2017;139(2):584-596.e10.
Jiao D, Liu Y, Lu X, Liu B, Pan Q, Liu Y, et al. Macrophages are the dominant effector cells responsible for IgG-mediated passive systemic anaphylaxis challenged by natural protein antigen in BALB/c and C57BL/6 mice. Cell Immunol. 2014;289(1–2):97–105.
Escribese MM, Rosace D, Chivato T, Fernández TD, Corbí AL, Barber D. Alternative anaphylactic routes: the potential role of macrophages. Front Immunol. 2017;8:8.
Jönsson F, Mancardi DA, Kita Y, Karasuyama H, Iannascoli B, van Rooijen N, et al. Mouse and human neutrophils induce anaphylaxis. J Clin Investig. 2011;121(4):1484–96.
Jönsson F, Mancardi DA, Albanesi M, Bruhns P. Neutrophils in local and systemic antibody-dependent inflammatory and anaphylactic reactions. J Leukoc Biol. 2013;94(4):643–56.
Francis A, Bosio E, Stone SF, Fatovich DM, Arendts G, Nagree Y, et al. Neutrophil activation during acute human anaphylaxis: analysis of MPO and sCD62L. Clin Exp Allergy. 2017;47(3):361–70.
Jiménez-Saiz R. Drug-induced IgG-neutrophil-mediated anaphylaxis in humans: uncovered! Vol. 75, Allergy. NLM (Medline); 2020. 484–5.
Edston E. Accumulation of eosinophils, mast cells, and basophils in the spleen in anaphylactic deaths. Forensic Sci Med Pathol. 2013;9(4):496–500.
Weg VB, Watson ML, Faccioli LH, Williams TJ. Investigation of the endogenous chemoattractants involved in 111In-eosinophil accumulation in passive cutaneous anaphylactic reactions in the guinea-pig. Br J Pharmacol. 1994;113(1):35–42.
Clemetson KJ, Clemetson JM. Platelet receptors. In: Platelets. Elsevier; 2019. 169–92.
Kasperska-Zajaç A, Rogala B. Platelet function in anaphylaxis. J Investig Allergol Clin Immunol. 2006;16(1):1–4.
Beutier H, Hechler B, Godon O, Wang Y, Gillis CM, de Chaisemartin L, et al. Platelets expressing IgG receptor FcγRIIA/CD32A determine the severity of experimental anaphylaxis. Sci Immunol. 2018;3(22).
Sun HJ, Wu ZY, Nie XW, Bian JS. Role of endothelial dysfunction in cardiovascular diseases: the link between inflammation and hydrogen sulfide. Front Pharmacol. 2020;21:10.
Bucci T, Parente R, de Feo G, Cardamone C, Triggiani M. Flow-mediated dilation shows impaired endothelial function in patients with mastocytosis. J Allergy Clin Immunol. 2019;144(4):1106–11.
Libby P, Lüscher T. COVID-19 is, in the end, an endothelial disease. Eur Heart J. 2020;41(32):3038–44.
Guilarte M, Sala-Cunill A, Luengo O, Labrador-Horrillo M, Cardona V. The mast cell, contact, and coagulation system connection in anaphylaxis. Front Immunol. 2017;26:8.
Yuste-Montalvo A, Fernandez-Bravo S, Oliva T, Pastor-Vargas C, Betancor D, Goikoetxea MJ, et al. Proteomic and biological analysis of an in vitro human endothelial system in response to drug anaphylaxis. Front Immunol. 2021;25:12.
Passia E, Jandus P. Using baseline and peak serum tryptase levels to diagnose anaphylaxis: a review. Vol. 58, Clinical Reviews in Allergy and Immunology. Springer; 2020. 366–76.
Butterfield JH, Ravi A, Pongdee T. Mast cell mediators of significance in clinical practice in mastocytosis. Immunol Allergy Clin North Am. 2018;38(3):397–410.
Alcañiz L, Vega A, Chacón P, el Bekay R, Ventura I, Aroca R, et al. Histamine production by human neutrophils. FASEB J. 2013;27:2902–10.
Pucillo C, Lorentz A, Sharma M, Saluja R, Thangam EB, Jemima EA, et al. The role of histamine and histamine receptors in mast cell-mediated allergy and inflammation: the hunt for new therapeutic targets. 2018;9:1873
Takeda J, Ueda E, Takahashi J, Fukushima K. Plasma N-methylhistamine concentration as an indicator of histamine release by intravenous d-tubocurarine in humans. Anesth Analg. 1995;80(5):1015–7.
Sala-Cunill A, Guilarte M, Cardona V. Phenotypes, endotypes and biomarkers in anaphylaxis: current insights. Curr Opin Allergy Clin Immunol. 2018;18(5):370–6.
Jackson CW, Pratt CM, Rupprecht CP, Pattanaik D, Krishnaswamy G. Mastocytosis and mast cell activation disorders: clearing the air. Int J Mol Sci. 2021;22(20).
Valent P, Akin C, Metcalfe DD. Mastocytosis: 2016 updated WHO classification and novel emerging treatment concepts. Blood. 2017;129(11):1420–7.
Payne V, Kam PCA. Mast cell tryptase: a review of its physiology and clinical significance. Anaesthesia. 2004;59(7):695–703.
Mukai K, Tsai M, Saito H, Galli SJ. Mast cells as sources of cytokines, chemokines, and growth factors. Immunol Rev. 2018;282(1):121–50.
Passia E, Jandus P. Using baseline and peak serum tryptase levels to diagnose anaphylaxis: a review. Clin Rev Allergy Immunol. 2020;58(3):366–76. This paper is a revision of all the data on the usefulness of measuring tryptase during anaphylaxis and at baseline.
Valent P, Akin C, Arock M, Brockow K, Butterfield JH, Carter MC, et al. Definitions, criteria and global classification of mast cell disorders with special reference to mast cell activation syndromes: a consensus proposal. Int Arch Allergy Immunol. 2012;157(3):215–25.
Sala-Cunill A, Cardona V, Labrador-Horrillo M, Luengo O, Esteso O, Garriga T, et al. Usefulness and limitations of sequential serum tryptase for the diagnosis of anaphylaxis in 102 patients. Int Arch Allergy Immunol. 2013;160(2):192–9.
Dua S, Dowey J, Foley L, Islam S, King Y, Ewan P, et al. Diagnostic value of tryptase in food allergic reactions: a prospective study of 160 adult peanut challenges. J Allergy Clin Immunol Pract. 6(5):1692–1698.e1.
Wongkaewpothong P, Pacharn P, Sripramong C, Boonchoo S, Piboonpocanun S, Visitsunthorn N, et al. The utility of serum tryptase in the diagnosis of food-induced anaphylaxis. Allergy Asthma Immunol Res. 2014;6(4):304–9.
Zhou X, Whitworth HS, M. EK, Brown TA, Goswami R, Eren E, et al. Mast cell chymase: a useful serum marker in anaphylaxis. Journal of Allergy and Clinical Immunology. 2011 Feb;127(2):AB143–AB143.
Atiakshin D, Kostin A, Trotsenko I, Samoilova V, Buchwalow I, Tiemann M. Carboxypeptidase A3—a key component of the protease phenotype of mast cells. Cells. 2022;11(3):570.
Caughey GH, Wilson TM, Carter MC, Metcalfe DD, Raymond WW, Su S, et al. α2-Macroglobulin capture allows detection of mast cell chymase in serum and creates a circulating reservoir of angiotensin II-generating activity. J Immunol. 2009;182:5770–7.
Nishio H, Takai S, Miyazaki M, Horiuchi H, Osawa M, Uemura K, et al. Usefulness of serum mast cell-specific chymase levels for postmortem diagnosis of anaphylaxis. Int J Legal Med. 2005;119(6):331–4.
Hellman L, Akula S, Fu Z, Wernersson S. Mast cell and basophil granule proteases - in vivo targets and function. Front Immunol. 2022;13:918305.
Zhou X, Buckley MG, Lau LC, Summers C, Pumphrey RSH, Walls AF. Mast cell carboxypeptidase as a new clinical marker for anaphylaxis. J Allergy Clin Immunol. 2006;117(2):S85.
Beck SC, Wilding T, Buka RJ, Baretto RL, Huissoon AP, Krishna MT. Biomarkers in human anaphylaxis: a critical appraisal of current evidence and perspectives. Front Immunol. 2019;5(10):494.
Mochizuki A, McEuen AR, Buckley MG, Walls AF. The release of basogranulin in response to IgE-dependent and IgE-independent stimuli: validity of basogranulin measurement as an indicator of basophil activation. J Allergy Clin Immunol. 2003;112(1):102–8.
Weidmann H, Heikaus L, Long AT, Naudin C, Schlüter H, Renné T. 2017 The plasma contact system, a protease cascade at the nexus of inflammation, coagulation and immunity. Biochim Biophys Acta Mol Cell Res. 1864;1864(11 Pt B):2118–27.
Wolters PJ, Pham CT, Muilenburg DJ, Ley TJ, Caughey GH. Dipeptidyl peptidase I is essential for activation of mast cell chymases, but not tryptases, in mice. J Biol Chem. 2001;276(21):18551–6.
Caughey GH. Mast cell proteases as pharmacological targets. Eur J Pharmacol. 2016;5(778):44–55.
Cyr M, Lepage Y, Blais C, Gervais N, Cugno M, Rouleau JL, et al. Bradykinin and des-Arg(9)-bradykinin metabolic pathways and kinetics of activation of human plasma. Am J Physiol Heart Circ Physiol. 2001;281(1):H275–83.
Cicardi M, Zuraw BL. Angioedema due to bradykinin dysregulation. J Allergy Clin Immunol Pract. 2018;6(4):1132–41.
Raithel M, Zopf Y, Kimpel S, Naegel A, Molderings GJ, Buchwald F, et al. The measurement of leukotrienes in urine as diagnostic option in systemic mastocytosis. J Physiol Pharmacol. 2011;62(4):469–72.
Morel DR, Skoskiewicz M, Robinson DR, Bloch KJ, Hoaglin DC, Zapol WM. Leukotrienes, thromboxane A2, and prostaglandins during systemic anaphylaxis in sheep. Am J Physiol-Heart Circ Physiol. 1991;261(3):H782–92.
Schulman ES, Newball HH, Demers LM, Fitzpatrick FA, Adkinson NF. Anaphylactic release of thromboxane A2, prostaglandin D2, and prostacyclin from human lung parenchyma. Am Rev Respir Dis. 1981;124(4):402–6.
Idborg H, Pawelzik SC. Prostanoid metabolites as biomarkers in human disease. Metabolites. 2022;12(8):721.
Nassiri M, Eckermann O, Babina M, Edenharter G, Worm M. Serum levels of 9α,11β-PGF2 and cysteinyl leukotrienes are useful biomarkers of anaphylaxis. J Allergy Clin Immunol. 2016;137(1):312-314.e7.
Upton JEM, Hoang JA, Leon-Ponte M, Finkelstein Y, Du YJ, Adeli K, et al. Platelet-activating factor acetylhydrolase is a biomarker of severe anaphylaxis in children. Allergy. 2022 Apr 9.
Vadas P, Gold M, Perelman B, Liss GM, Lack G, Blyth T, et al. Platelet-activating factor, PAF acetylhydrolase, and severe anaphylaxis. N Engl J Med. 2008;358(1):28–35.
Vadas P, Perelman B, Liss G. Platelet-activating factor, histamine, and tryptase levels in human anaphylaxis. J Allergy Clin Immunol. 2013;131(1):144–9.
Klos A, Tenner AJ, Johswich KO, Ager RR, Reis ES, Köhl J. The role of the anaphylatoxins in health and disease. Mol Immunol. 2009;46(14):2753–66.
Murakami Y, Imamichi T, Nagasawa S. Characterization of C3a anaphylatoxin receptor on guinea-pig macrophages. Immunology. 1993;79(4):633–8.
Elsner J, Oppermann M, Czech W, Dobos G, Schöpf E, Norgauer J, et al. C3a activates reactive oxygen radical species production and intracellular calcium transients in human eosinophils. Eur J Immunol. 1994;24(3):518–22.
Nguyen SMT, Rupprecht CP, Haque A, Pattanaik D, Yusin J, Krishnaswamy G. Mechanisms governing anaphylaxis: Inflammatory cells, mediators, endothelial gap junctions and beyond. Int J Mol Sci. 2021;22(15):7785.
Zhang S, Anderson DF, Bradding P, Coward WR, Baddeley SM, MacLeod JDA, et al. Human mast cells express stem cell factor. J Pathol. 1998;186(1):59–66.
Ptaschinski C, Rasky AJ, Fonseca W, Lukacs NW. Stem cell factor neutralization protects from severe anaphylaxis in a murine model of food allergy. Front Immunol [Internet]. 2021;12:604192.
Berra-Romani R, Faris P, Pellavio G, Orgiu M, Negri S, Forcaia G, et al. Histamine induces intracellular Ca 2+ oscillations and nitric oxide release in endothelial cells from brain microvascular circulation. J Cell Physiol. 2020;235(2):1515–30.
Stone SF, Cotterell C, Isbister GK, Holdgate A, Brown SGA. Elevated serum cytokines during human anaphylaxis: identification of potential mediators of acute allergic reactions. J Allergy Clin Immunol. 2009;124(4):786-792.e4.
Brown SGA, Stone SF, Fatovich DM, Burrows SA, Holdgate A, Celenza A, et al. Anaphylaxis: clinical patterns, mediator release, and severity. J Allergy Clin Immunol. 2013;132(5):1141-1149.e5.
Kang S, Tanaka T, Inoue H, Ono C, Hashimoto S, Kioi Y, et al. IL-6 trans-signaling induces plasminogen activator inhibitor-1 from vascular endothelial cells in cytokine release syndrome. Proc Natl Acad Sci. 2020;117(36):22351–6.
Lopez-Castejon G, Brough D. Understanding the mechanism of IL-1β secretion. Cytokine Growth Factor Rev. 2011;22(4):189–95.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human participants or animal performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Anaphylaxis.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Galvan-Blasco, P., Gil-Serrano, J. & Sala-Cunill, A. New Biomarkers in Anaphylaxis (Beyond Tryptase). Curr Treat Options Allergy 9, 303–322 (2022). https://doi.org/10.1007/s40521-022-00326-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40521-022-00326-1