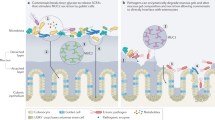
Abstract
Recent observations indicate that acute enteric infections may contribute to various intestinal and extra-intestinal disorders long after elimination of the inciting microorganism. The mechanisms remain unclear. This review discusses how alterations to the gut microbiota by enteropathogens during the acute stage of an infection may at least in part contribute to these presentations. After providing a critical discussion of the biology of the human intestinal microbiota, the review presents recent data that illustrate how enteropathogens may activate latent virulence genes in commensal bacteria, disrupt the microbiota biofilm phenotype, and promote the release of pathobionts from the commensal biofilm. Evidence suggests that, in turn, these planktonic pathobionts may spontaneously translocate across the mucus and the gut epithelium to trigger, at least in part, the pro-inflammatory events that lead to these long-term, post-infectious sequelae.
Similar content being viewed by others
References
O’Ryan M, Prado V, Pickering LK. A millennium update on pediatric diarrheal illness in the developing world. Semin Pediatr Infect Dis. 2005;16:125–36.
Riddle MS, Sanders JW, Putnam SD, et al. Incidence, etiology, and impact of diarrhea among long-term travelers (US military and similar populations): a systematic review. Am J Trop Med Hyg. 2006;74:891–900.
Letizia A, Riddle MS, Tribble D, et al. Effects of pre-deployment loperamide provision on use and travelers’ diarrhea outcomes among U.S. military personnel deployed to Turkey. Travel Med Infect Dis. 2014;12:360–3.
Pavli A, Silvestros C, Patrinos S, et al. Pre-travel preparation practices among business travellers to tropical and subtropical destinations: results from the Athens International Airport Survey. Travel Med Infect Dis. 2014;12:364–9.
Grady D. Gastrointestinal infection deaths more than doubled. New York Times; 2012. Available at: http://www.nytimes.com/2012/03/15/health/gastrointestinal-infection-deaths-more-than-doubled.html?_r=0. Accessed 8 Aug 2015.
Buret AG, Reti K. Acute enteric infections alter commensal microbiota: new mechanisms in post-infectious intestinal inflammatory disorders. In: Heidt PJ, Lang D, Riddle MS, et al, editors. Persisting Consequences of Intestinal Infections. Old Herborn University Monograph, OHUS; 2014. p. 87–106. ISBN: 3-923022-39-5.
Scallan E, Hoekstra RM, Angulo FJ, et al. Foodborne illness acquired in the United States—major pathogens. Emerg Infect Dis. 2011;17:7–15.
Liu J, Kabir F, Manneh J, et al. Development and assessment of molecular diagnostic tests for 15 enteropathogens causing childhood diarrhoea: a multicentre study. Lancet Infect Dis. 2014;14:716–24.
Payne DC, Vinjé J, Szilagyi PG, et al. Norovirus and medically attended gastroenteritis in U.S. children. N Engl J Med. 2013;368:1121–30.
Bartelt LA, Sartor RB. Advances in understanding Giardia: determinants and mechanisms of chronic sequelae. F1000Prime Rep. 2015;7:1–14.
Rees JH, Soudain SE, Gregson NA, et al. Campylobacter jejuni infection and Guillain-Barré syndrome. N Engl J Med. 1995;333:1374–9.
Thabane M, Kottachchi DT, Marshall JK. Systematic review and meta-analysis: the incidence and prognosis of post-infectious irritable bowel syndrome. Aliment Pharmacol Ther. 2007;26:535–44.
Gradel KO, Nielsen HL, Schønheyder HC, et al. Increased short- and long-term risk of inflammatory bowel disease after Salmonella or Campylobacter gastroenteritis. Gastroenterology. 2009;137:495–501.
García Rodríguez LA, Ruigómez A, Panés J. Acute gastroenteritis is followed by an increased risk of inflammatory bowel disease. Gastroenterology. 2006;130:1588–94.
Halliez MCM, Buret AG. Extra-intestinal and long term consequences of Giardia duodenalis infections. World J Gastroenterol. 2013;19:8974–85.
Network Investigators MAL-ED. The MAL-ED study: a multinational and multidisciplinary approach to understand the relationship between enteric pathogens, malnutrition, gut physiology, physical growth, cognitive development, and immune responses in infants and children up to 2 years of age in resource-poor environments. Clin Infect Dis. 2014;59:S193–206.
Buret A, Amat C, Manko A, et al. Giardia duodenalis: new research developments in pathophysiology, pathogenesis, and virulence factors. Curr Trop Med Rep. 2015;2:110–8.
Savioli L, Smith H, Thompson A. Giardia and Cryptosporidium join the ‘neglected diseases initiative’. Trends Parasitol. 2006;22:203–8.
World Health Organization. Campylobacter. Fact sheet N°255. 2011. Available at: http://www.who.int/mediacentre/factsheets/fs255/en/. Accessed 7 Aug 2015.
Connor BA, Schwartz E. Typhoid and paratyphoid fever in travellers. Lancet Infect Dis. 2005;5:623–8.
Evering T, Weiss LM. The immunology of parasite infections in immunocompromised hosts. Parasite Immunol. 2006;28:549–65.
Solaymani-Mohammadi S, Singer SM. Giardia duodenalis: the double-edged sword of immune responses in giardiasis. Exp Parasitol. 2010;126:292–7.
Cotton JA, Beatty JK, Buret AG. Host parasite interactions and pathophysiology in Giardia infections. Int J Parasitol. 2011;41:925–33.
von Allmen N, Christen S, Forster U, et al. Acute trichinellosis increases susceptibility to Giardia lamblia infection in the mouse model. Parasitology. 2006;133:139–49.
Ankarklev J, Hestvik E, Lebbad M, et al. Common coinfections of Giardia intestinalis and Helicobacter pylori in non-symptomatic Ugandan children. PLoS Negl Trop Dis. 2012;6:e1780.
Bhavnani D, Goldstick JE, Cevallos W, et al. Synergistic effects between rotavirus and coinfecting pathogens on diarrheal disease: evidence from a community-based study in northwestern Ecuador. Am J Epidemiol. 2012;176:387–95.
Mahmoudi MR, Kazemi B, Mohammadiha A, et al. Detection of Cryptosporidium and Giardia (oo)cysts by IFA, PCR and LAMP in surface water from Rasht, Iran. Trans R Soc Trop Med Hyg. 2013;107:511–7.
Mohamed AS, Levine M, Camp Jr JW, et al. Temporal patterns of human and canine Giardia infection in the United States: 2003–2009. Prev Vet Med. 2014;113:249–56.
Mukherjee AK, Chowdhury P, Rajendran K, et al. Association between Giardia duodenalis and coinfection with other diarrhea-causing pathogens in India. Biomed Res Int. 2014;2014:786480.
Moore SR. Early childhood diarrhea and helminthiases associated with long-term linear growth faltering. Int J Epidemiol. 2001;30:1457–64.
Jensen LA, Marlin JW, Dyck DD, et al. Prevalence of multi-gastrointestinal infections with helminth, protozoan and Campylobacter spp. in Guatemalan children. J Infect Dev Ctries. 2009;3:229–34.
Veenemans J, Schouten LRA, Ottenhof MJ, et al. Effect of preventive supplementation with zinc and other micronutrients on non-malarial morbidity in Tanzanian pre-school children: a randomized trial. PLoS One. 2012;7:e41630.
Cotton J, Motta JP, Schenk LP, et al. Giardia duodenalis infection reduces granulocyte infiltration in an in vivo model of bacterial toxin-induced colitis and attenuates inflammation in human intestinal tissue. PLoS One. 2014;9:e109087.
Maizels RM, Yazdanbakhsh M. Immune regulation by helminth parasites: cellular and molecular mechanisms. Nat Rev Immunol. 2003;3:733–44.
Chen CC, Louie S, Shi HN. Concurrent infection with an intestinal helminth parasite impairs host resistance to enteric Citrobacter rodentium and enhances Citrobacter-induced colitis in mice. Infect Immun. 2005;73:5468–81.
Osborne LC, Monticelli LA, Nice TJ, et al. Virus-helminth coinfection reveals a microbiota-independent mechanism of immunomodulation. Science. 2014;345:578–82.
Cotton JA, Bhargava A, Ferraz JG, et al. Giardia duodenalis cathepsin B proteases degrade intestinal epithelial interleukin-8 and attenuate interleukin-8-induced neutrophil chemotaxis. Infect Immun. 2014;82:2772–87.
Kamada N, Chen GY, Inohara N, et al. Control of pathogens and pathobionts by the gut microbiota. Nat Immunol. 2013;14:685–90.
Faith JJ, Guruge JL, Charbonneau M, et al. The long-term stability of the human gut microbiota. Science. 2013;341:1237439.
Dominianni C, Sinha R, Goedert JJ, et al. Sex, body mass index, and dietary fiber intake influence the human gut microbiome. PLoS One. 2015;10(4):e0124599.
Thompson JA, Oliveira RA, Djukovic A, et al. Manipulation of the quorum sensing signal AI-2 affects the antibiotic-treated gut microbiota. Cell Rep. 2015;10:1861–71.
Franzosa EA, Hunag K, Meadow JF, et al. Identifying personal microbiomes using metagenomic codes. Proc Natl Acad Sci U S A. 2015;112:E2930–8.
Magnusson KR, Hauck L, Jeffrey BM, et al. Relationship between diet-related changes in the gut microbiome and cognitive flexibility. Neuroscience. 2015;300:128–40.
Nobel YR, Cox LM, Kirigin FK, et al. Metabolic and metagenomic outcomes from early-life pulsed antibiotic treatment. Nat Commun. 2015;6:7486.
Collins SM, Surette M, Bercik P. The interplay between the intestinal microbiota and the brain. Nat Rev Microbiol. 2012;10:735–42.
Carding S, Verbeke K, Vipond DT, et al. Dysbiosis of the gut microbiota in disease. Microb Ecol Health Dis. 2015;26:26191.
Gao Z, Guo B, Gao R, et al. Microbiota dysbiosis is associated with colorectal cancer. Front Microbiol. 2015;6:20.
Ijssennagger N, Belzer C, Hooiveld GJ, et al. Gut microbiota facilitates dietary heme-induced epithelial hyperproliferation by opening the mucus barrier in colon. Proc Natl Acad Sci U S A. 2015;112:10038–43.
De Weerth C, Funetes S, Puylaert P, et al. Intestinal microbiota of infants with colic: development and specific signatures. Pediatrics. 2013;131:e550–8.
Lepage P, LeClerc MC, Joossens M, et al. Metagenomic insight into our gut’s microbiome. Gut. 2013;62:146–58.
de Vos WM. Microbial biofilms and the human intestinal microbiome. NPJ Biofilms Microbiomes. 2015;1:15005.
Motta JP, Flannigan KL, Agbor TA, et al. Hydrogen sulfide protects from colitis and restores intestinal microbiota biofilm and mucus production. Inflamm Bowel Dis. 2015;21:1006–17.
Hity M, Burke C, Pedro H, et al. Disordered microbial communities in asthmatic airways. PLoS One. 2010;5:e8578.
Scher JU, Abramson SB. The microbiome and rheumatoid arthritis. Nat Rev Rheumatol. 2011;7:569–78.
Kelly CR, Kahn S, Kashyap P, et al. Update on fecal microbiota transplantation 2015: indications, methodologies, mechanisms, and outlook. Gastroenterology. 2015;149:223–37.
Zoetendal EG, Rajilic-Stojanovic M, de Vos WM. High-throughput diversity and functionality analysis of the gastrointestinal tract microbiota. Gut. 2008;57:1605–15.
Dethlefsen L, Relman DA. Incomplete recovery and individualized responses of the human distal gut microbiota to repeated antibiotic perturbation. Proc Natl Acad Sci U S A. 2011;108 Suppl 1:4554–61.
Forsythe P, Kunze WA. Voices from within: gut microbes and the CNS. Cell Mol Life Sci. 2013;70:55–69.
Hopkins MJ, Sharp R, Macfarlane GT. Variation in human intestinal microbiota with age. Dig Liver Dis. 2002;34 Suppl 2:S12–8.
Tap J, Mondot S, Levenez F, et al. Towards the human intestinal phylogenetic core. Environ Microbiol. 2009;11:2574–84.
Frank DN, St Amand AL, Feldman RA, et al. Molecular-phylogenetic characterization of microbial community imbalances in human inflammatory bowel diseases. Proc Natl Acad Sci U S A. 2007;104:13780–5.
Macfarlane GT, Gibson GR, Cumming JH. Comparison of fermentation reactions in different regions of the human colon. J Appl Bacteriol. 1992;72:57–64.
Macfarlane S, Macfarlane G. Bacterial colonization of surfaces in the large intestine. In: Gibson G, Roberfroid M, editors. Colonic Microflora, Nutrition and Health. London: Chapman and Hall; 1999. p. 71–87.
Ganesh BP, Klopfleisch R, Loh G, et al. Commensal Akkermansia muciniphila exacerbates gut inflammation in Salmonella typhimurium-infected gnotobiotic mice. PLoS One. 2013;8:e74963.
Jakobsson HE, Rodríguez-Piñeiro AM, Schütte A, et al. The composition of the gut microbiota shapes the colon mucus barrier. EMBO Rep. 2015;16:164–77.
Swidsinsky A, Loering-Bauke V, Herber A. Mucosal flora in Crohn’s disease and ulcerative colitis – an overview. J Physiol Pharmacol. 2009;60 Suppl 6:61–71.
Darfeuille-Michaud A, Neut C, Barnich N, et al. Presence of adherent Escherichia coli strains in ileal mucosa of patients with Crohn’s disease. Gastroenterology. 1998;115:1405–13.
Chassaing B, Koren O, Goodrich JK, et al. Dietary emulsifiers impact the mouse gut microbiota promoting colitis and metabolic syndrome. Nature. 2015;519:92–6.
De Filippo C, Cavalieri D, Di Paola M, et al. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc Natl Acad Sci U S A. 2010;107:14691–6.
Xavier RJ, Podolsky DK. Unravelling the pathogenesis of inflammatory bowel disease. Nature. 2007;448:427–34.
Kalischuk LD, Buret AG. A role for Campylobacter jejuni-induced enteritis in inflammatory bowel disease? Am J Physiol Gastrointest Liver Physiol. 2009;298:G1–9.
Chassaing B, Darfeuille-Michaud A. The commensal microbiota and enteropathogens in the pathogenesis of inflammatory bowel diseases. Gastroenterology. 2011;140:1720–8.
Sokol H, Pigneur B, Watterlot L, et al. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proc Natl Acad Sci U S A. 2008;105:16731–6.
Quévrain E, Maubert MA, Michon C, et al. Identification of an anti-inflammatory protein from Faecalibacterium prausnitzii, a commensal bacterium deficient in Crohn’s disease. Gut. 2015. doi:10.1136/gutjnl-2014-307649 [Epub ahead of print].
Nelson AM, Walk ST, Taube S, et al. Disruption of the human gut microbiota following norovirus infection. PLoS One. 2012;7:e48224.
Lupp C, Robertson ML, Wickham ME, et al. Host-mediated inflammation disrupts the intestinal microbiota and promotes the overgrowth of Enterobacteriaceae. Cell Host Microbe. 2007;2:119–29.
Stecher B, Robbiani R, Walker AW, et al. Salmonella enterica serovar typhimurium exploits inflammation to compete with the intestinal microbiota. PLoS Biol. 2007;5:2177–89.
Abraham C, Medzhitov R. Interactions between the host innate immune system and microbes in inflammatory bowel disease. Gastroenterology. 2011;140:1729–37.
O’Hara JR, Feener TD, Fischer CD, et al. Campylobacter jejuni disrupts protective Toll-like receptor 9 signaling in colonic epithelial cells and increases the severity of dextran sulfate sodium-induced colitis in mice. Infect Immun. 2012;80:1563–71.
McCormick BA, Nusrat A, Parkos CA, et al. Unmasking of intestinal epithelial lateral membrane beta1 integrin consequent to transepithelial neutrophil migration in vitro facilitates inv-mediated invasion by Yersinia pseudotuberculosis. Infect Immun. 1997;65:1414–21.
Medzhitov R. Toll-like receptors and innate immunity. Nat Rev Immunol. 2001;1:135–45.
Muza-Moons MM, Koutsouris A, Hecht G. Disruption of cell polarity by enteropathogenic Escherichia coli enables basolateral membrane proteins to migrate apically and to potentiate physiological consequences. Infect Immun. 2003;71:7069–78.
O’Hara JR, Buret AG. Mechanisms of intestinal tight junctional disruption during infection. Front Biosci. 2008;13:7008–21.
Kalischuk LD, Inglis GD, Buret AG. Strain-dependent induction of epithelial cell oncosis by Campylobacter jejuni is correlated with invasion ability and is independent of cytolethal distending toxin. Microbiology. 2007;153:2952–63.
Lamb-Rosteski JM, Kalischuk LD, Inglis GD, et al. Epidermal growth factor inhibits Campylobacter jejuni-induced claudin-4 disruption, loss of epithelial barrier function, and Escherichia coli translocation. Infect Immun. 2008;76:3390–8.
Kalischuk LD, Inglis GD, Buret AG. Campylobacter jejuni induces transcellular translocation of commensal bacteria via lipid rafts. Gut Pathog. 2009;1:2.
Kalischuk LD, Leggett F, Inglis GD. Campylobacter jejuni induces transcytosis of commensal bacteria across the intestinal epithelium through M-like cells. Gut Pathog. 2010;2:14.
Knoop KA, McDonald KG, Kulkarni DH, et al. Antibiotics promote inflammation through the translocation of native commensal colonic bacteria. Gut. 2015. doi:10.1136/gutjnl-2014-309059 [Epub ahead of print].
Nazli A, Yang PC, Jury J, et al. Epithelia under metabolic stress perceive commensal bacteria as a threat. Am J Pathol. 2004;164:947–57.
Clark E, Hoare C, Tanianis-Hughes J, et al. Interferon gamma induces translocation of commensal Escherichia coli across gut epithelial cells via a lipid raft-mediated process. Gastroenterology. 2005;128:1258–67.
Söderholm JD, Streutker C, Yang PC, et al. Increased epithelial uptake of protein antigens in the ileum of Crohn’s disease mediated by tumour necrosis factor alpha. Gut. 2004;53:1817–24.
Swidsinski A, Weber J, Loening-Baucke V, et al. Spatial organization and composition of the mucosal flora in patients with inflammatory bowel disease. J Clin Microbiol. 2005;43:3380–9.
Kamada N, Kim YG, Sham HP, et al. Regulated virulence controls the ability of a pathogen to compete with the gut microbiota. Science. 2012;336:1325–9.
Chen TL, Chen S, Wu HW, et al. Persistent gut barrier damage and commensal bacterial influx following eradication of Giardia infection in mice. Gut Pathog. 2013;5:26.
Halliez M, Motta JP, Feener T, et al. Giardia duodenalis induces bacterial translocation and causes post-infectious visceral hypersensitity. Am J Physiol. (Gastrointestinal and Liver Physiol.) 2016;310(8):G574–85.
Gerbaba TK, Gupta P, Rioux K, et al. Giardia duodenalis-induced alterations of commensal bacteria kill Caenorhabditis elegans: a new model to study microbial-microbial interactions in the gut. Am J Physiol Gastrointest Liver Physiol. 2015;308:G550–61.
Pham TA, Clare S, Goulding D, et al. Epithelial IL-22RA1-mediated fucosylation promotes intestinal colonization resistance to an opportunistic pathogen. Cell Host Microbe. 2014;16:504–16.
Goto Y, Obata T, Kunisawa J, et al. Innate lymphoid cells regulate intestinal epithelial cell glycosylation. Science. 2014;345:1254009.
Pickard JM, Maurice CF, Kinnebrew MA, et al. Rapid fucosylation of intestinal epithelium sustains host-commensal symbiosis in sickness. Nature. 2014;514:638–41.
DuPont AW, DuPont HL. The intestinal microbiota and chronic disorders of the gut. Nat Rev Gastoenterol Hepatol. 2011;8:523–31.
Probert HM, Gibson GR. Bacterial biofilms in the human gastrointestinal tract. Curr Issues Intest Microbiol. 2002;3:23–7.
Pruteanu M, Hyland NP, Clarke DJ, et al. Degradation of the extracellular matrix components by bacterial-derived metalloproteases: implications for inflammatory bowel diseases. Inflamm Bowel Dis. 2010;17:1189–200.
Spiller R, Campbell E. Post-infectious irritable bowel syndrome. Curr Opin Gastroenterol. 2006;22:13–7.
Hanevik K, Dizdar V, Langeland N, et al. Development of functional gastrointestinal disorders after Giardia lamblia infection. BMC Gastroenterol. 2009;9:27.
Marshall JK, Thabane M, Garg AX, et al. Eight year prognosis of postinfectious irritable bowel syndrome following waterborne bacterial dysentery. Gut. 2010;59:605–11.
Wensaas KA, Langeland N, Hanevik K, et al. Irritable bowel syndrome and chronic fatigue 3 years after acute giardiasis: historic cohort study. Gut. 2012;61:214–9.
Cremon C, Stanghellini V, Pallotti F, et al. Salmonella gastroenteritis during childhood is a risk factor for irritable bowel syndrome in adulthood. Gastroenterology. 2014;147:69–103.
Ceri H, Olson ME, Stremick C, et al. The Calgary Biofilm Device: a new technology for the rapid determination of antibiotic susceptibility of bacterial biofilms. J Clin Microbiol. 1999;37:1771–6.
Sproule-Willoughby KM, Stanton MM, Rioux KP, et al. In vitro anaerobic biofilms of human colonic microbiota. J Microbiol Methods. 2010;83:296–301.
Beatty J, Akierman S, Rioux K, et al. Gut microbiota biofilm disruptions by Giardia: pathology in human enterocytes and germ-free mice. FASEB J 2013;27 [abstract 131.1].
Buret AG, Akierman S, Feener T, et al. Campylobacter jejuni or Giardia duodenalis-mediated disruptions of human intestinal microbiota biofilms: novel mechanisms producing post-infectious intestinal inflammatory disorders. Gastroenterology. 2013;144:S–309 [abstract Sa1802].
Reti KL, Tymensen LD, Davis Sp, et al. Campylobacter jejuni increases flagellar expression and adhesion of non-invasive Escherichia coli: effects on enterocytic TLR4 and CXCL8 expression. Infect Immun. 2015;83(12):4571–4581.
Acknowledgments
Critical proofreading by Drs. Jean-Paul Motta and James Cotton is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Some of the findings discussed in this article have been generated through the financial support of the Natural Sciences and Engineering Research Council of Canada, Crohn’s and Colitis Canada, Alberta Innovates Health Solution, and the Canadian Institutes of Health Research. Lupin Pharmaceuticals, Inc., Baltimore, MD, facilitated this article by providing financial support.
Andre G. Buret declares no other conflict of interest.
Human and Animal Rights and Informed Consent
This article may contain studies with human or animal subjects performed by the author. The author verifies that all current Ethical Standards for the conductance of prospective research were followed.
Additional information
This article is part of the Topical Collection on Protozoa
Rights and permissions
About this article
Cite this article
Buret, A.G. Enteropathogen-Induced Microbiota Biofilm Disruptions and Post-Infectious Intestinal Inflammatory Disorders. Curr Trop Med Rep 3, 94–101 (2016). https://doi.org/10.1007/s40475-016-0079-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40475-016-0079-x