Abstract
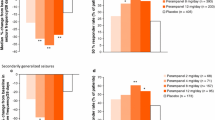
Perampanel (Fycompa®), a novel selective, noncompetitive antagonist of α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid-type glutamate receptors on post-synaptic neurons, is indicated as adjunctive therapy for patients aged ≥12 years with partial-onset seizures with or without secondarily generalized seizures. In this patient population, adjunctive once-daily perampanel is more effective than adjunctive placebo in reducing seizure frequency and is generally well-tolerated.

Similar content being viewed by others
References
Neligan A, Hauser WA, Sander JW. The epidemiology of the epilepsies. Handb Clin Neurol. 2012;107(3rd series):113–33.
Figueroa Garcia A. Partial epilepsies. 2012. http://emedicine.medscape.com/article/1186635-overview. Accessed 27 Feb 2013.
Berg AT, Berkovic SF, Brodie MJ, et al. Revised terminology and concepts for organization of seizures and epilepsies: report of the ILAE Commission on Classification and Terminology, 2005–2009. Epilepsia. 2010;51(4):676–85.
The National Society for Epilepsy. Epilepsy: an introduction to epileptic seizures. 2012. http://www.epilepsysociety.org.uk/AboutEpilepsy/Whatisepilepsy/Seizures. Accessed 27 Feb 2013.
Plosker GL. Perampanel as adjunctive therapy in patients with partial-onset seizures. CNS Drugs. 2012;26(12):1085–96.
Fycompa (perampanel) film-coated tablet: summary of product characteristics. London: European Medicines Agency; 2012.
Fycompa (perampanel) tablets: US prescribing information. Woodcliff Lake (NJ): Eisai Inc.; 2012.
French JA, Krauss GL, Biton V, et al. Adjunctive perampanel for refractory partial-onset seizures: randomized phase III study 304. Neurology. 2012;79(6):589–96.
French JA, Krauss GL, Steinhoff BJ, et al. Evaluation of adjunctive perampanel in patients with refractory partial-onset seizures: results of randomized global phase III study 305. Epilepsia. 2013;54(1):117–25.
Krauss GL, Serratosa JM, Villanueva V, et al. Randomized phase III study 306: adjunctive perampanel for refractory partial-onset seizures. Neurology. 2012;78(18):1408–15.
Steinhoff BJ, Ben-Menachem E, Ryvlin P, et al. Efficacy and safety of adjunctive perampanel for the treatment of refractory partial seizures: a pooled analysis of three phase III studies. Epilepsia. Epub 2013 May 10.
Krauss GL, Perucca E, Ben-Menachem E, et al. Perampanel, a selective, noncompetitive α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor antagonist, as adjunctive therapy for refractory partial-onset seizures: interim results from phase III, extension study 307. Epilepsia. 2013;54(1):126–34.
Acknowledgement
The manuscript was reviewed by: A. Husain, Duke University Medical Center, Medicine (Neurology), Durham, NC, USA.
Disclosure
This article was updated from CNS Drugs 2012;26(12):1085–96 [5]. The preparation of these reviews was not supported by any external funding. During the review process, the manufacturer of the agent under review was offered an opportunity to comment on the articles. Changes resulting from comments received were made by the authors on the basis of scientific and editorial merit.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Scott, L.J., Lyseng-Williamson, K.A. & Plosker, G.L. Perampanel: a guide to its use in partial-onset seizures. Drugs Ther Perspect 29, 189–193 (2013). https://doi.org/10.1007/s40267-013-0050-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40267-013-0050-z