Abstract
Background
Hallucinations are associated with earlier death in older adults with dementia, but antipsychotic medications are also associated with mortality, and comparisons of their relative harms are lacking.
Objective
To determine the individual and combined association between hallucinations, antipsychotic use, and mortality.
Methods
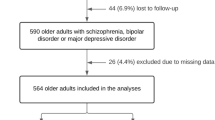
We performed a retrospective cohort study using Medicare-linked survey data from two nationally representative studies (the National Health and Aging Trends Study and the Health and Retirement Study) containing validated dementia identification algorithms and a screening question for hallucinations. Using Medicare claims, we identified participants with dementia who had no history of antipsychotic use during the year of or prior to entry. We used extended Cox regression with time-varying covariates to analyze the association between hallucinations, antipsychotic use, and mortality adjusting for confounders.
Results
We identified 1703 eligible subjects who contributed 4,819 person-years of follow-up. 555 (32.6%) had hallucinations at baseline, 705 (41.4%) reported hallucinations at least once during follow-up, and 284 (16.7%) received antipsychotics. Hallucinations were associated with an increased risk of death in unadjusted models (hazard ratio (HR) 1.36; 95% confidence interval (CI): 1.18–1.5), but antipsychotic use was not (HR 1.03; 95% CI 0.85–1.2). After adjusting for age, race, gender, dementia severity, and comorbidities, the HR for hallucinations attenuated and was no longer statistically significant (1.15, 95% CI 0.98–1.34). There was no significant interaction between hallucinations and antipsychotic use.
Conclusion
Hallucinations are associated with an increased risk of death that is greater than the risk associated with antipsychotic use, though this is partially confounded by dementia severity and comorbidities.


Similar content being viewed by others
References
Ropacki SA, Jeste DV. Epidemiology of and risk factors for psychosis of Alzheimer’s disease: a review of 55 studies published from 1990 to 2003. Am J Psychiatry. 2005;162:2022–30.
Zhu K, van Hilten JJ, Putter H, Marinus J. Risk factors for hallucinations in Parkinson’s disease: results from a large prospective cohort study. Mov Disord Off J Mov Disord Soc. 2013;28:755–62.
Savica R, Grossardt BR, Bower JH, Boeve BF, Ahlskog JE, Rocca WA. Incidence of dementia with Lewy bodies and Parkinson disease dementia. JAMA Neurol. 2013;70:1396–402.
Schneider LS, Dagerman KS, Insel P. Risk of death with atypical antipsychotic drug treatment for dementia: meta-analysis of randomized placebo-controlled trials. JAMA. 2005;294:1934–43.
Dorsey ER, Rabbani A, Gallagher SA, Conti RM, Alexander GC. Impact of FDA black box advisory on antipsychotic medication use. Arch Intern Med. 2010;170:96–103.
Menon GJ. Complex visual hallucinations in the visually impaired: a structured history-taking approach. Arch Ophthalmol Chic Ill. 1960;2005(123):349–55.
Chaudhuri KR, Prieto-Jurcynska C, Naidu Y, Mitra T, Frades-Payo B, Tluk S, et al. The nondeclaration of nonmotor symptoms of Parkinson’s disease to health care professionals: an international study using the nonmotor symptoms questionnaire. Mov Disord Off J Mov Disord Soc. 2010;25:704–9.
Simoni-Wastila L, Ryder PT, Qian J, Zuckerman IH, Shaffer T, Zhao L. Association of antipsychotic use with hospital events and mortality among medicare beneficiaries residing in long-term care facilities. Am J Geriatr Psychiatry Off J Am Assoc Geriatr Psychiatry. 2009;17:417–27.
Maust DT, Kim HM, Seyfried LS, Chiang C, Kavanagh J, Schneider LS, et al. Antipsychotics, other psychotropics, and the risk of death in patients with dementia: number needed to harm. JAMA Psychiat. 2015;72:438–45.
Rashid N, Wetmore JB, Irfan M, Abler V. Adverse outcomes associated with off-label agents used to treat dementia patients with psychosis: a case-control medicare database study. Am J Alzheimers Dis Other Demen. 2022;37:15333175221081374.
Kales HC, Valenstein M, Kim HM, McCarthy JF, Ganoczy D, Cunningham F, et al. Mortality risk in patients with dementia treated with antipsychotics versus other psychiatric medications. Am J Psychiatry. 2007;164:1568–76 (quiz 1623).
Park Y, Franklin JM, Schneeweiss S, Levin R, Crystal S, Gerhard T, et al. Antipsychotics and mortality: adjusting for mortality risk scores to address confounding by terminal illness. J Am Geriatr Soc. 2015;63:516–23.
Hamedani AG, Weintraub D, Willis AW. Medicare claims data underestimate hallucinations in older adults with dementia. Am J Geriatr Psychiatry Off J Am Assoc Geriatr Psychiatry. 2022;30:352–9.
Holiday KA, Pirogovsky-Turk E, Malcarne VL, Filoteo JV, Litvan I, Lessig SL, et al. Psychometric properties and characteristics of the north-east visual hallucinations interview in Parkinson’s disease. Mov Disord Clin Pract. 2017;4:717–23.
Niazi S, Krogh Nielsen M, Singh A, Sørensen TL, Subhi Y. Prevalence of Charles Bonnet syndrome in patients with age-related macular degeneration: systematic review and meta-analysis. Acta Ophthalmol (Copenh). 2020;98:121–31.
Vilalta-Franch J, López-Pousa S, Calvó-Perxas L, Garre-Olmo J. Psychosis of Alzheimer disease: prevalence, incidence, persistence, risk factors, and mortality. Am J Geriatr Psychiatry Off J Am Assoc Geriatr Psychiatry. 2013;21:1135–43.
Scarmeas N, Brandt J, Albert M, Hadjigeorgiou G, Papadimitriou A, Dubois B, et al. Delusions and hallucinations are associated with worse outcome in Alzheimer disease. Arch Neurol. 2005;62:1601–8.
Gardette V, Lapeyre-Mestre M, Coley N, Cantet C, Montastruc J-L, Vellas B, et al. Antipsychotic use and mortality risk in community-dwelling Alzheimer’s disease patients: evidence for a role of dementia severity. Curr Alzheimer Res. 2012;9:1106–16.
Wilson RS, Tang Y, Aggarwal NT, Gilley DW, McCann JJ, Bienias JL, et al. Hallucinations, cognitive decline, and death in Alzheimer’s disease. Neuroepidemiology. 2006;26:68–75.
Connors MH, Ames D, Boundy K, Clarnette R, Kurrle S, Mander A, et al. Predictors of mortality in dementia: the PRIME Study. J Alzheimers Dis JAD. 2016;52:967–74.
Lopez OL, Wisniewski SR, Becker JT, Boller F, DeKosky ST. Psychiatric medication and abnormal behavior as predictors of progression in probable Alzheimer disease. Arch Neurol. 1999;56:1266–72.
Mooldijk SS, Licher S, Wolters FJ. characterizing demographic, racial, and geographic diversity in dementia research: a systematic review. JAMA Neurol. 2021;78:1255–61.
NHATS Public Use Data. Rounds 1-7. Sponsored by the National Institute on Aging (grant number NIA U01AG032947) through a cooperative agreement with the Johns Hopkins Bloomberg School of Public Health. www.nhats.org. Accessed 14 Mar 2019.
Harnonized HRS Dataset and Codebook Version B, October 2018. Developed by the Gateway to Global Aging Data, funded by the National Institute on Aging (R01 AG030153 RC2 AG036619, 1R03Ag043052). www.g2aging.org. Accessed 14 Mar 2019.
Kasper J, Freedman V, Spillman B. Classification of Persons by Dementia Status in the National Health and Aging Trends Study. Technical Pper #5. Baltim Johns Hopkins Bloom Sch Public Health. 2013;
Langa K, Weir D, Kabeto M, Sonnega A. Langa-Weir Classification of Cognitive Function (1995 Onward). Survey Research Center, Institute for Social Research, University of Michigan; 2020.
Harrison KL, Ritchie CS, Patel K, Hunt LJ, Covinsky KE, Yaffe K, et al. Care settings and clinical characteristics of older adults with moderately severe dementia. J Am Geriatr Soc. 2019;67:1907–12.
Lee SJ, Lindquist K, Segal MR, Covinsky KE. Development and validation of a prognostic index for 4-year mortality in older adults. JAMA. 2006;295:801–8.
Kales HC, Gitlin LN, Lyketsos CG, Detroit Expert Panel on Assessment and Management of Neuropsychiatric Symptoms of Dementia. Management of neuropsychiatric symptoms of dementia in clinical settings: recommendations from a multidisciplinary expert panel. J Am Geriatr Soc. 2014;62:762–9.
Sweet RA, Bennett DA, Graff-Radford NR, Mayeux R, National Institute on Aging Late-Onset Alzheimer’s Disease Family Study Group. Assessment and familial aggregation of psychosis in Alzheimer’s disease from the National Institute on Aging Late Onset Alzheimer’s Disease Family Study. Brain J Neurol. 2010;133:1155–62.
Acknowledgements
The National Health and Aging Trends Study (NHATS) is sponsored by the National Institute on Aging (grant number NIA U01AG032947) through a cooperative agreement with the Johns Hopkins Bloomberg School of Public Health. The Health and Retirement Study (HRS) is supported by the National Institute on Aging, supplemented by the Social Security Agency, and operated from the Institute for Social Research (ISR) at the University of Michigan. This analysis uses data or information from the Harmonized HRS dataset and Codebook, Version B as of October 2018 developed by the Gateway to Global Aging Data. The development of the Harmonized HRS was funded by the National Institute on Aging (R01 AG030153, RC2 AG036619, 1R03AG043052). For more information, please refer to www.g2aging.org. The data have not been previously presented orally or by poster at scientific meetings.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was supported by the NIH (K23 EY033438-01 to AGH, R01 NS099129-05 to AWW), Parkinson Study Group (Mentored Clinical Research Award to AGH), and Acadia Pharmaceuticals (investigator-initiated award to AWW and DW). The funding organizations had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; or decision to submit the manuscript for publication.
Conflict of interest
Drs. Hamedani, Weintraub, and Willis declare that they have no conflicts of interest.
Ethics approval
This study was approved by the University of Pennsylvania Institutional Review Board.
Consent to participate
Informed consent was obtained from participants in the National Health and Aging Trends Study and Health and Retirement Study at the time of enrollment.
Consent for publication
Not applicable.
Availability of data and material
The data that support the findings of this study are available from the National Health and Aging Trends Study, Health and Retirement Study, and Centers for Medicare and Medicaid Services, but restrictions apply to the availability of some of these data, which were used under license for the current study, and so are not publicly available.
Code availability
The analytic code used to support the findings in this study are available from the corresponding author upon reasonable request.
Author contributions
Dr. Hamedani conceived of the study, performed the primary statistical analysis, and drafted the manuscript. Drs. Weintraub and Willis provided input in study design, results interpretation, and critical manuscript review. Dr. Hamedani had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. All authors have read and approved the final submitted manuscript and agree to be accountable for this work.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hamedani, A.G., Weintraub, D. & Willis, A.W. Hallucinations, Antipsychotic Use, and Mortality in Older Adults with Dementia: Retrospective Cohort Study of Two Medicare-Linked National Health Surveys. Drugs Aging 39, 967–974 (2022). https://doi.org/10.1007/s40266-022-00991-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40266-022-00991-6