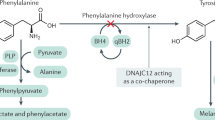
Abstract
Phenylalanine hydroxylase (PAH) deficiency is an inborn error of metabolism that results in elevated phenylalanine levels in blood. The classical form of the disease with phenylalanine level > 1200 µmol/L in blood is called phenylketonuria (PKU) and is associated with severe intellectual disability when untreated. In addition, phenylalanine levels above the therapeutic range in pregnant female patients lead to adverse fetal effects. Lowering the plasma phenylalanine level prevents intellectual disability, maintaining the level in the therapeutic range of 120–360 µmol/L is associated with good outcome for patients as well as their pregnancies. Patient phenotypes are on a continuous spectrum from mild hyperphenylalaninemia to mild PKU, moderate PKU, and severe classic PKU. There is a good correlation between the biochemical phenotype and the patient’s genotype. For over four decades the only available treatment was a very restrictive low phenylalanine diet. This changed in 2007 with the approval of cofactor therapy which is effective in up to 55% of patients depending on the population. Cofactor therapy typically is more effective in patients with milder forms of the disease and less effective in classical PKU. A new therapy has just been approved that can be effective in all patients with PAH deficiency regardless of their degree of enzyme deficiency or the severity of their phenotype. This article reviews the mainstay therapy, adjunct enzyme cofactor therapy, and the newly available enzyme substitution therapy for hyperphenylalaninemia. It also provides an outlook on emerging approaches for hyperphenylalaninemia treatment such as recruiting the microbiome into the therapeutic endeavor as well as therapies under development such as gene therapy.

Similar content being viewed by others
References
Kaufman S. Phenylketonuria: biochemical mechanisms, 1–32. In: Agranoff BW, Aprison MH (eds): Adv Neurochem, 1976, vol 2. Plenum Press, New York.
Camp KM, Parisi MA, Acosta PB, Berry GT, Bilder DA, Blau N, Bodamer OA, Brosco JP, Brown CS, Burlina AB, Burton BK, Chang CS, Coates PM, Cunningham AC, Dobrowolski SF, Ferguson JH, Franklin TD, Frazier DM, Grange DK, Greene CL, Groft SC, Harding CO, Howell RR, Huntington KL, Hyatt-Knorr HD, Jevaji IP, Levy HL, Lichter-Konecki U, Lindegren ML, Lloyd-Puryear MA, Matalon K, MacDonald A, McPheeters ML, Mitchell JJ, Mofidi S, Moseley KD, Mueller CM, Mulberg AE, Nerurkar LS, Ogata BN, Pariser AR, Prasad S, Pridjian G, Rasmussen SA, Reddy UM, Rohr FJ, Singh RH, Sirrs SM, Stremer SE, Tagle DA, Thompson SM, Urv TK, Utz JR, van Spronsen F, Vockley J, Waisbren SE, Weglicki LS, White DA, Whitley CB, Wilfond BS, Yannicelli S, Young JM. Phenylketonuria Scientific Review Conference: state of the science and future research needs. Mol Genet Metab. 2014;112:87–122 (PMID: 24667081).
Bickel H, Gerrard J, Hickmans EM. Influence of phenylalanine intake on phenylketonuria. Lancet. 1953;265(6790):812–3.
Guthrie R, Susi A. A simple phenylalanine method for detecting phenylketonuria in large populations of newborn infants. Pediatrics. 1963;32:338–43.
Mabry CC, Denniston JC, Nelson TL, Son CD. Maternal phenylketonuria. A cause of mental retardation in children without the metabolic defect. N Engl J Med. 1963;269:1404–8.
Williamson ML, Koch R, Azen C, Chang C. Correlates of intelligence test results in treated phenylketonuric children. Pediatrics. 1981;68(2):161–7.
Kaufman S. An evaluation of the possible neurotoxicity of metabolites of phenylalanine. J Pediatr. 1989;114:895–900.
Vockley J, Andersson HC, Antshel KM, Braverman NE, Burton BK, Frazier DM, Mitchell J, Smith WE, Thompson BH, Berry SA, American College of Medical Genetics and Genomics Therapeutics Committee. Phenylalanine hydroxylase deficiency: diagnosis and management guideline. Genet Med. 2014;16(2):188–200 (Erratum in: Genet Med. 2014;16(4):356.).
Acosta P, Yannicelli S. in The Ross Metabolic Formula System Nutrition Support Protocols. ed 4. Columbus: Ross Products Division/Abbot Laboratories; 2001. Protocol 1 – phenylketonuria (PKU).
Güttler F, Wamberg E. On indications for treatment of the hyperphenylalaninemic neonate. Acta Paediatr Scand. 1977;66(3):339–44.
MacLeod EL, Gleason ST, van Calcar SC, Ney DM. Reassessment of phenylalanine tolerance in adults with phenylketonuria is needed as body mass changes. Mol Genet Metab. 2009;98(4):331–7.
Walter JH, White FJ. Blood phenylalanine control in adolescents with phenylketonuria. Int J Adolesc Med Health. 2004;16(1):41–5 (PMID: 15148857).
Pietz J, Kreis R, Rupp A, Mayatepek E, Rating D, Boesch C, Bremer HJ. Large neutral amino acids block phenylalanine transport into brain tissue in patients with phenylketonuria. J Clin Invest. 1999;103(8):1169–78.
Bartholomé K, Byrd DJ, Kaufman S, Milstien S. Atypical phenylketonuria with normal phenylalanine hydroxylase and dihydropteridine reductase activity in vitro. Pediatrics. 1977;59(5):757–61.
Schaub J, Däumling S, Curtius HC, Niederwieser A, Bartholomé K, Viscontini M, Schircks B, Bieri JH. Tetrahydrobiopterin therapy of atypical phenylketonuria due to defective dihydrobiopterin biosynthesis. Arch Dis Child. 1978;53(8):674–6.
Trefz FK, Aulela-Scholz C, Blau N. Successful treatment of phenylketonuria with tetrahydrobiopterin. Eur J Pediatr. 2001;160(5):315.
Kuvan™ (Sapropterin Dihydrochloride) prescribing information, BioMarin Pharmaceutical Inc., Novato, CA, 2013. (v5/2015, revised: 07/2015).
Trefz FK, Burton BK, Longo N, Casanova MM, Gruskin DJ, Dorenbaum A, Kakkis ED, Crombez EA, Grange DK, Harmatz P, Lipson MH, Milanowski A, Randolph LM, Vockley J, Whitley CB, Wolff JA, Bebchuk J, Christ-Schmidt H, Hennermann JB, Sapropterin Study G. Efficacy of sapropterin dihydrochloride in increasing phenylalanine tolerance in children with phenylketonuria: a phase III, randomized, double-blind, placebo-controlled study. J Pediatr. 2009;154:700–7.
Blau N, Hennermann JB, Langenbeck U, Lichter-Konecki U. Diagnosis, classification, and genetics of phenylketonuria and tetrahydrobiopterin (BH4) deficiencies. Mol Genet Metab. 2011;104:S2–9 (PMID: 21937252).
Guldberg P, Levy HL, Hanley WB, Koch R, Matalon R, Rouse BM, Trefz F, de la Cruz F, Henriksen KF, Güttler F. Phenylalanine hydroxylase gene mutations in the United States: report from the Maternal PKU Collaborative Study. Am J Hum Genet. 1996;59(1):84–94.
Grange DK, Hillman RE, Burton BK, Yano S, Vockley J, Fong CT, Hunt J, Mahoney JJ, Phenylketonuria Demographics Outcomes and Safety (PKUDOS) registry; Maternal Phenylketonuria Observational Program (PKU MOMS) sub-registry. Cohen-Pfeffer JL Sapropterin dihydrochloride use in pregnant women with phenylketonuria: an interim report of the PKU MOMS sub-registry. Mol Genet Metab. 2014;112(1):9–16. https://doi.org/10.1016/j.ymgme.2014.02.016 (Epub 2014 Mar 12).
Hoskins JA, Jack G, Wade HE, Peiris RJ, Wright EC, Starr DJ, Stern J. Enzymatic control of phenylalanine intake in phenylketonuria. Lancet. 1980;1(8165):392–4.
Sarkissian CN, Shao Z, Blain F, Peevers R, Su H, Heft R, Chang TM, Scriver CR. A different approach to treatment of phenylketonuria: phenylalanine degradation with recombinant phenylalanine ammonia lyase. Proc Natl Acad Sci USA. 1999;96(5):2339–44 (PMID: 10051643).
Sarkissian CN, Gámez A. Phenylalanine ammonia lyase, enzyme substitution therapy for phenylketonuria, where are we now? Mol Genet Metab. 2005;86(Suppl 1):S22-6 (Epub 2005 Sep 13. Review).
Zori R, Thomas JA, Shur N, Rizzo WB, Decker C, Rosen O, Li M, Schweighardt B, Larimore K, Longo N. Induction, titration, and maintenance dosing regimen in a phase 2 study of pegvaliase for control of blood phenylalanine in adults with phenylketonuria. Mol Genet Metab. 2018;125:217–27 (PMID: 30146451).
Thomas J, Levy H, Amato S, Vockley J, Zori R, Dimmock D, Harding CO, Bilder DA, Weng HH, Olbertz J, Merilainen M, Jiang J, Larimore K, Gupta S, Gu Z, Northrup H, PRISM investigators. Pegvaliase for the treatment of phenylketonuria: Results of a long-term phase 3 clinical trial program (PRISM). Mol Genet Metab. 2018;124(1):27–38 (PMID: 29653686).
Longo N, Zori R, Wasserstein MP, Vockley J, Burton BK, Decker C, Li M, Lau K, Jiang J, Larimore K, Thomas JA. Long-term safety and efficacy of pegvaliase for the treatment of phenylketonuria in adults: combined phase 2 outcomes through PAL-003 extension study. Orphanet J Rare Dis. 2018;13:108 (PMID: 29973227).
Sarkissian CN, Gámez A, Wang L, Charbonneau M, Fitzpatrick P, Lemontt JF, Zhao B, Vellard M, Bell SM, Henschell C, Lambert A, Tsuruda L, Stevens RC, Scriver CR. Preclinical evaluation of multiple species of PEGylated recombinant phenylalanine ammonia lyase for the treatment of phenylketonuria. Proc Natl Acad Sci USA. 2008;105(52):20894–9. https://doi.org/10.1073/pnas.0808421105.
Palynziq prescribing information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/761079s000lbl.pdf. Accessed 9 April 2018.
Harding CO, Gillingham MB, Hamman K, Clark H, Goebel-Daghighi E, Bird A, Koeberl DD. Complete correction of hyperphenylalaninemia following liver-directed, recombinant AAV2/8 vector-mediated gene therapy in murine phenylketonuria. Gene Ther. 2006;13:457–62.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No financial assistance was obtained to prepare this publication.
Conflict of interest
ULK and JV have participated in clinical trials sponsored by BioMarin Pharmaceuticals, manufacturer of sapropterin and pegvaliase. JV is funded for research on PKU by the National Institutes of Health. JV has served as a consultant for Homology Pharmaceuticals, Moderna Pharmaceuticals, Synlogic Pharmaceuticals, ATG gene therapies, and Kaleido Pharmaceuticals.
Rights and permissions
About this article
Cite this article
Lichter-Konecki, U., Vockley, J. Phenylketonuria: Current Treatments and Future Developments. Drugs 79, 495–500 (2019). https://doi.org/10.1007/s40265-019-01079-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40265-019-01079-z