Abstract
Patients with viral infections of the central nervous system (CNS) may present with a variety of neurological symptoms, most commonly dominated by either encephalitis or meningitis. The aetiological panorama varies in different parts of the world as well as over time. Thus, virological first-line diagnostics must be adapted to the current epidemiological situation and to the individual patient history, including recent travels. This review focuses on the diagnostics and treatment of viral CNS infections in the immunocompetent host from a Northern European perspective. Effective vaccines are available for viruses such as poliovirus and tick-borne encephalitis virus (TBEV) and for the childhood diseases morbilli (measles), rubella (German measles), parotitis (mumps) and varicella (chickenpox). However, cases do appear due to suboptimal immunization rates. In viral CNS infections, epidemiological surveillance is essential for establishing preventive strategies and for detecting emerging viruses. Knowledge of the possibilities and limitations of diagnostic methods for specific viral CNS infections is vital. A positive cerebral spinal fluid (CSF) polymerase chain reaction (PCR) finding is usually reliable for aetiological diagnosis. The demonstration of intrathecal antibody synthesis is useful for confirming the aetiology in a later stage of disease, hitherto sufficiently evaluated in herpes simplex encephalitis (HSE) and tick-borne encephalitis (TBE). Despite improved virological and differential diagnostic methods, aetiology remains unknown in about half of the cases with suspected viral encephalitis. Antiviral treatment is available chiefly for infections caused by herpesviruses, and acyclovir (aciclovir) is the drug of choice for empirical therapy in suspected viral encephalitis. However, randomized, controlled antiviral trials have only been conducted for HSE, while such studies are lacking in other viral CNS infections. Viral cytolysis and immune-mediated mechanisms may contribute to varying extents to neurological damage. Although the brain damage is believed to depend, to a varying degree, on the intrathecal host immune response, the use of corticosteroids in viral CNS infections is scarcely studied, as is specific treatment for neuroinflammation. Improved antiviral and immunomodulating treatment is desirable. Since neurological sequelae are still abundant, follow-up after severe viral CNS disease must include a neuropsychological assessment and an individually adapted rehabilitation plan.
Similar content being viewed by others
1 General Considerations
Acute viral infections of the central nervous system (CNS) are caused by a wide range of viral agents that elicit various neurological manifestations through different pathogenic mechanisms. The clinical picture is not always distinct and clear-cut but rather reflects a continuous spectrum, with overlapping features of meningoencephalitis, meningomyelitis, or meningoencephalomyelitis. However, patients often present with predominant symptoms of either meningitis, myelitis or encephalitis.
Viral encephalitis in the adult is rare, with an estimated incidence of 1.4–2.2 per 100,000 [1–3], but a large percentage of the patients develop neurological sequelae, with substantial impairment of quality of life [4–8]. However, the majority of viral CNS infections comprise self-limiting acute aseptic meningitis, which is up to ten times more common [3, 9]. The true incidence of viral myelitis is not known, being even more rare than encephalitis.
1.1 Dynamics in the Aetiological Panorama
The aetiology of viral CNS infections may show substantial geographic differences due to variations in the vector and reservoir distribution and their viral agents. Epidemic outbreaks with spread of viral infections to new areas influence the panorama. A wide variety of emerging and re-emerging viruses with novel neuropathogenic properties also have the potential to alter the distribution of CNS diseases globally.
Immunization against a number of infections such as polio, measles, mumps and rubella has diminished the importance of these viruses as encephalitic agents. Improved diagnostic methods and possibly increased propensity to search for the aetiology may also contribute to the observation of a changing aetiological panorama.
Important viruses that cause CNS diseases, such as West Nile virus, Japanese B encephalitis virus, and other arthropod-borne viruses from the toga- and bunya-virus families, along with rabies, emphasize the importance of knowing the patient’s travel history and the viral epidemiology in the area visited. However, it is beyond the scope of this paper to review every possible virus that may cause CNS infection worldwide and we will concentrate on viral CNS infections from a Northern European epidemiological perspective.
1.2 Virological Diagnostics are Highly Valuable
In viral CNS infections, preceding or concomitant symptoms of infection, such as fever, respiratory or gastrointestinal symptoms, may occur, but may also be lacking. Virological aetiological diagnostic tests are essential, as they enable the detection of treatable serious infections. A verified viral aetiology enables correct medical intervention, and unnecessary examinations, antibacterial treatments and over-consumption of medical care further on may be avoided. From the individual patient’s point of view, the viral aetiological diagnosis is of benefit per se, especially in cases with a subsequent prolonged recovery or complicating events. Furthermore, by determining a causative agent, prognostic information is made possible.
The maintenance and development of viral diagnostics is the basis for epidemiological surveillance, and a prerequisite for detecting emerging and re-emerging viruses with changed clinical appearances. Surveillance is necessary for establishing immunization strategies, both within a country and globally.
1.3 Viral Diagnostics in Central Nervous System (CNS) Disease
During recent decades, the development and use of modern molecular techniques has greatly improved the diagnostic yield. Today, a specific aetiological agent is detected in approximately 50 % of cases of suspected infectious encephalitis [10, 11] and 60–70 % of cases with aseptic meningitis [3, 12].
Knowledge of the possibilities and limitations of the diagnostic methods for a specific viral CNS infection is of vital importance, since diagnostic sensitivity is highly dependent on the timing of sampling and the choice of analytic method (Fig. 1). The basis for interpretation of test results varies in solidity and this provides support for a causal association with different strengths. A positive virus isolation from cerebrospinal fluid (CSF) provides strong evidence for an on-going CNS infection with replicating virus, but the overall sensitivity is not sufficient. Nowadays, detection of viral RNA or DNA in the CSF is the primary choice of diagnostics in the acute phase of disease. Qualitative polymerase chain reaction (PCR) has been replaced by real-time PCR with equal or higher sensitivity and specificity, along with the potential additional advantages of quantitative information. However, the clinical significance of the finding of a certain amount of a specific viral genome in the CSF remains unclear. The quantitative data might be valuable for treatment monitoring as well as for giving prognostic information. Careful interpretation is required since, for example, Epstein Barr virus (EBV) DNA can be found concomitantly in the CSF as a possible bystander to other infectious CNS diseases, such as tuberculosis, bacterial meningitis and herpes simplex encephalitis (HSE) [13–16]. Detection of human herpesvirus (HHV)-6 DNA in the CSF might express an inherited integrated genome rather than being the cause of the CNS disease [17, 18].
The demonstration of intrathecal antibody synthesis is useful for verifying the aetiology in a later stage of disease, but the result must be interpreted in relation to the extent of the blood–brain barrier (BBB) damage. Correction for the damage of the BBB could be performed by Reibers formula or other methods in the laboratory. The intrathecal antibody synthesis has been evaluated in HSE and tick-borne encephalitis (TBE) and, to some extent, also in varicella zoster virus (VZV) neurological infections [19–21], while in other viral CNS infections, the diagnostic use of intrathecal antibodies has been even less evaluated hitherto. Seroconversion, IgM detection, or a significant serum antibody titre rise against a specific viral antigen indicates a present or recent infection, but does not necessarily prove a causal connection with the CNS symptoms, which is also the case with findings of a virus from peripheral sites, such as nasopharyngeal or fecal specimens or skin blisters.
On admission of a patient with severe neurological symptoms indicating an infectious disease, differential non-infectious causes, such as autoimmune or autoinflammatory conditions, vascular and metabolic disorders, drug toxicity and malignancies should be taken into consideration [10, 11, 22]. In viral disease, the pathogeneses may differ and comprise a spectrum of mechanisms. The distinction between a viral infectious and a post-infectious CNS disease presents a challenge. The virus infection may be cytolytic and/or noncytolytic and elicit a varying degree of immunologically mediated inflammation. The clinical manifestations are determined by the virus itself and its properties and, not least, the general immune status of the host, as well as factors involving the specific constitutive and adaptive immune defence.
In this review, we concentrate primarily on adult immunocompetent patients who present with a clinical syndrome consistent with viral encephalitis or meningitis. The first part focuses on definitions of suspected viral meningitis, encephalitis and myelitis, and suggested algorithms for diagnostic approaches and empiric antiviral therapy in the acute phase. Thereafter, important and common viruses are reviewed with regard to clinical features, epidemiology and diagnostics. Therapy recommendations are graded according to the Infectious Diseases Society of America grading system [23]. We emphasize that management algorithms must be adapted to the current epidemiological situation.
2 Definitions and Initial Management
It is of vital importance to aim at defining the patient’s present clinical condition, i.e. meningitis, encephalitis, myelitis (Table 1), since this directs acute management. For practical reasons, the diagnostic procedures and empiric antiviral therapy are chosen on the basis of the predominant symptoms. If encephalitic symptoms are present, management should follow the suggested algorithm for encephalitis (Fig. 2). A thorough patient history is of the utmost importance (Table 2) and may indicate viral agents to be searched for in the first-line diagnostics. In suspected viral encephalitis, the clinical diagnosis is based on the patient history, clinical symptoms and signs, along with CSF findings, neuroradiology, and electroencephalogram (EEG) (Fig. 2).
Initial management on suspicion of acute viral encephalitis. CRP C-reactive protein, CSF cerebrospinal fluid, CT computed tomography, DNA deoxyribonucleic acid, EEG electroencephalogram, HIV human immunodeficiency virus, HSV herpes simplex virus, IgM immunoglobulin M, IV intravenous, MRI magnetic resonance imaging, P plasma, PCR polymerase chain reaction, PC-INR prothrombin complex International normalized ratio, RLS reaction level scale, S serum, TBE tick-borne encephalitis, tid three times daily, TPC thrombocyte particle concentration, VZV varicella zoster virus
Antiviral therapy, i.e. aciclovir, should be administered on the mere suspicion of HSE and without awaiting the result of aetiological diagnostics, as early institution of antiviral treatment correlates with a better prognosis [4, 5]. If the first-line diagnostics turn out to be negative, a second lumbar puncture is compulsory to rule out false-negative herpes simplex virus (HSV) and VZV PCR analyses [24–29]. In the absence of clinical improvement, further investigation is needed (Fig. 3). When managing a patient with suspected viral encephalitis, bacteria and other infectious or non-infectious causes should always be borne in mind. Recently published British guidelines on suspected viral encephalitis contain a broad review of differential diagnostics [22]. The liberal approach to starting antiviral therapy demands a strategy for withdrawal of the therapy when treatable viruses have been excluded. A supportive algorithm is suggested in Fig. 4.
Diagnostics in acute encephalitis with unknown aetiology after first lumbar puncture. CSF cerebrospinal fluid, CMV cytomegalovirus, DNA deoxyribonucleic acid, EBV Epstein Barr virus, EEG electroencephalogram, HHV-6 human herpesvirus 6, HIV human immunodeficiency virus, HSV herpes simplex virus, IgG immunoglobulin G, IgM immunoglobulin M, MRI magnetic resonance imaging, PCR polymerase chain reaction, S serum, Tb tuberculosis, TBE tick-borne encephalitis, VZV varicella zoster virus
In suspected viral meningitis, a similar algorithm may be used (Fig. 5). Antiviral treatment in viral meningitis is recommended in primary HSV meningitis, in analogy with the treatment of primary genital herpes [30]. In recurrent HSV meningitis and in VZV meningitis, antiviral treatment is optional, while awaiting studies in this field. Further examinations may be needed if the results of first line-diagnostics are negative, particularly in the absence of spontaneous recovery (Fig. 6). Viral myelitis, which is a rare disease, should be promptly treated with aciclovir, similarly to encephalitis, after the initial magnetic resonance imaging (MRI), lumbar puncture and serum sampling. The suggested initial search for microbiological causes is shown in Fig. 7, but modifications may be needed according to the epidemiological circumstances.
Initial management on suspicion of acute viral meningitis. CRP C-reactive protein, CSF cerebrospinal fluid, DNA deoxyribonucleic acid, HIV human immunodeficiency virus, HSV herpes simplex virus, IgM immunoglobulin M, IV intravenous, P plasma, PCR polymerase chain reaction, po per oral, RNA ribonucleic acid, S serum, TBE tick-borne encephalitis, tid three times daily, VZV varicella zoster virus
Diagnostics in acute meningitis with unknown aetiology after first lumbal puncture and need for further examinations due to severe symptoms. CMV cytomegalovirus, CSF cerebrospinal fluid, CT computed tomography, DNA deoxyribonucleic acid, EBV Epstein Barr virus, HHV-6 human herpesvirus 6, HIV human immunodeficiency virus, IgM immunoglobulin M, MRI magnetic resonance imaging, PCR polymerase chain reaction, S serum, Tb tuberculosis, TBE tick-borne encephalitis
Initial management on suspicion of acute viral myelitis. CMV cytomegalovirus, CRP C-reactive protein, CSF cerebrospinal fluid, DNA deoxyribonucleic acid, EBV Epstein Barr virus, HHV-6 human herpesvirus 6, HIV human immunodeficiency virus, HSV herpes simplex virus, HTLV-1 human T-lymphotropic virus 1, IgM immunoglobulin M, IV intravenous, MRI magnetic resonance imaging, od once daily, P plasma, PCR polymerase chain reaction, RNA ribonucleic acid, S serum, TBE tick-borne encephalitis, tid three times daily, VZV varicella zoster virus
3 Herpes Simplex Encephalitis
3.1 Clinical Features and Epidemiology
Herpes simplex virus infection is common worldwide. In rare cases, the virus causes an acute, aggressive, focal, necrotizing inflammation mainly localized in the temporal and subfrontal areas of the brain: HSE. However, HSV is the most common viral cause of sporadic encephalitis. Clinically, a prodromal phase with high fever and headache may precede the neurological symptoms [31]. On admission, the HSE patient often presents with high fever and headache in conjunction with diffuse and focal neurological symptoms, including disorientation, altered consciousness, personality changes, behavioural alterations, seizures, dysphasia and paresis [31–33]. Later studies report a lower frequency of paresis and decreased consciousness, which possibly reflects increased awareness of HSE and thereby identification of patients in an earlier stage of disease [4–6]. Herpetic blisters are not more prevalent among HSE patients [33] and certain risk factors or triggering events have not been identified.
HSE is associated with high mortality (more than 70 %) without antiviral treatment and high morbidity in spite of antiviral treatment. The most common disabling sequelae are non-verbal and verbal memory impairment, and the anterograde memory is particularly affected. Personality and/or behavioural abnormalities are found in almost half of the patients, and psychiatric symptoms, depression, anxiety, insomnia and emotional lability in up to one third. Anosmia, epilepsia and dysphasia are also frequent [5, 34, 35].
HSE occurs globally, with an incidence of 2–4 per million [32, 36, 37]. More than 90 % of the cases are caused by HSV-1 and the remainder by HSV-2 [25, 38]. After the neonatal period, the disease appears at all ages, but more than 70 % of affected individuals are older than 50 years [37]. There is no gender difference.
3.2 Diagnostic Procedures
3.2.1 Cerebrospinal Fluid (CSF) Findings and Virological Analyses
A mild to moderate CSF pleocytosis with a predominance of lymphocytes is usually found, along with a moderately increased protein concentration. Fewer than 5 leukocytes × 106/L has been reported in 3–8 % of HSE cases on admission [4, 33, 38], but elevated leukocyte counts and protein concentrations can usually be demonstrated a day or two later [4, 33]. An increased number of erythrocytes is frequently found, and xanthochromia is sometimes found. As a rule, the glucose CSF/serum ratio is normal, although a slight decrease is occasionally found.
The HSE diagnosis is verified in the acute stage by detection of HSV DNA (HSV-1 or HSV-2) in the CSF by PCR [24]. The method is sensitive (>95 %), and DNA is present in the CSF during the first week from the start of antiviral treatment, and disappears during the following 1–2 weeks [24, 25]. False-negative results appear very early, i.e. 1–3 days from the onset of neurological symptoms [24, 25, 29]. In a later stage, the diagnosis may be confirmed by demonstrating HSV-specific intrathecal antibody synthesis, the sensitivity of which increases with time and approaches 100 % after 10–12 days [19]. Intrathecal HSV antibodies persist decades after HSE [39].
3.3 Neuroradiology and Neurophysiology
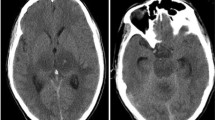
Urgent neuroradiological examination is essential, not least to differentiate encephalitis from focal expansive processes of other origin. MRI is superior to computed tomography (CT) for the detection of HSE lesions [40–43]. In the early phase, oedema is discerned mainly in the temporal lobe. MRI without pathological findings during any part of the course of HSE is rare. A CT scan typically shows low-attenuating changes, but abnormalities may be absent on admission [4, 44]. Repeated CT images demonstrate focal temporal lesions in almost 90 % of cases. Furthermore, haemorrhages may be visualized in the encephalitic area. Early encephalitic changes dominate on one side but may spread to both hemispheres. Lesions often involve the inferior frontal lobes but may also appear in the parietal and occipital lobes [4, 44] and elsewhere [40, 45, 46]. The inflammation may cause a visible mass effect.
An abnormal EEG is the rule in HSE, but the specificity is low and the pattern fluctuates during the course [47]. Non-specific slowing or asymmetric spikes and slow waves are frequent. Intermittent periodic lateralizing epileptiform discharges (PLED) are associated with HSE, although not pathognomonic [47]. EEG recordings are important for revealing nonconvulsive epilepsy in patients with decreased consciousness [48].
3.4 Therapy
Prompt administration of aciclovir is mandatory since delay is associated with a worsened prognosis. Without antiviral therapy, the mortality is high, exceeding 70 %, as is neurological morbidity among survivors. Intravenous aciclovir 10 mg/kg three times daily for 10 days significantly decreased mortality and morbidity, as shown in two large studies in the 1980s [32, 36]. However, considerable mortality (19–28 %) and morbidity was still found after 6 months, which has subsequently led to prolonged treatment of up to 14–21 days [23] (Table 3). Later studies have reported lower mortality (6–15 %), to which longer treatment duration, identification of milder cases, earlier initiation of antiviral therapy, and younger age might have contributed [4–6, 37]. The amount of HSV DNA in the CSF at admission has not been shown to be a prognostic factor, but persistent PCR positivity in the CSF after 10–14 days of treatment correlates with a poor outcome [49], and prolonged antiviral therapy is recommended in such cases [50]. A large randomized, placebo-controlled, multicentre study of long-term treatment with valacyclovir (valaciclovir), after termination of intravenous aciclovir, has recently been completed and is currently under evaluation [51]. A higher dose of intravenous aciclovir, i.e. 15 mg/kg three times daily, is often administered to young individuals without renal impairment, although it has not been evaluated in prospective studies. In one retrospective study, a high dose of aciclovir was not associated with a better outcome [52]. Corticosteroids are often administered in the acute phase in individuals with clinical signs of increased intracranial pressure (ICP) and mass effect. The role of ICP monitoring is uncertain. Approximately one third of HSE patients (n = 46) had initially elevated ICP with a peak on the 12th day of the disease [31]. Extensive brain cell destruction by viral cytolysis and also a vigorous intrathecal immune response contribute to the brain damage. In a retrospective study of 45 patients, corticosteroids in conjunction with aciclovir correlated with improved outcomes compared with aciclovir alone [53]. A prospective study of dexamethasone in addition to aciclovir in the initial phase of HSE is under way (GACHE; German trial of Acyclovir and Corticosteroids in Herpes-simplex-virus-Encephalitis) [54].
Seizures are common, and anticonvulsive therapy is often administered, although studies on seizure therapy and prophylaxis in HSE are lacking and the optimal duration of treatment is unknown. Initially, benzodiazepines and intravenous phenytoin are commonly used. In a Swedish national study, seizures were found to be the most frequent cause of rehospitalization in the course of HSE, occurring in as many as 20 % of 236 patients and often after several months (median 9.3 months) [37]. Anti-epileptic prophylaxis is therefore recommended during the first year in cases with seizures or findings of epileptic EEG activity. Episodes of relapsing encephalitic symptoms have been reported in up to 10 % of HSE cases. CSF PCR has mostly been negative and an immunologically mediated cytotoxicity has been suggested [55]. However, HSV DNA has been demonstrated in single cases and low-grade viral replication is not excluded, which supports the rationale for using intravenous aciclovir in relapse, often given for 2 weeks, along with corticosteroids in tapering doses.
4 Herpes Simplex Meningitis
4.1 Clinical Features and Epidemiology
HSV-2 causes vesicular lesions chiefly in the genital or lumbosacral region, but can also induce a wide spectrum of neurological manifestations, mainly aseptic meningitis often in association with myelitis and radiculitis or neuritis. Initial transient mild-to-moderate encephalitis symptoms occur. Reactivation of the virus may give rise to recurrent neurological symptoms, and HSV-2 is the major cause of recurrent aseptic meningitis [56, 57].
In primary HSV-2 meningitis, headache, usually described as intense, develops during 2–3 days [57], together with varying degrees of neck stiffness, photophobia, nausea and vomiting. Fever is common but not an obligatory finding [58]. The acute symptoms resolve spontaneously, although sometimes after protracted illness [57, 59, 60]. Symptoms such as headache, lability, concentration disabilities and fatigue may last for several weeks or months [57, 59–61, 65].
Herpetic mucocutaneous lesions may precede the meningitis by about 2–14 days [57, 59, 60, 62], but sometimes appear after the meningitis and the two manifestations may occur independently. However, more than 50 % of patients with HSV meningitis have never recognized any herpetic blisters [9, 58].
HSV-2 meningitis, particularly the first episode, is associated with neurological symptoms, like urinary retention, constipation, dysesthesia, radiating pain or weakness in the lumbosacral area and/or lower limbs, indicating sacral myeloradiculitis, in approximately half of the patients [57, 58, 61].
HSV-2 meningitis carries the risk of a broad spectrum of future neurological morbidity, including recurrent meningitis, myelitis and radiculitis [57, 61]. The recurrent meningitis episodes vary in intensity but tend to occur with milder clinical symptoms and be of shorter duration (2–5 days) than the primary attack [57, 63]. At least 20–30 % of patients experience recurrent bouts of meningitis [64, 65].
HSV-2 is one of the major causes of aseptic meningitis and accounted for almost 20 % of consecutive cases in a recent Swedish survey [12]. Most cases occur in young adults [12, 57–61]. A female predominance is striking, with a female-to-male ratio of about 2:1–6:1 [57, 60, 61, 66]. Although genital herpes caused by HSV-1 is increasing, meningitis due to HSV-1 is rare [12].
4.2 Diagnostic Procedures
The diagnosis of herpetic meningitis begins with a thorough history covering previous herpetic manifestations, such as mucocutaneous herpes and/or bouts of aseptic meningitis (Table 2).
4.2.1 CSF Findings and Virological Analyses
In primary meningitis, a mild or, more often, moderate pleocytosis with predominantly lymphocytes averaging around 400 × 106/L (range 5–1,100) is seen. The CSF protein is increased, and the CSF/serum glucose ratio and the CSF lactate are often normal, but hypoglycorrhea and a slightly increased lactate may be found [9, 60, 67]. In recurrent meningitis, the inflammatory reaction is usually less pronounced [9].
Detection of HSV-2 DNA in the CSF by PCR, preferably quantitative, verifies the diagnosis. The estimated sensitivity is almost 90 and 70 % in primary and recurrent meningitis, respectively [12]. The HSV-2 viral load is higher in primary than in recurrent meningitis and correlates with the degree of inflammation [9]. Isolation of HSV in the CSF is less sensitive and almost exclusively successful in primary meningitis [57, 59, 68, 69]. Demonstration of HSV-2 DNA or viral antigen from mucous membrane or skin lesions, if present, supports the diagnosis in PCR-negative cases.
A seroconversion to type-specific HSV antigen demonstrated by enzyme-linked immunosorbent assay (ELISA) may verify the diagnosis in primary infections. The majority of patients with HSV-2 meningitis have no HSV-1 antibodies in serum, thus indicating a lack of protection against symptomatic HSV-2 disease [65, 70]. Seroconversion to HSV-2 may appear late, after 4–5 weeks [57] or even later. In recurrent meningitis, significant serum titre rises are usually not observed [57]. The intrathecal antibody response in HSV meningitis has not been fully evaluated.
4.3 Therapy and Prophylaxis
Acute HSV meningitis is a self-limiting disease that also heals without specific antiviral treatment. Controlled trials of acute treatment of herpes meningitis are lacking. In primary HSV meningitis, with its protracted course and association with myelitic symptoms, it seems justified to give antiviral treatment in analogy with the recommendations in primary genital herpes [30]. If the patient presents with severe symptoms and/or nausea and vomiting, initial intravenous aciclovir therapy may be administered. Otherwise, oral therapy with valaciclovir may be given, in doses ensuring adequate CSF concentrations [71] (Fig. 5; Table 3). In recurrent meningitis, prompt treatment after the onset of symptoms may be beneficial, but since the recurrent episodes are often less severe, a decision to avoid antiviral medication may be arrived at.
Successful prevention of meningitis with antiviral suppression has been reported in small case series [72, 73]. Tailored suppressive treatment may be considered in patients with frequent meningitis recurrences, although recent data do not provide support for general antiviral suppression with valaciclovir 0.5 g twice daily following HSV-2 meningitis [65]. Identifiable triggering factors should, if possible, be eliminated to prevent recurrences. Vaccines against HSV are currently extensively investigated, but have not been shown to be sufficiently effective hitherto, and the impact on HSV CNS viral disease remains to be evaluated.
5 Varicella Zoster Virus
5.1 Clinical Features and Epidemiology
VZV causes a wide spectrum of CNS infections, including meningitis, acute cerebellar ataxia (ACA), meningoencephalitis, encephalitis, myelitis, cranial nerve involvement, encephalopathy, brain stem encephalitis and brain infarction or bleeding. Neurological complications are seen in both children and adults. Approximately half of the patients have concomitant varicella or shingles [74]. In adults, most CNS infections are caused by reactivated VZV. The reported frequency of VZV infections of the CNS is dependent on the diagnostic procedures performed and the national vaccination status. Without varicella immunization, an increasing incidence of CNS complications has been noted [8, 75–77], compared with a low incidence reported in countries practicing varicella vaccination [78].
ACA is the most common manifestation [79], occurring primarily in pre-school children, with the onset usually 1–3 weeks after the primary infection [80].
Meningitis occurs in all age groups, and meningoencephalitis, encephalitis and Ramsay Hunt syndrome, i.e. facial palsy with zoster oticus, or facial palsy without blisters, dominate in the older ages [8]. Ramsay Hunt syndrome is often accompanied by involvement of other cranial nerves with symptoms such as vertigo, deafness, tinnitus and nystagmus [81].
VZV may cause vasculopathies, which can induce ischaemic infarction, aneurysm, subarachnoid or cerebral haemorrhage and carotid dissection in adults and children [82–85]. Nagel et al [85] described 30 adult CNS vasculopathy cases with verified virological diagnosis either by findings of VZV DNA in the CSF or by demonstration of specific intrathecal VZV antibody synthesis. Ciccone et al. [83] conducted a literature review and listed 70 cases of stroke syndrome associated with chickenpox or herpes zoster in children. In the majority of the cases only a temporal association between infarction and varicella was reported, but some cases were virologically verified by detection of VZV antigen in cerebral vessels at autopsy or CSF findings [83, 86–88]. Recurrent transient ischaemic attacks or reinfarction may occur after stroke secondary to varicella [86].
The long-term neurological sequelae following VZV CNS infections are relatively scarcely documented. A spectrum from recovery to disabling cognition and memory disabilities is reported [8, 89].
5.2 Diagnostic Procedures
5.2.1 CSF Findings and Virological Analyses
Mononuclear pleocytosis and an elevated protein content are seen most often, but may be absent in CNS vasculopathy. The sensitivity and specificity of CSF PCR has not been studied systematically. A high viral load is seen in meningitis [8], meningoencephalitis and encephalitis [8, 90], while patients with cranial nerve affections have lower levels [8]. In patients with vasculopathy, PCR is less sensitive, possibly due to a localized infection within arteries or sampling in a later phase, and an intrathecal antibody analysis is often necessary [21, 85]. Serological analyses have been hampered by cross-reactivity between HSV and VZV [39, 91], which is overcome by using more specific antigens [92]. Viral DNA from blisters might support, but not necessarily prove, a causal connection with the CNS symptoms.
5.2.2 Neuroradiology and Neurophysiology
Radiological findings in VZV CNS infections are often absent [93], but contrast enhancement on MRI may be seen in the brain, brainstem and cranial nerves or spinal cord in both grey and white matter [94, 95]. MRI with diffusion sequences may show ischaemic lesions or infarctions. In CNS vasculopathy, conventional angiography and CT and MR angiography are all hampered by a relatively low sensitivity and may be normal or show segmental constrictions or occlusions with post-stenotic dilatation [85]. In immunocompetent patients, large vessels, such as arteria cerebri media and anterior, are most often involved [96], but involvement of smaller arteries has also recently been demonstrated [85]. Conventional angiography is required to visualize small vessels in the brain and may show VZV vascular changes.
5.2.3 Therapy and Prophylaxis
Reliable treatment studies on VZV infections are lacking and most recommendations are based on case series or case reports. Based on the alleviation of disease severity, antiviral treatment is usually recommended in adult immunocompetent patients with herpes zoster [97]. Intravenous aciclovir is recommended [98] in myelitis, meningoencephalitis, encephalitis, brain infarction and severe cases of ACA (Table 3). VZV is less sensitive to aciclovir than HSV, and a higher dose may be used in younger patients without renal impairment, while adjustment of the dose to renal function may be necessary in older individuals (Table 3). The pathogenesis in encephalitis and meningoencephalitis has not been fully elucidated, and the value of additional corticosteroids has not been investigated systematically. However, in VZV vasculopathy, addition of corticosteroids is advisable (Table 3).
Untreated Ramsay Hunt syndrome may be associated with defective healing, especially in the elderly and in patients with total facial paralysis [99]. Some studies support antiviral therapy [100–102], but in the only small randomized, controlled trial published, no effect of antiviral therapy was found [103]. A Cochrane analysis indicated that randomized controlled trials investigating adjuvant corticosteroids in Ramsay Hunt syndrome were lacking [104]. Two non-randomized studies indicated that patients given combined treatment had fewer sequelae than patients treated with corticosteroids only [100, 101]. When given, early treatment seems to be beneficial [102].
Varicella immunization has markedly decreased the incidence of primary infections [105], and CNS complications. Post-marketing surveillance has also shown the vaccine strain to be less virulent than the wild-type in terms of ability to reactivate [93]. Since a vaccine effective in preventing herpes zoster, used in older age groups [106], is also available, we can foresee a reduction in zoster incidence and neurological morbidity.
6 Cytomegalovirus, Epstein Barr Virus and Human Herpesvirus 6
Cytomegalovirus (CMV), EBV and HHV-6 are all members of the HHV family and thereby share some characteristics. Primary infection often occurs early in life and is usually asymptomatic, as is later reactivation of the virus. Immunosuppression increases the risk of symptomatic CNS infections with these viruses, but disease occasionally also occurs in the immunocompetent host.
6.1 Clinical Features and Epidemiology
CMV infects a large proportion of infants during the first year of life, and the seroprevalence steadily increases with age, reaching 60–90 % in an adult population. CMV infection is usually mild or asymptomatic, but serious disease, including neurological manifestations, occurs primarily in immunocompromised but also in immunocompetent adults [107]. CNS infection with CMV is manifested as meningitis, encephalitis or myelitis, and CMV infection may precede Guillain–Barre syndrome, while ventriculitis is seen in advanced immunodeficiency syndromes [107–109]. In encephalitis, there is an acute onset with fever and headache in combination with neurological symptoms such as personality changes, confusion, altered consciousness and seizures or focal neurological signs, including cranial nerve palsy.
EBV and HHV-6 are ubiquitous viruses that infect a vast majority early in life, and the seroprevalence is >90 % in adults. In spite of this, neurological complications are uncommon. In larger series of patients with encephalitis, with and without immunosuppression, each one of these herpesviruses is identified as the causative agent in 0.5–2.5 % of cases [11, 110]. In a recent publication, detectable HHV-6 DNA was demonstrated in a larger proportion of patients with encephalitis, but this finding needs to be confirmed [111]. There are two types of HHV-6: A and B. Both have recently been shown to be integrated in chromosomal DNA in a minor proportion of the population [17]. This phenomenon is not associated with any known clinical symptoms, but it can lead to diagnostic difficulties, since these individuals always have high amounts of viral DNA in the blood and often detectable viral DNA in the CSF [18, 112].
A symptomatic EBV infection typically causes mononucleosis, but when affecting the CNS, the virus causes a wide range of neurological complications, such as meningitis, encephalitis, cerebellitis, polyradiculitis, myelitis, cranial nerve palsy and peripheral neuropathy [113]. CNS affection is probably more common during primary EBV infection, but it may occur in reactivated infection. EBV is also associated with primary CNS lymphomas in patients with acquired immunodeficiency syndrome (AIDS).
HHV-6 is the causative agent of exanthema subitum in children. During primary infection, HHV-6 DNA can often be detected in the CSF without concurrent neurological symptoms [114]. In children with recurrent febrile convulsions, HHV-6 DNA has been detected in the CSF, possibly suggesting viral persistence or reactivation of the virus in the CNS [115]. However, the virus may also cause meningitis, encephalitis or myelitis. Neurological complications are most common in immunosuppressed individuals, especially after haematopoietic stem cell transplantation, but have also been reported in immunocompetent children and adults [116, 117].
6.2 Diagnostic Procedures
6.2.1 CSF Findings and Virological Analyses
CNS infection with CMV, EBV or HHV-6 is diagnosed mainly by CSF analysis. Typically, a mild to moderate lymphocytic pleocytosis, a normal or slightly elevated protein concentration and a normal glucose CSF/serum ratio are seen. A definitive diagnosis is made by detection of viral DNA by PCR. Detectable CMV DNA in CSF is strongly indicative of CMV-related CNS disease. However, EBV and HHV-6 positive DNA findings must be interpreted with some caution, since small amounts of viral DNA can be detected in CSF without definitive clinical significance and sometimes together with other microbiological findings [118, 119]. Thus, thorough differential diagnostics, adjusted to the patient’s symptoms, is recommended. The relationship between the amounts of viral DNA and the probability of a true EBV CNS infection remains to be clarified [14]. Chromosomal integration of HHV-6 DNA can lead to detectable levels of HHV-6 DNA in CSF without clinical significance. If viral integration is suspected, additional analyses (e.g. quantitative PCR comparing whole blood and serum, type-specific HHV-6 PCR or comparison with blood samples from the patient’s parents) can be done to confirm or exclude integration.
For other herpesviruses, e.g. HSV and VZV, detection of intrathecal antibody production is a reliable method for confirming CNS infection, although it usually requires repeat sampling a few weeks after the onset of symptoms. However, for CMV, EBV and HHV-6, the evidence supporting use of this method is weak. Intrathecal antibody production has been shown to be less sensitive and specific than PCR analysis for diagnosing CMV infection in AIDS patients [120]. There are case reports of CNS infections with EBV and HHV-6 describing intrathecal antibody production, but no systematic analysis comparing intrathecal antibody production with detection of viral DNA has been published [121, 122].
6.2.2 Neuroradiology
Neuroimaging findings in encephalitis due to CMV, EBV or HHV-6 can be normal or show variable degrees and localizations of abnormalities, appearing as low attenuation on CT or an increased T2-weighted signal on MRI. In CMV meningoencephalitis, the MRI findings are unspecific and similar to those of other viral encephalitides. In a review of 101 cases of EBV infection with CNS manifestations, it was suggested that the distribution of radiological abnormalities is a prognostic marker. The most favourable outcome was found in patients with isolated hemispheric involvement. Half of the patients with thalamic involvement developed sequelae, and the highest mortality was found in patients with isolated brain stem involvement [123].
6.3 Therapy
There are no controlled trials of antiviral treatment of CNS infection caused by CMV, EBV, or HHV-6. Data for immunocompetent individuals are particularly scarce. Antiviral medication is usually not recommended for isolated meningitis.
For patients with CMV encephalitis, antiviral treatment with ganciclovir, foscarnet, or a combination of the two, is usually recommended [23, 109] (Table 3). Combination therapy has not been clearly shown to be superior to single treatment when studied in immunocompromised patients with generalized infection [124].
In EBV encephalitis, the benefit from using antivirals is probably low, but such treatment can be considered in serious cases since there are case reports of successful therapy with aciclovir or ganciclovir [125, 126]. Corticosteroids, with or without concurrent antiviral treatment, have been reported to be beneficial in EBV encephalitis, but there are no confirming systematic studies [127].
In vitro, HHV-6 is sensitive to ganciclovir, foscarnet and cidofovir. Clinical response has been shown in immunocompetent individuals treated with single or combination therapy with ganciclovir and foscarnet [128, 129]. In haematopoietic stem cell transplant recipients, a reduced HHV-6 viral load in CSF and blood has been shown [130]. The penetration of cidofovir into the CNS is poorly studied, and reports on its use in HHV-6 encephalitis show conflicting results [131, 132].
7 Tick-Borne Encephalitis
7.1 Clinical Features and Epidemiology
TBE is one of the most important zoonoses in Europe, with about 10,000 reported cases annually. In Europe, excluding Russia, hospitals care for 3,000 cases annually [7]. The clinical course and long-term prognosis has been studied in three prospective studies from Sweden, Germany and Lithuania [133–135].
TBE virus (TBEV) belongs to the Flaviviridae, and three main TBEV groups are discerned: the European or Western, the Siberian, and the Far Eastern subtype, formerly named Russian spring-summer encephalitis virus [136, 137]. TBE is endemic in Europe. The main vectors are the ticks Ixodes ricinus and persulcatus. Alimentary transmission via raw milk has also been reported.
An increase in the distribution area of TBEV and the number of TBE cases has been observed in many European countries in the past 10 years [7]. More men than women are affected. TBE occurs at all ages, with a peak incidence in the age group 40–60 years. Children are more seldom affected by severe disease; in a retrospective study 2 % of the patients were under 7 years old and 10 % under 15 years [138].
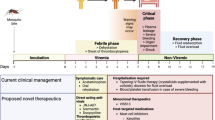
TBE is characterized by a biphasic course, seen in 70–90 % of cases [133, 134]. After an incubation period with a median of 8 days (4–28 days) [134], general signs of illness appear, such as headache, muscle ache, fatigue and fever, which persist for about 4 days (1–8 days). A short phase of viraemia with thrombocytopenia and granulocytopenia [133] is common. After a free interval with a median of 8 days (1–33 days), 20–30 % of those infected come down with signs of meningoencephalitis. Fever is obligatory, and blood leukocytosis is often seen in this second phase. A wide, continuous spectrum of neurological symptoms is seen, comprising meningitis, severe encephalitis and mixed syndromes, such as meningomyelitis and meningoencephalitis. About half of the cases are characterized by relatively severe diffuse encephalitis. The predominant symptoms of meningoencephalitis are ataxia (26 %), dysphasia, and cognitive dysfunction such as concentration and memory impairment (19 %), decreased consciousness, confusion (20 %), light and sound irritability (28 %) and tremor (9 %). Due to a TBEV preference for the anterior horn of the cervical spinal cord, a flaccid poliomyelitis-like paralysis can arise that, unlike poliomyelitis, usually affects the arms, shoulder and levator muscles of the head. In about 5–10 % of cases of European TBE, monoparesis, paraparesis and tetraparesis can develop, as well as paralysis of respiratory muscles, which requires ventilatory support [134]. Cranial nerve involvement is mainly associated with ocular, facial and pharyngeal motor function, but vestibular and hearing defects are also encountered. Seizures are rare. The elderly are found to suffer more severe disease [135]. The mortality rate in Europe is low (0.5–2 %) [7, 138].
Residual symptoms are seen in about 40–50 % of the patients at long-term follow-up. Cognitive dysfunctions and tremor show a tendency to increase during the early convalescence. One quarter of patients recover within 2 months. A second group has a prolonged course with cognitive defects and other residual neurological symptoms. A third group may develop myelitis and paralysis, which seems to occur without any direct connection with the initial severity of the disease [133]. Remaining palsy was found in 2.6–6 % of patients [135, 139].
7.2 Diagnostics Procedures
TBEV should be routinely included in the diagnosis of meningitis and meningoencephalitis in patients exposed in endemic areas during the tick season, regardless of known tick bite or not.
7.2.1 CSF Findings and Virological Analyses
The CSF pleocytosis is not as pronounced as in other viral meningoencephalitides. Initially, a predominance of polynuclear cells is observed, which after a few days is replaced by mononuclear cells [133].
Demonstration of TBEV by isolation or PCR is reported only in sporadic cases and cannot be used for routine diagnosis [140, 141]. Current infection is determined by detection of TBEV-specific IgM antibodies in serum together with specific IgG. IgM activity can be demonstrated in 96 % of patients with a median of 3 days after onset of encephalitis; later, serum from all patients is positive [20]. Maximum IgG activity can be detected in serum after 6 weeks and then decreases, but it persists for many years (>30 years). IgG should be analysed in paired serum samples. Intrathecal antibodies are seen in 97 % of patients after a median of 9 days [20] and the analysis may be of value in certain cases.
Cross-reaction with other flaviviruses occurs. It is therefore important to take account of any previous exposure to flavivirus and previous vaccinations. For immunity testing, the ELISA IgG is not a safe marker because of uncertainty about the lowest protective titres and cross-reaction with other flaviviruses.
In cases of suspected nonspecific IgG titres, neutralization (NT) may be performed to confirm the TBE diagnosis and immunity. NT requires the handling of infectious virus in a laboratory with biosafety level 3.
In TBE disease occurring in vaccinees [142], the characteristic kinetics consist in early detection of specific IgG with varying titres, low levels of neutralising antibodies to TBEV (NT), and a relatively late development of TBEV-specific IgM. Two to four weeks after the onset of encephalitis, new serum and CSF samples should be obtained for the detection of intrathecal antibody IgM and IgG activity and rising NT titres. Serum antibody responses may persist for a long time after natural infection or vaccination [142, 143].
7.2.2 Neuroradiology and Neurophysiology
Tissue destruction in the CNS is rare. Abnormalities on MRI are seen in up to 18 % of patients with TBE, with lesions confined to the thalamus, cerebellum, brainstem and nucleus caudatus [144, 145]. The EEG is abnormal in 77 % of patients [134]. Both MRI and EEG abnormalities are unspecific, not diagnostic, and no direct correlation with prognosis has been demonstrated.
7.3 Therapy and Prophylaxis
The treatment for TBE is currently symptomatic since no specific treatment is available. Antiviral therapy after the onset of encephalitis is of questionable value, since viral replication has most likely ceased when neurological symptoms appear. Anti-inflammatory therapy is a potential treatment, and further studies are necessary. The value of giving corticosteroids during TBE has not been convincingly demonstrated.
Previously, a specific immunoglobulin against TBE, post-exposure, was used, but this is not recommended due to doubts about the efficacy and even the risk of aggravated disease, which is supported by data from Germany [146]. There is no support for post-exposure vaccination after tick bites in endemic areas [147].
General vaccination is recommended in certain countries in Europe. In other countries, vaccination is recommended primarily for permanent and summer residents in endemic areas, as well as for travellers to specific risk areas [148].
Two vaccines against TBE are currently available. FSME-IMMUN® (Baxter™), and Encepur® (Novartis™). They are both whole virion vaccines and are produced by growing the virus from tick isolates in embryonal chicken fibroblasts. The adjuvant is aluminium hydroxide. The composition of the vaccines can be considered to be similar, but the stabilizer differs. Neither one of the vaccines contains thiomersal.
The adverse event profile is similar for both vaccines. The local side effects are usually mild and pass quickly. General malaise, body ache and headache are seen in approximately 10 %. Children tend to react more frequently with fever than adults. A transient post-vaccinal neuritis is reported in about 1/100,000 vaccinated individuals. A relative contraindication is hypersensitivity to egg white.
A protective effect of >95 % has been shown after three doses of FSME-IMMUN® [149], although no placebo-controlled study has been carried out. There is no support for significant differences between the vaccines. The protective effect can be expected from 14 days after the second dose.
Despite vaccination, TBE cases are observed after immunization with both the currently available vaccines [142, 143]. Individuals aged 50 years or above have a significantly lower antibody response [150], with a higher frequency of low responders and vaccine failures [151, 152]. Accordingly, rapid vaccination schedules [153–155] are considered to be less appropriate [156]. However, a recent study of the antibody avidity showed no difference between young or elderly individuals, and no correlation to the neutralization/ELISA ratio were found, suggesting that other factors affect the quality of the antibody response [157].
8 Enterovirus
8.1 Clinical Features and Epidemiology
Human enterovirus (HEV) infection is transmitted by the fecal–oral route, but drop and waterborne infection has also been documented. In the northern hemisphere, HEV disease is more common during summer and autumn, but sporadic cases and larger outbreaks can occur all year around [158]. In a given year, a certain enteroviral subtype may predominate and have an epidemic spread, whereas others may be more endemic.
Most cases of enteroviral infection are subclinical [159] or encompass acute febrile illness with upper respiratory tract symptoms, usually rather mild, but sometimes influenza-like. Vesicular lesions on the hands, feet and oral mucosa or herpangina as well as haemorrhagic conjunctivitis occur. The clinical presentation of enterovirus infection is influenced by age, rash being more common in small children. The name enterovirus is derived from the long carriage time in the intestine, although the virus rarely causes gastrointestinal symptoms.
After a couple of days of clinical amelioration, symptoms of meningitis may appear, with abrupt onset of headache, photophobia, nausea, vomiting and neck stiffness [159]. HEV is a predominant cause of aseptic meningitis and accounts for 30–75 % of all cases, or even more, depending on the season, age group and geographical region. In a family with an index case, all members are probably exposed, but >75 % of infections will lead to subclinical disease or mild upper respiratory symptoms. Thus, although secondary cases of viral meningitis are rare, small meningitis outbreaks have occasionally been reported. HEV CNS infection usually causes a benign and self-limiting disease [159]. However, encephalitis or meningoencephalitis may occur, with global symptoms such as confusion and altered consciousness, as well as seizures and focal neurological signs. Enterovirus is found in about 1 % of suspected encephalitis cases [10, 11]. The clinical course is often benign, and permanent sequelae are unusual [160]. A progressive and fatal course, with severe systemic disease, may occur in neonates, and chronic meningoencephalitis is seen in individuals with agammaglobulinaemia or patients undergoing immunosuppressive therapy [161].
Polio is a well known enterovirus on the verge of extinction, with now fewer than 1,000 yearly confirmed cases globally [162]. Subtype 2 is already eradicated. However, the eradication campaign with immunization has not fully succeeded, and subtypes 1 and 3 are endemic in Pakistan, Afghanistan and Nigeria, with rare imported cases to other countries.
While the importance of polio as a predominant myelitic agent has diminished in most countries, outbreaks of other neuropathogenic enterovirus strains, i.e. enterovirus 71 and other echo-and coxsackieviruses, are observed throughout the world. These strains may cause acute flaccid paralysis, brainstem-encephalitis mimicking paralytic poliomyelitis and cardiac complications [163, 164].
8.2 Diagnostic Procedures
8.2.1 CSF Findings and Virological Analysis
Usually 10–300 × 106 cells/L are found, with a predominance of polynuclear cells in the early stage but later with increasing numbers of mononuclear cells. However, it has been well reported that enterovirus might be present in the CSF without a cellular reaction. Molecular diagnostics using PCR for detection of HEV RNA is the method of choice for enteroviral CNS infection [165, 166]. Overall sensitivity is high, but the viral load in the early phase of the infection may not reach the threshold for detection. One week after the onset of neurological symptoms, the PCR signal usually disappears [167]. All subtypes, including polioviruses, are usually detected in test panels.
Several investigations have pointed out that PCR diagnostics may save costs by reducing hospital stay and avoiding unnecessary investigations [168–170]. Virus isolation, from CSF and feces, is mainly obsolete in the acute setting, but could be used for classification and for epidemiological purposes. Sequence analysis of HEV has recently led to considerable reclassification [171, 172].
Serology using IgM analysis for HEV is indicated primarily in PCR-negative cases, but is hampered by suboptimal specificity and sensitivity [173, 174]. Paired sera for IgG analysis can be used for diagnosis in a later stage.
8.2.2 Neuroradiology
In meningitis cases, radiological examinations are seldom necessary, but may be used to rule out other diseases. For encephalitis cases, MRI is an asset.
8.3 Therapy and Prophylaxis
No effective antiviral treatment can presently be offered for HEV infections. Pleconaril has been tried and a somewhat shortened duration of symptoms in meningitis was found, and symptoms seemed to be moderated in severe disease [175]. If made available, the compound could be an option in severe cases. No effect was shown in a small controlled study on pleconaril treatment in neonatal enteroviral sepsis syndrome [176].
Ribavirin appears to be effective in animal experiments, but clinical experience is lacking [177]. Immunoglobulin has been used in agammaglobulinaemic patients, neonates with HEV sepsis and meningitis, and in patients with severe enterovirus 71 disease [178–180]. Effective polio vaccines, oral and parenteral, have been available for more than 50 years. Clinical trials of enterovirus 71 vaccines are on-going [181].
9 Influenza Virus
9.1 Clinical Features and Epidemiology
All types of influenza viruses, capable of infecting humans, might also cause CNS complications. Influenza-associated acute encephalopathy/encephalitis (IAE) presents with high fever, altered consciousness and seizures, mainly documented in children. Acute necrotizing encephalopathy/encephalitis (ANE) has been reported in young children, mainly from Asian countries, since 1995 [182]. Outside South-East Asia, documentation of ANE is restricted to case reports [183–185].
In aetiological studies on encephalitis, seasonal influenza has been identified in 5–7 % of children [186, 187], in 1 % of both adults and children [10, 11], and in 1–7.4 % of adults [1, 27, 78]. Neurological complications of pandemic Influenza A H1N1/2009 in 69 children from Europe and the USA have recently been reviewed [188].
9.2 Diagnostic Procedures
9.2.1 CSF Findings and Virological Analyses
CSF analysis often reveals a lack of pleocytosis or merely a modest elevation of mononuclear leukocytes. The protein concentration and CSF/serum glucose rate are usually normal, although a slightly increased protein level may be present. A positive virus isolation from the CSF is rarely reported [189]. Influenza RNA may be demonstrated by reverse transcriptase (RT) PCR in cases with a rapid onset of CNS symptoms [190], but has not been detected in the majority of patients. The assumption that the encephalitis has been elicited by the influenza virus has, in most cases, been based on a temporal connection of the neurological symptoms and clinical influenza verified by nasopharyngeal virus isolation, antigen detection or PCR, or a significant serum antibody titre rise. The diagnostic value of intrathecally produced specific antibodies has not been evaluated.
9.2.2 Neuroradiology
Neuroimaging studies mostly review paediatric cases. CT or MRI abnormalities might be absent, especially early in the course. Later on, low attenuated and high signalling changes in, respectively, the cerebral cortex and the subcortical white matter may appear [191]. Reversible splenial lesions are associated with delirious symptoms [192]. In ANE, signs of multifocal symmetric lesions are prominent, often localized in the thalamus and brain stem with or without brain oedema [182]. Pathological findings on CT or MRI have been associated with a less favourable outcome [193].
9.3 Treatment and Prophylaxis
There are no randomized controlled trials on treatment of CNS complications in influenza. The emerging view of the pathogenesis of influenza encephalopathy is that the cytokine-mediated host response plays a key role. Genetic susceptibility, as well as metabolic disorders, might contribute to the pathogenesis [194]. The relative importance of a viral invasion of the CNS needs to be further clarified.
Antiviral drugs for influenza are directed against the receptor proteins: neuraminidase (NA) and membrane channel protein (M2). Early antiviral treatment, with NA inhibitors, such as oseltamivir or zanamivir, suppresses viral replication and prevents further stimulation of the host inflammatory response, thereby possibly reducing the risk of influenza complications in general [195]. However, a direct therapeutic effect on CNS manifestations is uncertain, since CSF concentrations of NA inhibitors have been shown to be low, although this has only been investigated in healthy volunteers [196]. The M2 inhibitors (amantadine and rimantadine) penetrate well into the CSF [197], but they are seldom used due to primary resistance or resistance development. Immunomodulatory treatments and hypothermia have been used experimentally [198]. Whether general influenza vaccination in adults diminishes CNS complications in influenza is unclear [199], but this has been suggested in Japanese children [200].
10 Measles, Mumps, Rubella
Since the introduction of highly effective combined measles, mumps, rubella vaccines in most countries, the observed incidence of CNS complications has markedly decreased.
10.1 Clinical Features and Epidemiology
Morbillivirus (measles virus) is spread by droplets from respiratory secretions or aerosol and is highly contagious. The clinical picture is characterized by cough, conjunctivitis, high fever and exanthema. Complications involve the respiratory tract and/or the CNS. Acute ‘post-infectious’ measles encephalitis, presumed to be autoimmune in nature and manifested 5–14 days after the onset of rash, is estimated to occur in 1/1,000–2,000 cases of measles in immunocompetent patients, primarily children. Decreased consciousness, fever, headache, seizures and focal neurological signs are common, as are neurological sequelae [201]. A few adult cases have been reported, suggesting that adults may suffer from more severe manifestations [202]. Progressive infectious encephalitis after the acute infection seriously affects immunocompromised individuals [203, 204]. A third entity, subacute sclerosing panencephalitis (SSPE), is a chronic, very rare complication appearing 2–24 years after primary measles infection, and inevitably leads to mental deterioration, seizures, coma and death [201, 205].
Parotitis (mumps) is a childhood disease, often with prominent clinical symptoms with tenderness and swelling of the salivary and parotid glands. However, up to one third of patients are asymptomatic. Epididymo-orchitis is a feared complication. Before the introduction of the vaccine, mumps was the most common cause of viral meningitis; 1–10 % of mumps cases showed signs of meningitis [206], usually emerging about 5 days after onset of disease, but sometimes preceding the mumps. The course is usually benign, but mumps meningitis can lead to unilateral deafness, described in 1/20,000 of mumps cases [206]. Encephalitis with seizures, sensorineural hearing loss and altered consciousness is a rare manifestation [207]. Other rare complications are facial palsy, ataxia, myelitis, polyradiculitis and Guillain–Barré syndrome [206].
Rubella infection most often causes a mild or subclinical disease characterized by fever, conjunctivitis, rash and lymphadenopathy. Acute encephalitis is rare (1/5,000–8,000) [208] and appears on average 4 days after an acute infection. The clinical signs are headache, disturbance of consciousness, convulsions, ataxia and focal neurological deficits. Survivors usually have no sequelae [201]. The progressive rubella panencephalitis (PRP) is an unusual neurodegenerative condition that appears 8–21 years after a congenital or prenatal infection [209].
10.2 Diagnostic Procedures
10.2.1 CSF Findings and Virological Analyses
In acute measles ‘post-infectious’ encephalitis, the CSF findings are normal or show pleocytosis 10–500 × 106 cells/L [201]. Measles may be diagnosed by virus isolation or RT-PCR of the CSF, serum, urine, nasopharyngeal aspirate or biopsy samples and a significant serological response [210]. Antibody detection in serum has a high sensitivity and specificity [211]. In the acute phase of encephalitis, specific intrathecal antibodies may be lacking, and are only reported in some few cases [202], while very high titres of antibodies in the CSF are detected in SSPE [212].
Most patients with mumps meningitis have pronounced CSF pleocytosis, and the lumbar puncture specimen often appears cloudy due to the high leukocyte content (1,000–2,000 × 106/L). Pleocytosis is even present in the CSF of half of mumps cases, disregarding signs of meningitis [213]. In mumps encephalitis, the cell count may be normal or elevated and the CSF/serum glucose ratio is normal or slightly depressed [207]. Virus isolation or PCR of saliva/throat, nasopharynx, blood, CSF, urine and sperm samples are diagnostic during the first week of infection [213]. CSF PCR, with its high sensitivity (70–96 %), is recommended for the diagnosis of meningitis. Serum IgM antibodies can be detected a week after onset of disease [206].
In the rare cases of rubella encephalitis, the lumbar puncture shows pleocytosis and elevated protein [214]. Rubella RNA can be detected by RT-PCR in nasopharyngeal, oral fluid, CSF and serum samples [215]. Serology with detection of IgM and seroconversion confirms a rubella infection [23], and intrathecal antibodies might be detected [214].
10.2.2 Neuroradiology and Neurophysiology
In acute ‘post-infectious’ encephalitis after measles, the MRI shows a picture similar to acute disseminated encephalomyelitis (ADEM), with generalized pathology in the white matter. EEG shows diffuse slow waves but does not reveal any distinctive pattern [210]. Half of the patients with measles without neurological symptoms will present with a pathological EEG recording efficacy [216].
MRI studies in mumps encephalitis are lacking [217]. EEG has shown generalized disturbances with slight to moderate slow activity, and in some cases lateralized signs [207].
In acute rubella encephalitis, radiological examinations of the brain are scarcely documented; normal images as well as brain oedema have been reported [214].
10.3 Therapy
Acute ‘post-infectious’ measles encephalitis, which is believed to be an autoimmune reaction [218], is treated symptomatically and supportively since randomized controlled treatment studies are lacking. Ribavirin has been used experimentally in patients with progressive infectious encephalitis [23]. A proven specific antiviral treatment is lacking for mumps and rubella encephalitis [23].
11 Neurorehabilitation
Follow-up after a severe viral CNS infection is mandatory and should include an appraisal of the need for neurorehabilitation. Neurological sequelae are common, despite efforts to start early medication for treatable viral CNS infections. Patients at high risk of developing neurological sequelae are primarily those with encephalitis, brain-stem encephalitis, meningoencephalitis or myelitis. However, patients with a clinical picture suggestive of meningitis in the acute phase may also develop cognitive deficits needing an assessment of the neuropsychological functions.
Neurological sequelae after viral CNS infections are scarcely documented in general, except for HSE and TBE. Recently, a large 3-year follow-up study of consecutive encephalitis cases in France was published [256]. After HSE, up to two thirds of the surviving patients are found to have neurological deficits [4–6, 32, 34–36]. Data on sequelae in VZV CNS disease are more scarce and the prognosis varies [8, 89, 219, 220, 257]. In TBE, residual symptoms are observed in 40–50 % of patients at long-term follow-up [20, 135, 139].
In neurorehabilitation, a multi-professional assessment of the patient’s neuropsychological status, leading to an individually adapted rehabilitation plan, is important. The goal is to reach the highest level of function and independence possible, with a focus on quality of life, often including the organization of additional practical support for the patient. The neurorehabilitation plan focuses on motor deficits and cognitive and communicative impairments and is aimed at facilitating the evolvement of strategies to handle brain tiredness. Neurorehabilitation is an expanding field in which new discoveries will be made regarding enriched environments, and physical, social and cultural activities, and pharmacological treatment aiming at stimulating neuroregeneration and neuroplasticity. As neurocognitive dysfunction is common after viral CNS infections, studies on structural tools for assessment of cognition in the follow-up are warranted.
12 Therapy
Today, antivirals are available for most herpesvirus CNS infections. However, randomized, controlled antiviral trials have only been conducted for HSE, where two independent trials in the 1980s showed superiority of aciclovir over vidarabine [32, 36], while such studies are lacking in other herpesvirus CNS infections. Aciclovir is also used against herpes meningitis and VZV-induced CNS infections, but controlled trials of these acute CNS infections have not been performed. Neurological sequelae after all herpesvirus infections are frequent, and improved treatment, antiviral as well as immunomodulatory, is warranted.
No specific antiviral treatments exist for the regionally prevalent encephalitic flaviviruses such as Japanese B encephalitis virus, TBEV, West Nile virus and Murray Valley fever virus, [23]. Effective antiviral treatment cannot presently be offered for enteroviral CNS infections, including polio.
Immunomodulating therapies are currently under investigation for West Nile encephalitis, but to our knowledge not for TBE, which is the predominate flavivirus encephalitis in Europe.
Effective vaccines, protective against CNS manifestations, are available for polio and TBE as well as for the childhood diseases measles, rubella and mumps. Childhood immunization against VZV introduced in several countries has markedly decreased the incidence of primary infections [105], and a reduction of CNS complications in conjunction with chickenpox will most likely be seen. In contrast, eventual protection of such vaccines against herpes zoster and its frequent neurological complications is under debate. However, VZV vaccine given at a high dose to the elderly decreases the zoster incidence [106], and the neurological morbidity is expected to be less common.
12.1 Aciclovir
Aciclovir is the drug of choice for HSV and VZV infections of the CNS and is often administered empirically to all patients with suspected viral encephalitis. Since bioavailability of oral aciclovir is poor, intravenous aciclovir is required to combat CNS infections. The pharmacokinetics of aciclovir in the brain and CSF has not been extensively examined.
Aciclovir is a guanosine analogue that is phosphorylated by viral thymidine kinase (TK) in infected cells, and, after further phosphorylations by human enzymes, is subsequently incorporated in the viral DNA chain, the elongation of which is then interrupted. The compound also possesses a direct inhibiting effect on viral DNA polymerase. Due to the constriction of drug distribution to virally infected cells, the adverse effects are few.
Although aciclovir shows antiviral activity in vitro against HSV, VZV, CMV, EBV and HHV-6 [221], the impact on the clinical course in CNS infections caused by the blood-borne herpesviruses, CMV, EBV and HHV-6, is currently unknown.
While the use of aciclovir in HSE patients is solidly based on randomized studies, the recommendation for treatment of severe VZV infections is less evidence based [222]. Administration in the early stage of herpes zoster has been shown to decrease the duration and severity of symptoms [223, 224]. Although the pathogenesis of neurological complications of VZV infections is not fully understood, a viral presence in the CNS is indicated by PCR data, and aciclovir administration aiming at inhibiting viral replication is advisable [225]. VZV is less sensitive than HSV for this drug, with a three times higher half maximal inhibitory concentration (IC50) value [226]; accordingly, higher doses of aciclovir might be needed.
Higher doses (intravenous aciclovir 15 mg/kg three times daily) are often administered to young patients with HSE without renal impairment, with the aim of achieving an adequate concentration in the focal inflammatory area of the brain parenchyma, although prospective dose response studies are lacking. It is speculated that a higher dose might be of benefit in adult HSE, in analogy with mortality reduction in neonatal disseminated HSV infection [227] seen with higher doses of aciclovir. However, a retrospective study of adult HSE could not confirm a clinical benefit of a higher dose [52].
Toxicity of aciclovir correlates with serum concentrations. Aciclovir is excreted by glomerular filtration and tubular secretion. A well known adverse effect of aciclovir is renal dysfunction, which is usually reversible. The risk of nephrotoxicity can be diminished by slow infusion, ensuring adequate hydration, and adjustment of the dosage regimen in renal dysfunction [228, 229]. Other adverse effects are rash, gastrointestinal symptoms and myelosuppression.
Potential neurotoxicity, with CNS function disturbances including agitation, hallucinations, disorientation, tremor and myoclonus has been highlighted, especially in patients with renal impairment. Analyses of aciclovir concentrations and those of its possibly neurotoxic metabolite, 9-carboxymethoxymethylguanine (CMMG), may be used [230] for diagnosing CNS toxicity and for guidance of dose modification. Consequently, proper attention to side effects is especially important in the elderly and in patients receiving potentially nephrotoxic drugs.
12.2 Valaciclovir
The l-valyl ester of aciclovir, valaciclovir, is an oral prodrug, and converts to aciclovir by hepatic and plasma esterases. The drug has 3–5 times greater bioavailability than oral aciclovir [231]. The compound has been scarcely studied in treatment and prophylaxis of viral CNS infections. Recently, CSF concentrations of aciclovir have been measured in patients with acute HSE treated with valaciclovir in resource-limited settings (Vietnam). Higher concentrations of aciclovir in the CSF were found after administration of valaciclovir 1 g three times daily in HSE [232], than in patients with multiple sclerosis [71], potentially reflecting the impaired and more permeable BBB in the early stage of HSE. Valaciclovir is well tolerated, with few reported adverse effects similar to those of aciclovir. Precautions similar to those for aciclovir regarding renal dysfunction and dose adjustment should be taken. The plasma aciclovir area under the concentration–time curve (AUC) values after administration of high-dose valaciclovir (2 g four times daily) are similar to those observed with intravenous aciclovir 10 mg/kg three times daily, but the peak concentrations are deemed to be less [231]. In severe CNS infections, treatment with oral valaciclovir in the acute stage cannot be recommended until controlled trials have shown satisfactory clinical effects on mortality and morbidity, equivalent to the effect of intravenous aciclovir treatment. In HSE, prolonged antiviral therapy with valaciclovir (2 g three times daily) after the intravenous therapy and its impact on mortality and sequelae is currently being evaluated in a large randomized, placebo-controlled, prospective, multicentre study by the Collaborative Antiviral Study Group of The National Institute of Allergy and Infectious Diseases [51].
In primary and recurrent HSV-2 meningitis, antiviral treatment in the acute phase has not been assessed in controlled trials. Nevertheless, antiviral treatment with 1 g of valaciclovir three times daily for 1 week is often used as acute phase treatment. Recently, a randomized, controlled trial did not provide support for suppression with valaciclovir 0.5 g twice daily in general after HSV-meningitis to avoid further meningitis episodes [65]. However, it cannot be ruled out that tailored suppressive treatment may be of benefit to some patients with frequent recurrences.
12.3 Ganciclovir
Ganciclovir is ten times more potent in vitro against CMV and EBV than aciclovir and is equally effective against HSV-1, HSV-2 and VZV [233]. Furthermore, an antiviral effect against HHV-6 has been reported [234]. The compound is, like aciclovir, a nucleoside analogue that undergoes triphosphorylation catalysed by UL97-encoded kinase and cellular kinases. The triphosphate inhibits viral DNA synthesis through competitive incorporation into the DNA, resulting in chain termination [235].
Ganciclovir treatment of viral CNS infections is almost exclusively described in immunocompromised patients [109] with varying efficacy [236]. In immunocompetent patients with severe CNS infections caused by CMV or HHV-6, intravenous ganciclovir is currently advised, but the benefit should always be weighed against the potential toxicity. The most common side effects are reversible myelodepression and nephrotoxicity [235]. Other adverse effects are rash, pruritus, gastrointestinal symptoms, and increased levels of liver enzymes. Neurotoxicity may occur occasionally.
12.4 Valganciclovir
Valganciclovir is the l-valyl ester prodrug and is rapidly hydrolyzed to ganciclovir. Pharmacokinetic studies in adults have shown that oral valganciclovir 900 mg results in similar serum concentrations as those after intravenous ganciclovir 5 mg/kg [237]. Treatment of CMV-retinitis in immunosuppressed individuals with valganciclovir appears to be as effective as intravenous ganciclovir treatment [238], but systematic studies have not been performed in treatment of CNS infections such as encephalitis or myelitis caused by this virus.
12.5 Foscarnet
Foscarnet suppresses replication of all herpesviruses [239] and is available for intravenous use only. The compound is a non-nucleoside pyrophosphate analogue that directly inhibits the viral DNA polymerase, thus maintaining activity against herpesviruses with TK or UL97 kinase mutations [240].
The use of foscarnet is limited by its relatively high level of toxicity. Surveillance of mineral and electrolyte homeostasis as well as kidney function is compulsory. Hyperhydration during the treatment may prevent the tubular toxicity [239].
In immunocompetent patients, use of foscarnet may be considered in those with aciclovir- or ganciclovir-resistant infections caused by herpesviruses, or in cases of allergy or intolerance to ganciclovir. In immunosuppressed patients with CMV infection of the CNS, addition of, or alteration with, foscarnet can be suggested if single therapy with ganciclovir is non-effective [241].
12.6 Cidofovir
Cidofovir is a nucleoside analogue with effect on herpesviruses in vitro. The drug is poorly investigated as regards effects in the treatment of CNS infections, [131, 132] and pharmacological data on penetration to the CSF or brain is lacking.
12.7 Pleconaril
Pleconaril exerts its antiviral effect on picorna viruses by inhibiting attachment of the virion to the cell as well as the uncoating of viral RNA, and thus interrupts the infection cycle [242]. Clinical studies of patients suffering from meningitis caused by enteroviruses showed a slightly shortened duration of symptoms after pleconaril treatment, and the clinical course of severe disease seemed to be moderated [175]. If made available, the compound could be a treatment option in severe cases. No effect was shown in a small controlled study on pleconaril treatment in neonatal enteroviral sepsis syndrome [176]. New antivirals are needed for the treatment of severe enterovirus infections.
12.8 Neuraminidase Inhibitors
Oseltamivir phosphate is a prodrug of oseltamivir carboxylate, which selectively blocks viral NA, an enzyme that is essential in the replication of influenza A and B viruses. Another NA inhibitor, zanamivir, is inhaled and has been used intravenously in critically ill patients, while oseltamivir is administered orally. Two new NA inhibitors, laninamivir and peramivir, are under investigation.
Although NA inhibitors have demonstrated beneficial effects in the form of reduced durations of clinical symptoms during influenza infection, the efficacy of antiviral treatment for CNS manifestations of this virus has not been well documented. CSF concentrations of NA inhibitors have been determined in healthy volunteers and shown to be low [196]. The most commonly reported adverse effects of oseltamivir are gastrointestinal symptoms, and insomnia. Furthermore, vertigo and neuropsychiatric adverse effects were mainly reported from Japan [243]. The M2 inhibitors (amantadine and rimantadine) penetrate well into the CSF [197], but are not used in clinical practice due to primary viral resistance, or to resistance developing during treatment.
12.9 Aciclovir Resistance
Despite the use of antivirals such as aciclovir for several decades, viral resistance has hitherto not been a serious issue for immunocompetent individuals. Primary resistance is uncommon, and induced resistance develops mainly in immunosuppressed individuals with high viral loads and antiviral treatment of long duration. However, with the increased number of patients receiving immunosuppressive treatments due to organ transplantation, autoimmune diseases or malignancies, drug-resistant viral infections are no longer rare events.
Aciclovir resistance is mainly associated with mutations in the TK coding region of the genomes of herpes simplex [244, 245] and VZV [246]. In large population studies of mucocutaneous herpes, only 0.3 % of HSV strains isolated from immunocompetent hosts were aciclovir resistant [247]. HSV-1 strains deficient of TK have been demonstrated to be less neurovirulent [248]. However, an immunocompetent HSE patient with a suspected primary aciclovir-resistant virus has recently been described, [249] and resistance has also been reported in neonatal herpes [250]. Recently, compartmentalization of aciclovir-resistant VZV, in stem cell transplant recipients, has been described with resistant virus/mutant sequences in the CSF, while wild-virus was found in plasma samples [251].
12.10 Immunomodulating Therapy
Immunomodulating therapy is discussed for several viral CNS infections, but randomized studies are lacking. The extent to which the brain cell damage is due to direct viral cytolysis or mediated by the host-immune response during the different stages of infection is insufficient elucidated. The use of corticosteroids in viral CNS infections are scarcely studied, although the brain damage is believed to depend to a varying degree, on the intrathecal host immune response.
In HSE, in which the pathogenesis includes a cytolytic infection of neurons, a vigorous acute inflammatory response, as well as long-term persistence of intrathecal cellular and humoral immune activation, is demonstrated [252, 253]. Studies on supplementary steroid treatment in experimental HSE are scarce [254, 255], and only one study in humans has been performed [53] and further studies are warranted [54]. In CNS vasculopathy caused by VZV infection, corticosteroids may be used in conjunction with antivirals [98].
In clinical practice, steroids are used as anti-oedema therapy in cases of viral encephalitis with a high ICP, and in patients with viral myelitis. A short duration of the treatment, such as 3–5 days, is generally recommended to avoid adverse effects.
In influenza and TBE encephalitis the inflammatory component in the pathogenesis is believed to play a predominant role [7, 194]. Specific immunomodulatory therapies for neuro-inflammation are currently being developed and under investigation for treatment. If shown to be effective and safe, such drugs might be translated into clinical practice.
13 Conclusion
Virological diagnostic procedures for CNS infections, including PCR and serology, have been substantially improved during the recent years. The choice of diagnostics must be adapted to the current epidemiological situation and the individual patient history, including recent travel. Furthermore, the virological results should be interpreted based on knowledge of the different possibilities and limitations of the methods used. Continuous epidemiological surveillance is necessary for establishing effective preventive strategies and for the detection of emerging viruses. Despite the recent advancements of diagnostics during the last decades and the early administration of antiviral treatment, neurological sequelae are still frequent in viral CNS infections. Further advances in both antiviral and immunomodulating treatment are awaited. However, while controlled treatment trials of viral CNS infections are difficult to conduct due to the relative paucity of cases and need for multicentre cooperation, the advances may possibly rather be expected within the field of viral vaccines.
References
Rantalaiho T, Färkkilä M, Vaheri A, et al. Acute encephalitis from 1967 to 1991. J Neurol Sci. 2001;184(2):169–77.
Davison KL, Crowcroft NS, Ramsay ME, et al. Viral encephalitis in England, 1989–1998: what did we miss? Emerg Infect Dis. 2003;9(2):234–40.
Kupila L, Vuorinen T, Vainionpää R, et al. Etiology of aseptic meningitis and encephalitis in an adult population. Neurology. 2006;66(1):75–80.
Raschilas F, Wolff M, Delatour F, et al. Outcome of and prognostic factors for herpes simplex encephalitis in adult patients: results of a multicenter study. Clin Infect Dis. 2002;35(3):254–60.
McGrath N, Anderson NE, Croxson MC, et al. Herpes simplex encephalitis treated with acyclovir: diagnosis and long term outcome. J Neurol Neurosurg Psychiatry. 1997;63(3):321–6.
Domingues RB, Tsanaclis AM, Pannuti CS, et al. Evaluation of the range of clinical presentations of herpes simplex encephalitis by using polymerase chain reaction assay of cerebrospinal fluid samples. Clin Infect Dis. 1997;25(1):86–91.
Lindquist L, Vapalahti O. Tick-borne encephalitis. Lancet. 2008;371(9627):1861–71.
Persson A, Bergström T, Lindh M, et al. Varicella zoster virus CNS disease: viral load, clinical manifestations and sequels. J Clin Virol. 2009;46(3):249–53.
Franzen-Röhl E, Tiveljung-Lindell A, Grillner L, et al. Increased detection rate in diagnosis of herpes simplex virus type 2 meningitis by real-time PCR using cerebrospinal fluid samples. J Clin Microbiol. 2007;45(8):2516–20.
Mailles A, Stahl JP. Infectious encephalitis in France in 2007: a national prospective study. Clin Infect Dis. 2009;49(12):1838–47.
Graneröd J, Ambrose HE, Davies NW, et al. Causes of encephalitis and differences in their clinical presentations in England: a multicentre, population-based prospective study. Lancet Infect Dis. 2010;10(12):835–44.
Franzen-Röhl E, Larsson K, Skoog E, et al. High diagnostic yield by CSF-PCR for entero- and herpes simplex viruses and TBEV serology in adults with acute aseptic meningitis in Stockholm. Scand J Infect Dis. 2008;40(11–12):914–21.
Studahl M, Hagberg, Rekabdar E, et al. Hematogenously spread herpesviruses are detected as frequently as neuronally spread herpesviruses in cerebrospinal fluid by polymerase chain reaction assay. Clin Infect Dis. 1999;29(1):216–8.
Weinberg A, Bloch KC, Li S, Tang YW, Palmer M, Tyler KL. Dual infections of the central nervous system with Epstein–Barr virus. J Infect Dis. 2005;191(2):234–7.
Kleines M, Schiefer J, Stienen A, et al. Expanding the spectrum of neurological disease associated with Epstein–Barr virus activity. Eur J Clin Microbiol Infect Dis. 2011;30(12):1561–9.
Martelius T, Lappalainen M, Palomäki M, et al. Clinical characteristics of patients with Epstein–Barr virus in cerebrospinal fluid. BMC Infect Dis. 2011;11:281.
Leong HN, Tuke PW, Tedder RS, et al. The prevalence of chromosomally integrated human herpesvirus 6 genomes in the blood of UK blood donors. J Med Virol. 2007;79(1):45–51.
Ward KN, Leong HN, Thiruchelvam AD, et al. Human herpesvirus 6 DNA levels in cerebrospinal fluid due to primary infection differ from those due to chromosomal viral integration and have implications for diagnosis of encephalitis. J Clin Microbiol. 2007;45(4):1298–304.
Forsgren M, Sköldenberg B, Jeansson S, et al. Serodiagnosis of herpes encephalitis by indirect enzyme-linked immunosorbent assay, experience from a Swedish trial. Serodiagn Immunother Infect Dis. 1989;3(4):259–71.
Gunther G, Haglund M, Lindquist L, et al. Intrathecal IgM, IgA and IgG antibody response in tick-borne encephalitis: long-term follow-up related to clinical course and outcome. Clin Diagn Virol. 1997;8(1):17–29.
Gregoire SM, van Pesch V, Goffette S, et al. Polymerase chain reaction analysis and oligoclonal antibody in the cerebrospinal fluid from 34 patients with varicella-zoster virus infection of the nervous system. J Neurol Neurosurg Psychiatry. 2006;77(8):938–42.
Solomon T, Michael BD, Smith PE, Sanderson F, Davies NW, Hart IJ, et al. Management of suspected viral encephalitis in adults. Association of British Neurologists and British Infection Association National Guidelines. J Infect. 2012;64(4):347–73.
Tunkel AR, Glaser CA, Bloch KC, et al. The management of encephalitis: clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis. 2008;47(3):303–27.
Aurelius E, Johansson B, Sköldenberg B, et al. Rapid diagnosis of herpes simplex encephalitis by nested polymerase chain reaction assay of cerebrospinal fluid. Lancet. 1991;337(8735):189–92.
Aurelius E, Johansson B, Sköldenberg B, et al. Encephalitis in immunocompetent patients due to herpes simplex virus type 1 or 2 as determined by type-specific polymerase chain reaction and antibody assays of cerebrospinal fluid. J Med Virol. 1993;39(3):179–86.
Guffond T, Dewilde A, Lopert PE, et al. Significance and clinical relevance of the detection of herpes simplex virus DNA by the polymerase chain reaction in cerebrospinal fluid from patients with presumed encephalitis. Clin Infect Dis. 1994;18(5):744–9.
Studahl M, Bergström T, Hagberg L. Acute viral encephalitis in adults: a prospective study. Scand J Infect Dis. 1998;30(3):215–20.
Puchhammer-Stöckl E, Presterl E, Aberle S, et al. Screening for possible failure of herpes simplex virus PCR in cerebrospinal fluid for the diagnosis of herpes simplex encephalitis. J Med Virol. 2001;64(4):531–6.
Weil AA, Glaser CA, Amad Z, et al. Patients with suspected herpes simplex encephalitis: rethinking an initial negative polymerase chain reaction result. Clin Infect Dis. 2002;34(8):1154–7.
Wald A, Zeh J, Selke S, et al. Reactivation of genital herpes simplex virus type 2 infection in asymptomatic seropositive persons. N Engl J Med. 2000;342(12):844–50.
Kennedy PG. A retrospective analysis of forty-six cases of herpes simplex encephalitis seen in Glasgow between 1962 and 1985. Q J Med. 1988;68(255):533–40.
Sköldenberg B, Forsgren M, Alestig K, et al. Acyclovir versus vidarabine in herpes simplex encephalitis: randomised multicentre study in consecutive Swedish patients. Lancet. 1984;2(8405):707–11.
Whitley RJ, Soong SJ, Linneman C, et al. Herpes simplex encephalitis: clinical assessment. JAMA. 1982;247(3):317–20.
Utley TFM, Ogden JA, Gibb A, McGrath N, Anderson NE. The long-term neuro-psychological outcome of herpes simplex encephalitis in a series of unselected survivors. Neuropsychiatry Neuropsychol Behav Neurol. 1997;10:180–9.
Gordon B, Selnes OA, Hart J Jr, Hanley DF, Whitley RJ. Long-term cognitive sequelae of acyclovir-treated herpes simplex encephalitis. Arch Neurol. 1990;47:646–7.
Whitley RJ, Alford CA, Hirsch MS, et al. Vidarabine versus acyclovir therapy in herpes simplex encephalitis. N Engl J Med. 1986;314(3):144–9.
Hjalmarsson A, Blomqvist P, Sköldenberg B. Herpes simplex encephalitis in Sweden, 1990–2001: incidence, morbidity, and mortality. Clin Infect Dis. 2007;45(7):875–80.
Dennett C, Cleator GM, Klapper PE. HSV-1 and HSV-2 in herpes simplex encephalitis: a study of sixty-four cases in the United Kingdom. J Med Virol. 1997;53(1):1–3.
Vandvik B, Sköldenberg B, Forsgren M, et al. Long-term persistence of intrathecal virus-specific antibody responses after herpes simplex virus encephalitis. J Neurol. 1985;231(6):307–12.
Domingues RB, Fink MC, Tsanaclis AM, et al. Diagnosis of herpes simplex encephalitis by magnetic resonance imaging and polymerase chain reaction assay of cerebrospinal fluid. J Neurol Sci. 1998;157(2):148–53.
Schlesinger Y, Buller RS, Brunstrom JE, et al. Expanded spectrum of herpes simplex encephalitis in childhood. J Pediatr. 1995;126(2):234–41.
Schmidbauer M, Podreka I, Winberger D, et al. SPECT and MR imaging in herpes simplex encephalitis. J Comput Assist Tomogr. 1991;15(5):811–5.
Schroth G, Gawehn J, Thron A, et al. Early diagnosis of herpes simplex encephalitis by MRI. Neurology. 1987;37(2):179–83.
Hindmarsh T, Lindqvist M, Olding-Stenkvist E, et al. Accuracy of computed tomography in the diagnosis of herpes simplex encephalitis. Acta Radiol Suppl. 1986;369:192–6.
Bergey GK, Coyle PK, Krumholz A, et al. Herpes simplex encephalitis with occipital localization. Arch Neurol. 1982;39(5):312–3.
Studahl M, Sköldenberg B. Herpes simplex encephalitis and other neurological syndromes caused by herpes simplex virus-1. In: Studahl M, Cinque P, Bergström T, editors. Herpes simplex viruses. New York: Taylor & Francis; 2006. p. 275–316.
Brodtkorb E, Lindqvist M, Jonsson M, et al. Diagnosis of herpes simplex encephalitis: a comparison between electro-encephalography and computed tomography findings. Acta Neurol Scand. 1982;66(4):462–71.
Towne AR, Waterhouse EJ, Boggs JG, et al. Prevalence of nonconvulsive status epilepticus in comatose patients. Neurology. 2000;54(2):340–5.
Schloss L, Falk KI, Skoog E, et al. Monitoring of herpes simplex virus DNA types 1 and 2 viral load in cerebrospinal fluid by real-time PCR in patients with herpes simplex encephalitis. J Med Virol. 2009;81(8):1432–7.
Cinque P, Cleator GM, Weber T, et al. The role of laboratory investigation in the diagnosis and management of patients with suspected herpes simplex encephalitis: a consensus report. The EU Concerted Action on Virus Meningitis and Encephalitis. J Neurol Neurosurg Psychiatry. 1996;61(4):339–45.
National Institute of Allergy and Infectious Diseases (NIAID). Long term treatment of herpes simplex encephalitis (HSE) with valacyclovir [Clinicaltrials.gov identifier NCT00031486]. US National Institutes of Health, ClinicalTrials.gov. http://www.clinicaltrials.gov. Accessed 2 May 2012.
Stahl JP, Mailles A, De Broucker T, et al. Herpes simplex encephalitis and management of acyclovir in encephalitis patients in France. Epidemiol Infect. 2012;140(2):372–81.
Kamei S, Sekizawa T, Shiota H, et al. Evaluation of combination therapy using aciclovir and corticosteroid in adult patients with herpes simplex virus encephalitis. J Neurol Neurosurg Psychiatry. 2005;76(11):1544–9.
Martinez-Torres F, et al. Protocol for German trial of Acyclovir and corticosteroids in Herpes-simplex-virus-encephalitis (GACHE): a multicenter, multinational, randomized, double-blind, placebo-controlled German, Austrian and Dutch trial [ISRCTN45122933]. BMC Neurol. 2008;29(8):40.
Sköldenberg B, Aurelius E, Hjalmarsson A, et al. Incidence and pathogenesis of clinical relapse after herpes simplex encephalitis in adults. J Neurol. 2006;253(2):163–70.
Tedder DG, Ashley R, Tyler KL, et al. Herpes simplex virus infection as a cause of benign recurrent lymphocytic meningitis. Ann Intern Med. 1994;121(5):334–8.
Bergström T, Vahlne A, Alestig K, et al. Primary and recurrent herpes simplex virus type 2-induced meningitis. J Infect Dis. 1990;162(2):322–30.
O’Sullivan CE, Aksamit AJ, Harrington JR, et al. Clinical spectrum and laboratory characteristics associated with detection of herpes simplex virus DNA in cerebrospinal fluid. Mayo Clin Proc. 2003;78(11):1347–52.
Sköldenberg B, Jeansson S, Wolontis S. Herpes simplex virus type 2 and acute aseptic meningitis: clinical features of cases with isolation of herpes simplex virus from cerebrospinal fluids. Scand J Infect Dis. 1975;7(4):227–32.
Corey L, Adams HG, Brown ZA, et al. Genital herpes simplex virus infections: clinical manifestations, course, and complications. Ann Intern Med. 1983;98(6):958–72.
Aurelius E, Forsgren M, Gille E, et al. Neurologic morbidity after herpes simplex virus type 2 meningitis: a retrospective study of 40 patients. Scand J Infect Dis. 2002;34(4):278–83.
Terni M, Caccialanza P, Cassai E, et al. Aseptic meningitis in association with herpes progenitalis. N Engl J Med. 1971;285(9):503–4.
Bachmeyer C, de la Blanchardière A, Lepercq J, et al. Recurring episodes of meningitis (Mollaret’s meningitis) with one showing an association with herpes simplex virus type 2. J Infect. 1996;32(3):247–8.
Kallio-Laine K, Seppänen M, Kautiainen H, et al. Recurrent lymphocytic meningitis positive for herpes simplex virus type 2. Emerg Infect Dis. 2009;15(7):1119–22.
Aurelius E, Franzen-Röhl E, Glimåker M, et al. Long-term valacyclovir suppressive treatment after herpes simplex virus type 2 meningitis: a double-blind, randomised, controlled trial. Clin Infect Dis. 2012;54(9):1304–13.
Read S, Kurtz JB. Laboratory diagnosis of common viral infections of the central nervous system by using a single multiplex PCR screening assay. J Clin Microbiol. 1999;37(5):1352–5.
Brenton DW. Hypoglycorrhachia in herpes simplex type 2 meningitis. Arch Neurol. 1980;37(5):317.
Sköldenberg B, Jeansson S, Wolontis S. Herpes simplex virus type 2 in acute aseptic meningitis. Br Med J. 1973;2(5866):611.
Kupila L, Vainionpää R, Vuorinen T, et al. Recurrent lymphocytic meningitis: the role of herpesviruses. Arch Neurol. 2004;61(10):1553–7.
Aurelius E. Neurological disease in herpes simplex virus type 2 (HSV-2) infection. In: Studahl M, Cinque P, Bergström T, editors. Herpes simplex viruses. New York: Taylor & Francis; 2006. p. 317–38.
Lycke J, Malmeström C, Ståhle L. Acyclovir levels in serum and cerebrospinal fluid after oral administration of valacyclovir. Antimicrob Agents Chemother. 2003;47(8):2438–41.
Bergström T, Alestig K. Treatment of primary and recurrent herpes simplex virus type 2 induced meningitis with acyclovir. Scand J Infect Dis. 1990;22(2):239–40.
Berger JR. Benign aseptic (Mollaret’s) meningitis after genital herpes. Lancet. 1991;115(1):19–21.
Koskiniemi M, Piiparinen H, Rantalaiho T, et al. Acute central nervous system complications in varicella zoster virus infections. J Clin Virol. 2002;25(3):293–301.
Ziebold C, von Kries R, Lang R, et al. Severe complications of varicella in previously healthy children in Germany: a 1-year survey. Pediatrics. 2001;108(5):1–6.
Frenos S, Galli L, Chiappini E, et al. An increasing incidence of chickenpox central nervous system complications in children: what’s happening in Tuscany? J Clin Virol. 2007;38(4):358–61.
Rack AL, Grote V, Streng A, et al. Neurologic varicella complications before routine immunization in Germany. Pediatr Neurol. 2010;42(1):40–8.
Glaser CA, Gilliam S, Schnurr D, et al. In search of encephalitis etiologies: diagnostic challenges in the California Encephalitis Project, 1998–2000. Clin Infect Dis. 2003;36(6):731–42.
Guess HA, Broughton DD, Melton LJ 3rd, et al. Population-based studies of varicella complications. Pediatrics. 1986;78(4Pt2):723–7.
Connolly AM, Dodson E, Prensky AL, et al. Course and outcome of acute cerebellar ataxia. Ann Neurol. 1994;35(6):673–9.
Sweeney CJ, Gilden DH. Ramsay Hunt syndrome. J Neurol Neurosurg Psychiatry. 2001;71(2):149–54.
Gilden D, Cohrs RJ, Mahalingam R, et al. Varicella zoster virus vasculopathies: diverse clinical manifestations, laboratory features, pathogenesis, and treatment. Lancet Neurol. 2009;8(8):731–40.
Ciccone S, Faggioli R, Calzolari F, et al. Stroke after varicella-zoster infection: report of a case and review of the literature. Pediatr Infect Dis J. 2010;29(9):864–7.
Sebire G, Meyer L, Chabrier S. Varicella as a risk factor for cerebral infarction in childhood: a case–control study. Ann Neurol. 1999;45(5):679–80.
Nagel MA, Cohrs RJ, Mahalingam R, et al. The varicella zoster virus vasculopathies: clinical, CSF, imaging, and virologic features. Neurology. 2008;70(11):853–60.
Hattori H, Higuchi Y, Tsuji M. Recurrent strokes after varicella. Ann Neurol. 2000;47(1):136.
Berger TM, Caduff JH, Gebbers JO. Fatal varicella-zoster antigen-positive giant cell arteritis of the central nervous system. Pediatr Infect Dis J. 2000;19(7):653–6.
Okanishi T, Kondo A, Inoue T, Maegaki Y, Ohno K, Togari H. Bilateral middle cerebral artery infarctions following mild varicella infection: a case report. Brain Dev. 2009;31(1):86–9.
Hokkanen L, Launes J, Poutiainen E, et al. Subcortical type cognitive impairment in herpes zoster encephalitis. J Neurol. 1997;244(4):239–45.
Aberle SW, Aberle JH, Steininger C, et al. Quantitative real time PCR detection of varicella zoster virus DNA in cerebrospinal fluid in patients with neurological disease. Med Microbiol Immunol. 2005;194(1–2):7–12.
Mathiesen T, Linde A, Olding-Stenkvist E, et al. Antiviral IgM and IgG subclasses in varicella zoster associated neurological syndromes. J Neurol Neurosurg Psychiatry. 1989;52(5):578–82.
Grahn A, Studahl M, Nilsson S, et al. Varicella-zoster virus (VZV) glycoprotein E is a serological antigen for detection of intrathecal antibodies to VZV in central nervous system infections, without cross-reaction to herpes simplex virus 1. Clin Vaccine Immunol. 2011;18(8):1336–42.
Pahud BA, Glaser CA, Dekker CL, et al. Varicella zoster disease of the central nervous system: epidemiological, clinical, and laboratory features 10 years after the introduction of the varicella vaccine. J Infect Dis. 2011;203(3):316–23.
Haanpää M, Dastidar P, Weinberg A, et al. CSF and MRI findings in patients with acute herpes zoster. Neurology. 1998;51(5):1405–11.
Tien RD, Felsberg GJ, Osumi AK. Herpesvirus infections of the CNS: MR findings. Am J Roentgenol. 1993;161(1):167–76.
Küker W. Cerebral vasculitis: imaging signs revisited. Neuroradiology. 2007;49(6):471–9.
Gnann JW, Whitley RJ. Clinical practice. Herpes zoster. N Engl J Med. 2002;347(5):340–6.
Gilden D. Varicella zoster virus and central nervous system syndromes. Herpes. 2004;11(Suppl 2):89A–94A.
Devriese PP, Moesker WH. The natural history of facial paralysis in herpes zoster. Clin Otolaryngol Allied Sci. 1988;13(4):289–98.
Kinishi M, Amatsu M, Mohri M, et al. Acyclovir improves recovery rate of facial nerve palsy in Ramsay Hunt syndrome. Auris Nasus Larynx. 2001;28(3):223–6.
Furuta Y, Othani F, Mesuda Y, et al. Early diagnosis of zoster sine herpete and antiviral therapy for the treatment of facial palsy. Neurology. 2000;55(5):708–10.
Murakami S, Hato N, Horiuchi J, et al. Treatment of Ramsay Hunt syndrome with acyclovir-prednisone: significance of early diagnosis and treatment. Ann Neurol. 1997;41(3):353–7.
Uscategui T, Doree C, Chamberlain IJ, et al. Antiviral therapy for Ramsay Hunt syndrome (herpes zoster oticus with facial palsy) in adults. Cochrane Database Syst Rev. 2008;8(4):CD006851.
Uscategui T, Doree C, Chamberlain IJ, et al. Corticosteroids as adjuvant to antiviral treatment in Ramsay Hunt syndrome (herpes zoster oticus with facial palsy) in adults. Cochrane Database Syst Rev. 2008;16(3):CD006852.
Seward JF, Watson BM, Peterson CL, et al. Varicella disease after introduction of varicella vaccine in the United States, 1995–2000. JAMA. 2002;287(5):606–11.
Oxman MN, Levin MJ, Johnson GR, et al. A vaccine to prevent herpes zoster and postherpetic neuralgia in older adults. N Engl J Med. 2005;352(22):2271–84.
Rafailidis PI, Mourtzoukou EG, Varbobitis IC, et al. Severe cytomegalovirus infection in apparently immuncompetent patients: a systematic review. Virol J. 2008;5:47.
Arribas JR, Storch GA, Clifford DB, et al. Cytomegalovirus encephalitis. Ann Intern Med. 1996;125(7):577–87.
Griffiths P. Cytomegalovirus infection of the central nervous system. Herpes. 2004;11(Suppl 2):95A–104A.
Koskiniemi M, Rantalaiho T, Piiparinen H, et al. Infections of the central nervous system of suspected viral origin: a collaborative study from Finland. J Neurovirol. 2001;7(5):400–8.
Yao K, Honarmand S, Espinosa A, et al. Detection of human herpesvirus-6 in cerebrospinal fluid of patients with encephalitis. Ann Neurol. 2009;65(3):257–67.
Ward KN, Leong HN, Nacheva EP, et al. Human herpes virus 6 chromosomal integration in immunocompetent patients results in high levels of viral DNA in blood, sera, and hair follicles. J Clin Microbiol. 2006;44(4):1571–4.
Volpi A. Epstein-Barr virus and human herpesvirus type 8 infections of the central nervous system. Herpes. 2004;11(Suppl 2):120A–7A.
Caserta MT, Hall CB, Schnabel K, et al. Neuroinvasion and persistence of human herpesvirus 6 in children. J Infect Dis. 1994;170(6):1586–9.
Hall CB, Long CE, Schnabel KC, et al. Human herpesvirus-6 infection in children: a prospective study of complications and reactivation. N Engl J Med. 1994;331(7):432–8.
Ljungman P, Singh N. Human herpesvirus-6 infection in solid organ and stem cell transplant recipients. J Clin Virol. 2006;37(Suppl. 1):S87–91.
McCullers JA, Lakeman FD, Whitley RJ. Human herpesvirus 6 is associated with focal encephalitis. Clin Infect Dis. 1995;21(3):571–6.
Studahl M, Hagberg L, Rekabdar E, et al. Hematogenously spread herpesviruses are detected as frequently as neuronally spread herpesviruses in cerebrospinal fluid by polymerase chain reaction assay. Clin Infect Dis. 1999;29(1):216–8.
Davies NW, Brown LJ, Gonde J, et al. Factors influencing PCR detection of viruses in cerebrospinal fluid of patients with suspected CNS infections. J Neurol Neurosurg Psychiatry. 2005;76(1):82–7.
Weber T, Beck R, Stark E, et al. Comparative analysis of intrathecal antibody synthesis and DNA amplification for the diagnosis of cytomegalovirus infection of the central nervous system in AIDS patients. J Neurol. 1994;241(7):407–14.
Majid A, Galetta SL, Sweeney CJ, et al. Epstein–Barr virus myeloradiculitis and encephalomyeloradiculitis. Brain. 2002;125(Pt 1):159–65.
Patnaik M, Peter JB. Intrathecal synthesis of antibodies to human herpesvirus 6 early antigen in patients with meningitis/encephalitis. Clin Infect Dis. 1995;21(3):715–6.
Abul-Kasim K, Palm L, Maly P, et al. The neuroanatomic localization of Epstein–Barr virus encephalitis may be a predictive factor for its clinical outcome: a case report and review of 100 cases in 28 reports. J Child Neurol. 2009;24(6):720–6.
Mattes FM, Hainsworth EG, Geretti AM, et al. A randomized, controlled trial comparing ganciclovir to ganciclovir plus foscarnet (each at half dose) for preemptive therapy of cytomegalovirus infection in transplant recipients. J Infect Dis. 2004;189(8):1355–61.
Demey HE, Martin JJ, Leus RM, et al. Coma as a presenting sign of Epstein–Barr encephalitis. Arch Intern Med. 1988;148(6):1459–61.
Dellemijn PL, Brandenburg A, Niesters HG, et al. Successful treatment with ganciclovir of presumed Epstein–Barr meningo-encephalitis following bone marrow transplant. Bone Marrow Transplant. 1995;16(2):311–2.
Straus SE, Cohen JI, Tosato G, et al. NIH conference. Epstein–Barr virus infections: biology, pathogenesis and management. Ann Intern Med. 1993;118(1):45–58.
Troy SB, Blackburn BG, Yeom K, et al. Severe encephalomyelitis in an immunocompetent adult with chromosomally integrated human herpesvirus 6 and clinical response to treatment with foscarnet plus ganciclovir. Clin Infect Dis. 2008;47(12):e93–6.
Birnbaum T, Padovan CS, Sporer B, et al. Severe meningoencephalitis caused by human herpesvirus 6 type B in an immunocompetent woman treated with ganciclovir. Clin Infect Dis. 2005;40(6):887–9.
Zerr DM, Gupta D, Huang ML, et al. Effect of antivirals on human herpesvirus 6 replication in hematopoietic stem cell transplant recipients. Clin Infect Dis. 2002;34(3):309–17.
Pöhlmann C, Schetelig J, Reuner U, et al. Cidofovir and foscarnet for treatment of human herpesvirus 6 encephalitis in a neutropenic stem cell transplant recipient. Clin Infect Dis. 2007;44(12):e118–20.
Astriti M, Zeller V, Boutolleau D, et al. Fatal HHV-6 associated encephalitis in an HIV-1 infected patient treated with cidofovir. J Infect. 2006;52(4):237–42.
Gunther G, Haglund M, Lindquist L, et al. Tick-bone encephalitis in Sweden in relation to aseptic meningo-encephalitis of other etiology: a prospective study of clinical course and outcome. J Neurol. 1997;244(4):230–8.
Kaiser R. The clinical and epidemiological profile of tick-borne encephalitis in southern Germany 1994–98: a prospective study of 656 patients. Brain. 1999;122(Pt 11):2067–78.
Mickiene A, Laiskonis A, Gunther G, et al. Tickborne encephalitis in an area of high endemicity in Lithuania: disease severity and long-term prognosis. Clin Infect Dis. 2002;35(6):650–8.
Ecker M, Allison SL, Meixner T, et al. Sequence analysis and genetic classification of tick-borne encephalitis viruses from Europe and Asia. J Gen Virol. 1999;80(Pt 1):179–85.
Holzmann H, Vorobyova MS, Ladyzhenskaya IP, et al. Molecular epidemiology of tick-borne encephalitis virus: cross-protection between European and Far Eastern subtypes. Vaccine. 1992;10(5):345–9.
Holmgren EB, Forsgren M. Epidemiology of tick-borne encephalitis in Sweden 1956–1989: a study of 1116 cases. Scand J Infect Dis. 1990;22(3):287–95.
Haglund M, Forsgren M, Lindh G, et al. A 10-year follow-up study of tick-borne encephalitis in the Stockholm area and a review of the literature: need for a vaccination strategy. Scand J Infect Dis. 1996;28(3):217–24.
Puchhammer-Stockl E, Kunz C, Mandl CW, et al. Identification of tick-borne encephalitis virus ribonucleic acid in tick suspensions and in clinical specimens by a reverse transcription-nested polymerase chain reaction assay. Clin Diagn Virol. 1995;4(4):321–6.
Saksida A, Duh D, Lotric-Furlan S, et al. The importance of tick-borne encephalitis virus RNA detection for early differential diagnosis of tick-borne encephalitis. J Clin Virol. 2005;33(4):331–5.
Andersson CR, Vene S, Insulander M, et al. Vaccine failures after active immunisation against tick-borne encephalitis. Vaccine. 2010;28(16):2827–31.
Stiasny K, Holzmann H, Heinz FX. Characteristics of antibody responses in tick-borne encephalitis vaccination breakthroughs. Vaccine. 2009;27(50):7021–6.
Lorenzl S, Pfister HW, Padovan C, et al. MRI abnormalities in tick-borne encephalitis. Lancet. 1996;347(9002):698–9.
Marjelund S, Tikkakoski T, Tuisku S, et al. Magnetic resonance imaging findings and outcome in severe tick-borne encephalitis: report of four cases and review of the literature. Acta Radiol. 2004;45(1):88–94.
Waldvogel K, Bossart W, Huisman T, et al. Severe tick-borne encephalitis following passive immunisation. Eur J Pediatr. 1996;155(9):775–9.
Broker M, Kollaritsch H. After a tick bite in a tick-borne encephalitis virus endemic area: current positions about post-exposure treatment. Vaccine. 2008;26(7):863–8.
Stefanoff P, Polkowska A, Giambi C, et al. Reliable surveillance of tick-borne encephalitis in European countries is necessary to improve the quality of vaccine recommendations. Vaccine. 2011;29(6):1283–8.
Heinz FX, Holzmann H, Essl A, et al. Field effectiveness of vaccination against tick-borne encephalitis. Vaccine. 2007;25(43):7559–67.
Weinberger B, Keller M, Fischer KH, et al. Decreased antibody titers and booster responses in tick-borne encephalitis vaccinees aged 50–90 years. Vaccine. 2010;28(20):3511–5.
Hainz U, Jenewein B, Asch E, et al. Insufficient protection for healthy elderly adults by tetanus and TBE vaccines. Vaccine. 2005;23(25):3232–5.
Loew-Baselli A, Poellabauer EM, Pavlova BG, et al. Seropersistence of tick-borne encephalitis antibodies, safety and booster response to FSME-IMMUN 0.5 ml in adults aged 18–67 years. Hum Vaccin. 2009;5(8):551–6.
Loew-Baselli A, Konior R, Pavlova BG, et al. Safety and immunogenicity of the modified adult tick-borne encephalitis vaccine FSME-IMMUN: results of two large phase 3 clinical studies. Vaccine. 2006;24(24):5256–63.
Zent O, Jilg W, Plentz A, et al. Kinetics of the immune response after primary and booster immunization against tick-borne encephalitis (TBE) in adults using the rapid immunization schedule. Vaccine. 2003;21(32):4655–60.
Beran J, Douda P, Gniel D, et al. Long-term immunity after vaccination against tick-borne encephalitis with encepur using the rapid vaccination schedule. In J Med Microbiol. 2004;293(Suppl 37):130–3.
Schöndorf I, Beran J, Cizkova D, et al. Tick-borne encephalitis (TBE) vaccination: applying the most suitable vaccination schedule. Vaccine. 2007;25(8):1470–5.
Stiasny K, Aberle JH, Keller M, et al. Age affects quantity but not quality of antibody responses after vaccination with an inactivated flavivirus vaccine against tick-borne encephalitis. PLoS One. 2012;7(3):e34145.
Lee BE, Davies HD. Aseptic meningitis. Curr Opin Infect Dis. 2007;20(3):272–7.
Ropka SL, Jubelt B. Enteroviruses. In: Nath A, Berger JR, editors. Clinical neurovirology. New York: Marcel-Dekker; 2003. p. 359–77.
Sawyer MH, Rotbart HA. Viral meningitis and aseptic meningitis syndrome. In: Scheld WM, Whitley RJ, Marra CM, editors. Infections of the central nervous system. Philadelphia: Lippincott Williams & Wilkins; 2004. p. 75–93.
Rotbart HA. Enteroviral infections of central nervous system. Clin Infect Dis. 1995;20(4):971–81.
Polio Global Eradication Initiative. Wild poliovirus weekly update. http://www.polioeradication.org/. Accessed 8 Feb 2012.
Ho M. Enterovirus 71: the virus, its infections and outbreaks. J Microbiol Immunol Infect. 2000;33(4):205–16.
Kelly H, Brussen KA, Lawrence A, et al. Polioviruses and other enteroviruses isolated from faecal samples of patients with acute flaccid paralysis in Australia, 1996–2004. J Paediatr Child Health. 2006;42(6):370–6.
Glimåker M, Johansson B, Olcen P, et al. Detection of enteroviral RNA by polymerase chain reaction in cerebrospinal fluid from patients with aseptic meningitis. Scand J Infect Dis. 1993;25(5):547–57.
Mohamed N, Elfaitouri A, Fohlman J, et al. A sensitive and quantitative single-tube real-time reverse transcriptase-PCR for detection of enteroviral RNA. J Clin Virol. 2004;30(2):150–6.
Sato M, Hosoya M, Honzumi K, et al. Cytokine and cellular inflammatory sequence in enteroviral meningitis. Pediatrics. 2003;112(5):1103–7.
King RL, Lorch SA, Cohen DM, et al. Routine cerebrospinal fluid enterovirus polymerase chain reaction testing reduces hospitalization and antibiotic use for infants 90 days of age or younger. Pediatrics. 2007;120(3):489–96.
Nigrovic LE, Chiang VW. Cost analysis of enteroviral polymerase chain reaction in infants with fever and cerebrospinal fluid pleocytosis. Arch Pediatr Adolesc Med. 2000;154(8):817–21.
Ninove L, Tan C, Nougairede A, et al. Impact of diagnostic procedures on patient management and hospitalization cost during the 2000 and 2005 enterovirus epidemics in Marseilles, France. Clin Microbiol Infect. 2010;16(6):651–6.
Fauquet C, Mayo M, Maniloff J, et al., editors. Virus taxonomy: eighth report of the International Committee on Taxonomy of Viruses. San Diego: Elsevier Inc.; 2005.
ICTVdB management. 00.052. Picornaviridae. In: Büchen-Osmond C, editor. ICTVdB: the universal virus database, version 4. New York: Columbia University; 2006.
Glimåker M, Samuelson A, Magnius L, et al. Early diagnosis of enteroviral meningitis by detection of specific IgM antibodies with a solid-phase reverse immunosorbent test (SPRIST) and m-capture EIA. J Med Virol. 1992;36(3):193–201.
Elfaitouri A, Mohamed N, Fohlman J, et al. Quantitative PCR-enhanced immunoassay for measurement of enteroviral immunoglobulin M antibody and diagnosis of aseptic meningitis. Clin Diagn Lab Immunol. 2005;12(2):235–41.
Desmond RA, Accortt NA, Talley L, et al. Enteroviral meningitis: natural history and outcome of pleconaril therapy. Antimicrob Agents Chemother. 2006;50(7):2409–14.
Abzug MJ, Cloud G, Bradley J, Sánchez PJ, Romero J, National Institute of Allergy and Infectious Diseases Collaborative Antiviral Study Group, et al. Double blind placebo-controlled trial of pleconaril in infants with enterovirus meningitis. Pediatr Infect Dis J. 2003;22(4):335–41.
Li ZH, Li CM, Ling P, et al. Ribavirin reduces mortality in enterovirus 71-infected mice by decreasing viral replication. J Infect Dis. 2008;197(6):854–7.
Quartier P, Foray S, Casanova JL, et al. Enteroviral meningoencephalitis in X-linked agammaglobulinemia: intensive immunoglobulin therapy and sequential viral detection in cerebrospinal fluid by polymerase chain detection. Pediatr Infect Dis J. 2000;19(11):1106–8.
Abzug MJ, Keyserling HL, Lee ML, et al. Neonatal enterovirus infection: virology, serology, and effects of intravenous immune globulin. Clin Infect Dis. 1995;20(5):1201–6.
Wang SM, Lei HY, Huang MC, et al. Modulation of cytokine production by intravenous immunoglobulin in patients with enterovirus 71-associated brainstem encephalitis. J Clin Virol. 2006;37(1):47–52.
McMinn PC. Recent advances in the molecular epidemiology and control of human enterovirus 71 infection. Curr Opin Virol. 2012;2(2):199–205.
Mizuguchi M, Abe J, Mikkaichi K, et al. Acute necrotising encephalopathy of childhood: a new syndrome presenting with multifocal, symmetric brain lesions. J Neurol Neurosurg Psychiatry. 1995;58(5):555–61.
Kirton A, Busche K, Ross C, et al. Acute necrotizing encephalopathy in caucasian children: two cases and review of the literature. J Child Neurol. 2005;20(6):527–32.
Mastroyianni SD, Gionnis D, Voudris K, et al. Acute necrotizing encephalopathy of childhood in non-Asian patients: report of three cases and literature review. J Child Neurol. 2006;21(10):872–9.
Fasano A, Natoli GF, Cianfoni A, et al. Acute necrotizing encephalopathy: a relapsing case in a European adult. J Neurol Neurosurg Psychiatry. 2008;79(2):227–8.
Amin R, Ford-Jones E, Richardson SE, et al. Acute childhood encephalitis and encephalopathy associated with influenza: a prospective 11-year review. Pediatr Infect Dis J. 2008;27(5):390–5.
Fowler A, Stödberg T, Eriksson M, et al. Childhood encephalitis in Sweden: etiology, clinical presentation and outcome. Eur J Paediatr Neurol. 2008;12(6):484–90.
Surana P, Tang S, McDougall M, et al. Neurological complications of pandemic influenza A H1N1 2009 infection: European case series and review. Eur J Pediatr. 2011;170(8):1007–15.
Studahl M, Linde A. Influenza and CNS complications. In: Nath A, Berger JR, editors. Clinical neurovirology. 1st ed. New York: Marcel-Dekker; 2003. p. 453–68.
Fujimoto S, Kobayashi M, Uemura O, et al. PCR on cerebrospinal fluid to show influenza-associated acute encephalopathy or encephalitis. Lancet. 1998;352(9131):873–5.
Kimura S, Ohtuki N, Nezu A, et al. Clinical and radiological variability of influenza-related encephalopathy or encephalitis. Acta Paediatr Jpn. 1998;40(3):264–70.
Takanashi J. Two newly proposed infectious encephalitis/encephalopathy syndromes. Brain Dev. 2009;31(7):521–8.
Studahl M. Influenza virus and CNS manifestations. J Clin Virol. 2003;28(3):225–32.
Wang GF, Li W, Li K. Acute encephalopathy and encephalitis caused by influenza virus infection. Curr Opin Neurol. 2010;23(3):305–11.
Falagas ME, Koletsi PK, Vouloumanou EK, et al. Effectiveness and safety of neuraminidase inhibitors in reducing influenza complications: a meta-analysis of randomized controlled trials. J Antimicrob Chemother. 2010;65(7):1330–46.
Jhee SS, Yen M, Ereshefsky L, et al. Low penetration of oseltamivir and its carboxylate into cerebrospinal fluid in healthy Japanese and Caucasian volunteers. Antimicrob Agents Chemother. 2008;52(10):3687–93.
Kornhuber J, Quack G, Danysz W, et al. Therapeutic brain concentration of the NMDA receptor antagonist amantadine. Neuropharmacology. 1995;34(7):713–21.
Munakata M, Kato R, Yokoyama H, et al. Combined therapy with hypothermia and anticytokine agents in influenza A encephalopathy. Brain Dev. 2000;22(6):373–7.
Jefferson T, Di Pietrantonj C, Rivetti A, et al. Vaccines for preventing influenza in healthy adults. Cochrane Database Syst Rev. 2010;7:CD001269.
Sugaya N, Takeuchi Y. Mass vaccination of schoolchildren against influenza and its impact on the influenza-associated mortality rate among children in Japan. Clin Infect Dis. 2005;41(7):939–47.
Miller HG, Stanton JB, Gibbons JL. Para-infectious encephalomyelitis and related syndromes: a critical review of the neurological complications of certain specific fevers. Q J Med. 1956;25(100):427–505.
Baba Y, Tsuboi Y, Inoue H, et al. Acute measles encephalitis in adults. J Neurol. 2006;253(1):121–4.
Aicardi J, Goutieres F, Arsenio-Nunes ML, et al. Acute measles encephalitis in children with immunosuppression. Pediatrics. 1977;59(2):232–9.
Norrby E, Kristensson K. Measles virus in the brain. Brain Res Bull. 1997;44(3):213–20.
Gutierrez J, Issacson RS, Koppel BS. Subacute sclerosing panencephalitis: an update. Dev Med Child Neurol. 2010;52(10):901–7.
Hviid A, Rubin S, Mühlemann K. Mumps. Lancet. 2008;371(9616):932–44.
Koskiniemi M, Donner M, Pettay O. Clinical appearance and outcome in mumps encephalitis in children. Acta Paediatr Scand. 1983;72(4):603–9.
Griffin DE. Measles and rubella. In: Scheld WM, Whitley RJ, Marra CM, editors. Infections of the central nervous system. 3rd ed. Philadelphia: Lippincott Williams & Wilkins; 2004. p. 111–22.
Nath A. Rubella. In: Nath A, Berger JR, editors. Clinical neurovirology. 1st ed. London: Taylor and Francis; 2003. p 447–52.
Weissbrich B, Schneider-Schaulies J, ter Meulen V. Measles and its neurological complications. In: Nath A, Berger JR, editors. Clinical neurovirology. 1st ed. London: Taylor and Francis; 2003. p 401–30.
Health Protection Agency UK. Investigation of viral encephalitis and meningitis. National Standard Method QSOP. 2004;48(2):1–29.
Garg RK. Subacute sclerosing panencephalitis. J Neurol. 2008;255(12):1861–71.
Baum SG, Litman N. Mumps virus. In: Mandell GL, Bennett JE, Dolin R, editors. Mandell, Douglas and Bennett’s principles and practice of infectious diseases. 5th ed. Philadelphia: Churchill-Livingstone; 2000. p. 1776–81.
Lau KK, Lai ST, Lai JY, et al. Acute encephalitis complicating rubella. Hong Kong Med J. 1998;4(3):325–8.
Bosma TJ, Corbett KM, O′Shea S, et al. PCR for detection of rubella virus RNA in clinical samples. J Clin Microbiol. 1995;33(5):1075–9.
Schneider-Schaulies J, ter Meulen V, Schneider-Schaulies S. Measles infection of the central nervous system. J Neurovirol. 2003;9(2):247–52.
Sugita K, Ando M, Minamitani K, et al. Magnetic resonance imaging in a case of mumps postinfectious encephalitis with asymptomatic optic neuritis. Eur J Pediatr. 1991;150(11):773–5.
Liebert UG. Measles virus infections of the central nervous system. Intervirology. 1997;40(2–3):176–84.
Wetzel K, Asholt I, Herrmann E, et al. Good cognitive outcome of patients with herpes zoster encephalitis: a follow-up study. J Neurol. 2002;249(11):1612–4.
Adour KK. Otological complications of herpes zoster. Ann Neurol. 1994;35 Suppl:S62–4.
Wagstaff AJ, Faulds D, Goa KL, et al. Aciclovir: a reappraisal of its antiviral activity, pharmacokinetic properties and therapeutic efficacy. Drugs. 1994;47(1):153–205.
Gnann JW Jr. Varicella zoster virus: atypical presentations and unusual complications. J Infect Dis. 2002;186(Suppl. 1):S91–8.
Wood MJ, Kay R, Dworkin RH, et al. Oral acyclovir therapy accelerates pain resolution in patients with herpes zoster: a meta-analysis of placebo-controlled trials. Clin Infect Dis. 1996;2282:341–7.
Wood MJ, Shukla S, Fiddian AP, et al. Treatment of acute herpes zoster: effect of early (<48 h) versus late (48–72 h) therapy with acyclovir and valacyclovir on prolonged pain. J Infect Dis. 1998;178(Suppl. 1):S81–4.
Gilden DH, Mahalingam R, Cohrs RJ, et al. Herpesvirus infections of the central nervous system. Nat Clin Pract Neurol. 2007;3(2):82–94.
Sekiyama T, Hatsua S, Tanaka Y, et al. Synthesis and antiviral activity of novel acyclic nucleosides: discovery of a cyclopropyl nucleoside with potent inhibitory activity against herpesviruses. J Med Chem. 1998;41(8):1284–98.
Kimberlin DW, Lin CY, Jacobs RF, et al. Safety and efficacy of high-dose intravenous acyclovir in the management of neonatal herpes simplex virus infections. Pediatrics. 2001;108(2):230–8.
Bean B, Aeppli D. Adverse effects of high-dose intravenous acyclovir in ambulatory patients with acute herpes zoster. J Infect Dis. 1985;151(2):362–5.
Perazella MA. Crystal-induced acute renal failure. Am J Med. 1999;106(4):45–65.
Hellden A, Lycke J, Vander T, et al. The acyclovir metabolite CMMG is detectable in the CSF of subjects with neuropsychiatric symptoms during acyclovir and valaciclovir treatment. J Antimicrob Chemother. 2006;57(5):945–9.
Weller S, Blum R, Doucette M, et al. Pharmacokinetics of the acyclovir prodrug valacyclovir after escalating single-and multiple-dose administration to normal volunteers. Clin Pharmacol Ther. 1993;54(6):595–605.
Pouplin T, Pouplin JN, Van Toi P, et al. Valacyclovir for herpes simplex encephalitis. Antimicrob Agents Chemother. 2011;55(7):3624–6.
Frank KB, Chiou JF, Cheng YC. Interaction of herpes simplex virus-induced DNA polymerase with 9-(1,3-dihydroxy-2-propoxymethyl) guanine triphosphate. J Biol Chem. 1984;259(3):1566–9.
Russler SK, Tapper MA, Carrigan DR. Susceptibility of human herpesvirus 6 to acyclovir and ganciclovir. Lancet. 1989;2(8659):382.
Crumpacker CS. Ganciclovir. N Engl J Med. 1996;335(10):721–9.
Cinque P, Cleator GM, Weber T, et al. Diagnosis and clinical management of neurological disorders caused by cytomegalovirus in AIDS patients. J Neurovirol. 1998;4(1):120–32.
Brown F, Banken L, Saywell K, et al. Pharmacokinetics of valganciclovir and ganciuclovir following multiple oral dosages of valganciclovir in HIV-and CMV-seropositive volunteers. Clin Pharmacokinet. 1999;37(2):161–76.
Martin DF, Sierra-Mader J, Walmsley S, et al. A controlled trial of valganciclovir as induction therapy for cytomegalovirus retinitis. N Engl J Med. 2002;346(15):1119–26.
Gerard L, Salmon-Ceron D. Pharmacology and clinical use of foscarnet. Int J Antimicrob Agents. 1995;5(4):209–17.
Jabs DA, Enger C, Forman M, The cytomegalovirus retinitis and viral resistance study group, et al. Incidence of foscarnet resistance and cidofovir resistance in patients treated for cytomegalovirus retinitis. Antimicrob Agents Chemother. 1998;42(9):2240–4.
Peters M, Timm U, Schürmann D, et al. Combined and alternating ganciclovir and foscarnet in acute and maintenance therapy of human immunodeficiency virus-related cytomegalovirus encephalitis refractory to ganciclovir alone: a case report and review of the literature. Clin Invest. 1992;70(5):456–8.
Peavear DC, Tull TM, Seipel ME, et al. Activity of pleconaril against enteroviruses. Antimicrob Agents Chemother. 1999;43(9):2109–15.
L′Huillier AG, Lorenzini KI, Crisinel PA, et al. Polymorphisms and neuropsychiatric adverse events in oseltamivir-treated children during influenza H1N1/09 pandemia. Pharmacogenomics. 2011;12(10):1493–501.
Field HJ, Darby G, Wildy P. Isolation and characterization of acyclovir resistant mutants of herpes simplex virus. J Gen Virol. 1980;49(1):115–24.
Pottage JC Jr, Kessler HA. Herpes simplex virus resistance to acyclovir: clinical relevance. Infect Agents Dis. 1995;4(3):115–24.
Biron KK, Fyfe JA, Noblin JE, et al. Selection and preliminary characterization of acyclovir resistant mutants of varicella zoster virus. Am J Med. 1982;73(1A):383–6.
Bacon TH, Boon RJ, Schultz M, et al. Surveillance for antiviral-agent-resistant herpes simplex virus in the general population with recurrent herpes labialis. Antimicrob Agents Chemother. 2002;46:3042–4.
Grey F, Sowa M, Collins P, et al. Characterization of a neurovirulent acyclovir-resistant variant of herpes simplex virus. J Gen Virol. 2003;84:1403–10.
Schulte EC, Sauerbrei A, Hoffman D, et al. Acyclovir resistance in herpes simplex encephalitis. Ann Neurol. 2010;67(6):830–3.
Kakiuchi S, Nonoyama S, Wakamatsu H, et al. Neonatal herpes encephalitis caused by a virologically confirmed acyclovir resistant herpes simplex virus type 1. J Clin Microbiol. 2013;51(1):356–9.
Brink AA, van Gelder M, Wolffs PF, et al. Compartmentalization of acyclovir-resistant varicella zoster virus: implications for sampling in molecular diagnostics. Clin Infect Dis. 2011;52(8):982–7.
Aurelius E, Forsgren M, Sköldenberg B, et al. Persistent intrathecal immune activation in patients with herpes simplex encephalitis. J Infect Dis. 1993;168(5):1248–52.
Aurelius E, Andersson B, Forsgren M, et al. Cytokines and other markers of intrathecal immune response in patients with herpes simplex encephalitis. J Infect Dis. 1994;170(3):678–81.
Thompson KA, Blessing WW, Wesselingh SL. Herpes simplex replication and dissemination is not increased by corticosteroid treatment in a rat model of focal Herpes encephalitis. J Neurovirol. 2000;6(1):25–32.
Meyding-Lamade UK, Oberlinner C, Rau PR, et al. Experimental herpes simplex virus encephalitis: a combination therapy of acyclovir and glucocorticoids reduces long-term magnetic resonance imaging abnormalities. J Neurovirol. 2003;9(1):118–25.
Mailles A, De Broucker T, Costanzo P, et al. Long-term outcome of patients presenting with acute infectious encephalitis of various causes in France. Clin Infect Dis. 2012;54(10):1455–64.
De Broucker T, Mailles A, Chabrier S, et al. Acute varicella zoster encephalitis without evidence of primary vasculopathy in a case series of 20 patients. Clin Microbiol Infect. 2012;18(8):808–19.
Acknowledgments
This work was supported by The Swedish Society of Infectious Diseases and the LUA Foundation of the Sahlgrenska University Hospital.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Studahl, M., Lindquist, L., Eriksson, BM. et al. Acute Viral Infections of the Central Nervous System in Immunocompetent Adults: Diagnosis and Management. Drugs 73, 131–158 (2013). https://doi.org/10.1007/s40265-013-0007-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40265-013-0007-5