Abstract
Background and objective
Type 2 diabetes mellitus (T2DM) is frequently associated with co-morbidities that exacerbate cardiovascular (CV) risk. CV disease is the leading cause of death in people with diabetes across the world and accounts for approximately half the deaths in the T2DM population. Hence, the objective of present study was to evaluate the cost-effectiveness of empagliflozin, in addition to standard of care (SoC), for the treatment of adult patients with T2DM and high CV risk in Greece.
Methods
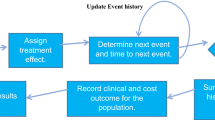
A health economic model was used to project clinical and economic outcomes of patients receiving empagliflozin plus SoC compared with those receiving SoC alone over a lifetime horizon. CV and renal event rates were derived from patient level data from the EMPA-REG-OUTCOME® trial by fitting time-dependent parametric survival functions. 5000 individual patient profiles randomly sampled from the trial were simulated using a time-to-event approach. Model extrapolated outcomes included life years (LYs), quality-adjusted life years (QALYs), costs, and incremental cost-effectiveness ratio (ICER). Following a Greek third-party payer perspective, only direct medical costs related to drug acquisition as well as fatal and non-fatal diabetes-related complications were considered (€2016). Cost units and utility data were extracted from the literature and publicly available official sources. Sensitivity analyses explored the impact of changes in input data.
Results
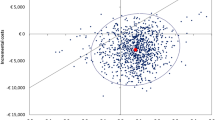
Over a patient’s lifetime, empagliflozin was predicted to result in longer mean survival (14.01 LY vs. 11.87 LY with SoC) and reduced rate of clinical events accumulating 7.75 QALYs versus 6.83 QALYs on SoC alone at additional costs of €4235. The generated ICER of empagliflozin was €4633 per QALY gained. One-way sensitivity analysis confirmed empagliflozin’s cost-effective profile. At the defined willingness-to-pay threshold of €34,000 per QALY gained, probabilistic sensitivity analysis showed that empagliflozin was estimated to have a 100% probability of being cost-effective relative to SoC.
Conclusions
Empagliflozin added to SoC was estimated to be a highly cost-effective treatment option for the treatment of T2DM in adults with increased CV disease risk in Greece.


Similar content being viewed by others
References
World Health Organization. Global report on Diabetes. 2016; Available from: http://apps.who.int/iris/bitstream/10665/204871/1/9789241565257_eng.pdf?ua=1. Accessed 11 Jan 2017.
Mathers CD, Loncar D. Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med. 2006;3(11):e442.
Liatis S, Dafoulas GE, Kani C, Politi A, Litsa P, Sfikakis PP, et al. The prevalence and treatment patterns of diabetes in the Greek population based on real-world data from the nation-wide prescription database. Diabetes Res Clin Pract. 2016;118:162–7.
Meigs JB. Epidemiology of cardiovascular complications in type 2 diabetes mellitus. Acta Diabetol. 2003;40(Suppl 2):S358–61.
Boldys A, Okopien B. Inhibitors of type 2 sodium glucose co-transporters–a new strategy for diabetes treatment. Pharmacol Rep. 2009;61(5):778–84.
Emerging Risk Factors, C., Di Angelantonio E, Kaptoge S, Wormser D, Willeit P, Butterworth AS, et al. Association of cardiometabolic multimorbidity with mortality. JAMA. 2015;314(1):52–60.
Zinman B, Inzucchi SE, Lachin JM, Wanner C, Ferrari R, Fitchett D, et al. Rationale, design, and baseline characteristics of a randomized, placebo-controlled cardiovascular outcome trial of empagliflozin (EMPA-REG OUTCOME). Cardiovasc Diabetol. 2014;13:102.
International Diabetes Federation. Diabetes, ATLAS Seventh Edition. 2015; Available from: http://www.diabetesatlas.org/. Accessed 14 Sept 2016.
Ninomiya T, Perkovic V, de Galan BE, Zoungas S, Pillai A, Jardine M, et al. Albuminuria and kidney function independently predict cardiovascular and renal outcomes in diabetes. J Am Soc Nephrol. 2009;20(8):1813–21.
Bennett WL, Maruthur NM, Singh S, Segal JB, Wilson LM, Chatterjee R, et al. Comparative effectiveness and safety of medications for type 2 diabetes: an update including new drugs and 2-drug combinations. Ann Intern Med. 2011;154(9):602–13.
Rosenstock J, Marx N, Kahn SE, Zinman B, Kastelein JJ, Lachin JM, et al. Cardiovascular outcome trials in type 2 diabetes and the sulphonylurea controversy: rationale for the active-comparator CAROLINA trial. Diabetes Vasc Dis Res. 2013;10(4):289–301.
Zinman B, Wanner C, Lachin JM, Fitchett D, Bluhmki E, Hantel S, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373(22):2117–28.
Gallo LA, Wright EM, Vallon V. Probing SGLT2 as a therapeutic target for diabetes: basic physiology and consequences. Diabetes Vasc Dis Res. 2015;12(2):78–89.
Grempler R, Thomas L, Eckhardt M, Himmelsbach F, Sauer A, Sharp DE, et al. Empagliflozin, a novel selective sodium glucose cotransporter-2 (SGLT-2) inhibitor: characterisation and comparison with other SGLT-2 inhibitors. Diabetes Obes Metab. 2012;14(1):83–90.
Fitchett D, Zinman B, Wanner C, Lachin JM, Hantel S, Salsali A, et al. Heart failure outcomes with empagliflozin in patients with type 2 diabetes at high cardiovascular risk: results of the EMPA-REG OUTCOME(R) trial. Eur Heart J. 2016;37(19):1526–34.
Kansal A, Zheng Y, Proskorovsky I, Krotneva S, Kandaswamy P, Ruffolo A. PDB37—modeling cardiovascular outcomes of treatment with empagliflozin in typer 2 diabetes based on hard outcomes data. Value Health. 2016;19(3):A203.
Mettam SR, Bajaj H, Kansal AR, Kandaswamy P. PDB52—cost effectiveness of empagliflozin in patients with T2DM and high CV risk in Canada. Value Health. 2016;19(7):A674.
Daacke I, Kandaswamy P, Tebboth A, Kansal A, Reifsnider O. PDB46—cost-effectiveness of empagliflozin (jardiance) in the treatment of patients with type 2 diabetes mellitus (T2DM) in the UK based on empa-reg outcome data. Value Health. 2016;19(7):A673.
World Health Organization (WHO). Greece life tables. Available from: http://apps.who.int/gho/data/view.main.60640. Accessed 12 Sept 2016.
Kourlaba G, Rapti V, Alexopoulos A, Relakis J, Koumakis G, Chatzikou M, et al. Everolimus plus exemestane versus bevacizumab-based chemotherapy for second-line treatment of hormone receptor-positive metastatic breast cancer in Greece: an economic evaluation study. BMC Health Serv Res. 2015;15:307.
Maniadakis N, Kaitelidou D, Siskou O, Spinthouri M, Liaropoulos L, Fragoulakis B, et al. Economic evaluation of treatment strategies for patients suffering acute myocardial infarction in Greece. Hellenic J Cardiol. 2005;46(3):212–21.
Gourzoulidis G, Kourlaba G, Kakisis J, Matsagkas M, Giannakoulas G, Gourgoulianis KI, et al. Cost-effectiveness analysis of rivaroxaban for treatment of deep vein thrombosis and pulmonary embolism in Greece. Clin Drug Investig. 2017;37(9):833–44.
Sullivan PW, Ghushchyan VH. EQ-5D scores for diabetes-related comorbidities. Value Health. 2016;19(8):1002–8.
Greek Ministry of Health. Drug price bulletin 2016. Available from: http://www.moh.gov.gr/articles/times-farmakwn/deltia-timwn. Accessed 31 Dec 2016.
Tzanetakos C, Melidonis A, Verras C, Kourlaba G, Maniadakis N. Cost-effectiveness analysis of liraglutide versus sitagliptin or exenatide in patients with inadequately controlled type 2 diabetes on oral antidiabetic drugs in Greece. BMC Health Serv Res. 2014;14:419.
Tzanetakos C, Tentolouris N, Kourlaba G, Maniadakis N. Cost-effectiveness of dapagliflozin as add-on to metformin for the treatment of type 2 diabetes mellitus in Greece. Clin Drug Investig. 2016;36(8):649–59.
Hellenic Statistical Authority (EL. STAT.). Inflation rates. Available from: http://www.statistics.gr/portal/page/portal/ESYE. Accessed 15 Dec 2016.
Greek Ministry of Health. Diagnostic Related Groups. Available from: http://www.yyka.gov.gr/articles/health/domes-kai-draseis-gia-thn-ygeia/kwdikopoihseis/709-kleista-enopoihmena-noshlia-1. Accessed 20 Dec 2016.
National Organisation for HealthcareServices Provision. Official web site of EOPYY. 2016; Available from: http://www.eopyy.gov.gr/Home/StartPage?a_HomePage=Index. Accessed 22 Dec 2016.
WHO. Choosing interventions that are cost effective (WHO-CHOICE): cost-effectiveness thresholds. 2013; Available from: http://www.who.int/choice/costs/CER_thresholds/en/index.html. Accessed 22 Jan 2017.
International Monetary Fund. World Economic Outlook Database. 2017; Available from: http://www.imf.org/external/pubs/ft/weo/2017/01/weodata/index.aspx. Accessed 18 Jan 2017.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by Βoehringer Ingelheim Hellas. However, the study sponsor had no interference in the study design, data collection, data analysis, or writing of the manuscript.
Conflict of interest and financial disclosure
II received honoraria for lectures from Boehringer Ingelheim. NM received speaker honoraria and consulting fees from Boehringer Ingelheim. AT received consulting fees and honoraria for lectures from Boehringer Ingelheim.GP is a Boehringer Ingelheim employee. None of the other authors has any personal or financial conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gourzoulidis, G., Tzanetakos, C., Ioannidis, I. et al. Cost-Effectiveness of Empagliflozin for the Treatment of Patients with Type 2 Diabetes Mellitus at Increased Cardiovascular Risk in Greece. Clin Drug Investig 38, 417–426 (2018). https://doi.org/10.1007/s40261-018-0620-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40261-018-0620-x