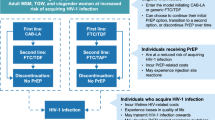
Abstract
Background
Currently, two-drug antiretroviral regimens are emerging fields in life-long treatment in people living with HIV.
Objectives
This randomized non-inferiority open-label controlled trial was designed to compare the 48-week efficacy and safety of tenofovir alafenamide plus dolutegravir versus the standard triple therapy in virologically suppressed people living with HIV. To the best of our knowledge this combination has not been studied before.
Methods
This open-label randomized controlled trial was conducted in treatment-experienced people with HIV who had HIV-RNA < 47 copies/mL for at least two years. Patients received either tenofovir alafenamide plus dolutegravir combination (26 patients) or a standard three-drug regimen (29 patients). The primary outcome was the proportion of patients maintaining HIV-RNA < 47 copies/mL during 48 weeks, and the secondary outcomes were CD4 cell count changes, the adherence rate, and adverse drug reactions, all over 48 weeks of study.
Results
HIV viral load remained undetectable (HIV-RNA < 47 copies/mL) during the 48 weeks of the study in both arms. The absolute CD4 cell count change was not significant between the two groups. The overall proportion of adverse effects in each group was comparable. The rate of adherence to treatment was acceptable in both groups, and no significant difference was observed.
Conclusions
Treatment simplification with tenofovir alafenamide plus dolutegravir regimen as maintenance therapy was non-inferior in terms of efficacy and safety compared to the standard triple therapy.
Graphical abstract
Comparing efficacy of antiretroviral therapy



Similar content being viewed by others
Data availability
The datasets used and analyzed during the current study are available from the corresponding author upon request.
References
Iacob SA, Iacob DG, Jugulete G. Improving the adherence to antiretroviral therapy, a difficult but essential task for a successful HIV treatment-clinical points of view and practical considerations. Front Pharmacol. 2017;8:831. https://doi.org/10.3389/fphar.2017.00831.
De Miguel BR, Montejano R, Stella-Ascariz N, Arribas JR. New strategies of ARV: the road to simplification. Curr HIV/AIDS Rep. 2018;15(1):11–9. https://doi.org/10.1007/s11904-018-0371-6.
Khalili H, Rohani R, Seyedalinaghi S, Hajiabdolbaghi M, Dashti-Khavidaki S, Talasaz AH. Adherence to Antiretroviral therapy among Iranian HIV/AIDS Patients. Curr Clin Pharmacol. 2012;7:111–5. https://doi.org/10.2174/157488412800228910.
Moradi A, Alavi SM, Fahim far N, Haghighizadeh MH, Mirzaei B. Study of determinants of adherence to antiretroviral treatment among HIV Patients covered by Ahwaz Jundishapur University of Medical Sciences. Int J Med Res Health Sci. 2016;5:477–84.
Bojdy A, Arian M, Najafi M, Mottaghi M. Adherence to antiretroviral therapy and its determinants in HIV patients in Mashhad, IRAN, a prospective study. Rev Clin Med. 2018;7:157–62.
Morowatisharifabad MA, Movahed E, Farokhzadian J, Nikooie R, Hosseinzadeh M, Askarishahi M, et al. Antiretroviral therapy adherence and its determinant factors among people living with HIV/AIDS: a case study in Iran. BMC Res Notes. 2019;12:162. https://doi.org/10.1186/s13104-019-42045.
Chastain D, Badowski M, Huesgen E, Pandit NS, Pallotta A, Michienzi S. Optimizing antiretroviral therapy in treatment-experienced patients living with HIV: a critical review of switch and simplification strategies. An opinion of the HIV practice and research network of the American College of clinical pharmacy. J Int Assoc Provid AIDS Care. 2019;18:2325958219867325. https://doi.org/10.1177/2325958219867325.
Corado KC, Caplan MR, Daar ES. Two-drug regimens for treatment of naïve HIV-1 infection and as maintenance therapy. Drug Des Devel Ther. 2018;12:3731–40. https://doi.org/10.2147/DDDT.S140767.
Badowski M, Pérez SE, Silva D, Lee A. Two’s a company, three’s a crowd: a review of initiating or switching to a two-drug antiretroviral regimen in treatment-naïve and treatment-experienced patients living with HIV-1. Infect Dis Ther. 2020;9:185–208. https://doi.org/10.1007/s40121-020-00290-w.
Waters L, Church H. Two drug regimens for HIV. Curr Opin Infect Dis. 2020;33:28–33. https://doi.org/10.1097/QCO.0000000000000615.
Brooks KM, Sherman EM, Egelund EF, Brotherton A, Durham S, Badowski ME, et al. Integrase inhibitors: after 10 years of experience, is the best yet to come? Pharmacotherapy. 2019;39:576–98. https://doi.org/10.1002/phar.2246.
Wandeler G, Buzzi M, Anderegg N, Sculier D, Béguelin C, Egger M, et al. Virologic failure and HIV drug resistance on simplified, dolutegravir-based maintenance therapy: Systematic review and meta-analysis. F1000Res. 2018;7:1359. https://doi.org/10.12688/f1000research.15995.2.
Punekar YS, Parks D, Joshi M, Kaur S, Evitt L, Chounta, et al. Effectiveness and safety of dolutegravir two-drug regimens in virologically suppressed people living with HIV: a systematic literature review and meta-analysis of real-world evidence. HIV Med. 2021;22:423–33. https://doi.org/10.1111/hiv.13050.
Cahn P, Sierra Madero J, Arribas JR, Antinori A, Ortiz R, Clarke AE, et al. Three-year durable efficacy of dolutegravir plus lamivudine in antiretroviral therapy - naive adults with HIV-1 infection. AIDS. 2022;36:39–48. https://doi.org/10.1097/QAD.0000000000003070.
Maggiolo F, Gulminetti R, Pagnucco L, Digaetano M, Cervo A, Valenti D, et al. Long-term outcome of lamivudine/dolutegravir dual therapy in HIV-infected, virologically suppressed patients. BMC Infect Dis. 2022;22:782. https://doi.org/10.1186/s12879-022-07769-6.
Girard PM, Cabié A, Michelet C, et al. A randomized trial of two-drug versus three-drug tenofovir-containing maintenance regimens in virologically controlled HIV-1 patients. J Anti microb Chemother. 2009;64:126–34.
Pinola M, Lazzarin A, Antinori A, et al. Lopinavir/ ritonavir? tenofovir dual therapy versus lopinavir/ ritonavir-based triple therapy in HIV-infected antiretroviral naïve subjects: the Kalead Study. J Antivir Antiretrovir. 2010;2:56–62. https://doi.org/10.4172/jaa.1000024.
Wassner C, Bradley N, Lee Y. A review and clinical understanding of tenofovir: tenofovir disoproxil fumarate versus tenofovir alafenamide. J Int Assoc Provid AIDS Care. 2020;19:2325958220919231. https://doi.org/10.1177/2325958220919231.
Pilkington V, Hughes SL, Pepperrell T, McCann K, Gotham D, Pozniak AL, et al. Tenofovir alafenamide vs. tenofovir disoproxil fumarate: an updated meta-analysis of 14 894 patients across 14 trials. AIDS. 2020;34:2259–68. https://doi.org/10.1097/QAD.0000000000002699.
Emamzadeh-Fard S, Fard SE. Author, Paydary K, Adherence to anti-retroviral therapy and its determinants in HIV/AIDS patients: a review. Infect Disord Drug Targets. 2012;12:346–56. https://doi.org/10.2174/187152612804142251.
https://eacs.sanfordguide.com/art/virological-failure. Accessed 8 Jan 2023.
Sculier D, Wandeler G, Yerly S, Marinosci A, Stoeckle M, Bernasconi E, et al. Efficacy and safety of dolutegravir plus emtricitabine versus standard ART for the maintenance of HIV-1 suppression: 48-week results of the factorial, randomized, non-inferiority SIMPL’HIV trial. PLoS Med. 2020;17:e1003421. https://doi.org/10.1371/journal.pmed.1003421.
Ward D, Ramgopal M, Riedel DJ, Garris C, Dhir S, Waller J, et al. Real-world experience with dolutegravir-based two-drug regimens. AIDS Res Treat. 2020;7:5923256. https://doi.org/10.1155/2020/5923256.
Atta MG, De Seigneux S, Lucas GM. Clinical pharmacology in HIV therapy. Clin J Am Soc Nephrol. 2019;14:435–44. https://doi.org/10.2215/CJN.02240218.
https://programme.aids2022.org/PAGMaterial/PPT/3733_4919/IAS_ADVANCE_W192POSTER_2022_AQ13JUL22.pdf. Accessed 10 Jan 2023.
Mallon PW, Brunet L, Hsu RK, Fusco JS, Mounzer KC, Prajapati G, et al. Weight gain before and after switch from TDF to TAF in a U.S. cohort study. J Int AIDS Soc. 2021;24:e25702. https://doi.org/10.1002/jia2.25702.
Surial B, Mugglin C, Calmy A, Cavassini M, Günthard HF, Stöckle M, et al. Swiss HIV cohort study. Weight and metabolic changes after switching from tenofovir disoproxil fumarate to tenofovir alafenamide in people living with HIV: a cohort study. Ann Intern Med. 2021;174:758–67. https://doi.org/10.7326/M20-4853.
Darnell J, Jain S, Sun X, Qin H, Reynolds T, Karris MY, et al. Impact of switching to tenofovir alafenamide on weight gain as compared to maintaining a non-tenofovir alafenamide containing regimen. Medicine (Baltimore). 2021;100:e27047. https://doi.org/10.1097/MD.0000000000027047.
Kanters S, Renaud F, Rangaraj A, Zhang K, Limbrick-Oldfield E, Hughes M, et al. Evidence synthesis evaluating body weight gain among people treating HIV with antiretroviral therapy - a systematic literature review and network meta-analysis. E Clin Med. 2022;48:101412. https://doi.org/10.1016/j.eclinm.2022.101412.
Emond B, Rossi C, Rogers R, Lefebvre P, Lafeuille MH, Donga P. Real-world analysis of weight gain and body mass index increase among patients with HIV-1 using antiretroviral regimen containing tenofovir alafenamide, tenofovir disoproxil fumarate, or neither in the United States. J Health Econ Outcomes Res. 2022;9:39–49. https://doi.org/10.36469/jheor.2022.31825.
Wood BR, Huhn GD. Excess weight gain with integrase inhibitors and tenofovir alafenamide: what is the mechanism and does it matter? Open Forum Infect Dis. 2021;8:ofab542.
Petrakis V, Panagopoulos P, Papachristou S, Papanas N, Terzi I, Trypsianis G, et al. Tenofovir alafenamide fumarate therapy for HIV treatment: cardiometabolic and renal safety. AIDS Res Hum Retroviruses. 2020;36:697–702. https://doi.org/10.1089/AID.2019.0280.10.1093/ofid/ofab542.
Acknowledgements
We are grateful to Dr Kasra Kolahdouzan MD, Dr Hossein Daliry MD, and all personnel of the Voluntary Counseling and Testing Center for their kind help and cooperation. The authors acknowledge with the support of Tadbir Kalaye Jam (Tekaje) Company for providing a part of tenofovir alafenamide.
Funding
This study was supported by Tehran University of Medical Sciences (IR.TUMS.TIPS.REC.1398.103).
Author information
Authors and Affiliations
Contributions
LA, HK, GA—conceptualized and implemented the study, GA wrote the first draft; SASA conducted statistical analyses, GA, MFA, SADM, MH—collected data; LA, SASA, HK—reviewed and provided intellectual and editorial content. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Authors have no any to declare regarding this submission.
Ethics approval and consent to participate
The study was approved by the Ethics Committees of Tehran University of Medical Sciences (IR.TUMS.TIPS.REC.1398.087).
Consent for publication
Not applicable.
Conflict of interest
The authors have no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Alavian, G., Abbasian, L., Khalili, H. et al. Tenofovir alafenamide plus dolutegravir as a switch strategy in HIV-infected patients: a pilot randomized controlled trial. DARU J Pharm Sci 31, 145–153 (2023). https://doi.org/10.1007/s40199-023-00470-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40199-023-00470-2