Abstract
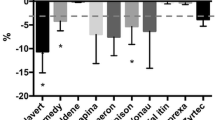
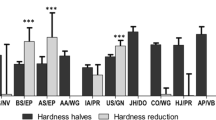
Tablet splitting is widely practiced worldwide. Several studies have considered weight variation of split tablets as a mean of estimating drug content uniformity but the analysis of their drug content and physical factors that may affect splitting are limited. The aim of this study is to evaluate the impact of manufacturing parameters and splitting on content and weight uniformity of atenolol tablets. Atenolol tablets (100 and 50 mg) were prepared under the same manufacturing conditions and using the same excipients. The obtained tablets were checked for hardness, weight, and disintegration. The weight and the content of the two strength atenolol tablets after splitting into two halves were evaluated. Atenolol tablets (100 mg) showed higher values of hardness, disintegration time and diameter than atenolol tablets (50 mg). Atenolol tablets (100 mg) passed both weight and content uniformity while atenolol tablets (50 mg) failed these tests. Half tablet weight appears to be directly correlated with its drug content. Manufacturers should investigate physical factors such as tablet hardness, diameter, and disintegration time that may play an important role in achieving both weight and content uniformity in the resultant tablet halves.
Similar content being viewed by others
References
Cook TJ, Edwards S, Gyemah C, Shah M, Shah I, Fox T (2004) Variability in tablet fragment weights when splitting unscored cyclobenzaprine 10 mg tablets. Am Pharm Assoc (2003) 44:583–586
Duncan MC, Castle SS, Streetman DS (2002) Effect of tablet splitting on serum cholesterol concentrations. Ann Pharmacother 36:205–209
Emilien G, Maloteaux JM (1998) Current therapeutic uses and potential of beta-adrenoceptor agonists and antagonists. Eur J Clin Pharmacol 53:389–404
European Pharmacopoeia (2002) Suppl 4.1, Council of Europe; European Directorate for the Quality of Medicine, Strasbourg, Tablets Monograph 0478
European Pharmacopoeia (2008) Suppl 6.4, Council of Europe; European Directorate for the Quality of Medicine, Strasbourg, Tablets Monograph 0478
Fawell NG, Cookson TL, Scranton SS (1999) Relationship between tablet splitting and compliance, drug acquisition cost, and patient acceptance. Am J Health Syst Pharm 56:2542–2545
Gee M, Hasson NK, Hahn T, Ryono R (2002) Effects of a tablet-splitting program in patients taking HMG-Co A reductase inhibitors: analysis of clinical effects, patient satisfaction, compliance, and cost avoidance. J Managed Care Pharm 8:453–458
Green G, Berg C, Polli J, Barends D (2009) Pharmacopeial standards for the subdivision characteristics of scored tablets. Pharmacopeial Forum 35:1598–1603
Hill SW, Varker AS, Karlage K, Myrdal PB (2009) Analysis of drug content and weight uniformity for half-tablets of 6 commonly split medications. J Manag Care Pharm 15:253–261
Katori N, Aoyagi N, Kojima S (2001) The study of the applicability of content uniformity and weight variation test- the state of commercial tablets and capsules in Japan. Chem Pharm Bull 49:1412–1419
Kristensen HG, Jorgensen GH, Moller-Sonnergaard JJ (1995) Mass uniformity of tablets broken by hand. Pharmeuropa 7:298–302
Polli JE, Kim S, Martin BR (2003) Weight uniformity of split tablets required by a Veterans Affairs policy. J Manag Care Pharm 9:401–407
Rindone JP (2000) Evaluation of tablet-splitting in patients taking lisinopril for hypertension. J Clin Outcomes Manage 7:22–24
Teng J, Song CK, Williams RL, Polli JE (2002) Lack of medication dose uniformity in commonly split tablets. J Am Pharm Assoc (Wash) 42:195–199
United States Pharmacopeial Convention (eds) (2005) The United States Pharmacopeia, 29th Rev., and the National Formulary, 24th edn. Rockville, p 212
Van Santen E, Barends DM, Frijlink HW (2002) Breaking of scored tablets: a review. Eur J Pharm Biopharm 53:139–145
Vranić E, Uzunović A (2007) Influence of tablet splitting on content uniformity of lisinopril/hydrochlorthiazide tablets. Bosn J Basic Med Sci 7:328–334
Zaid AN, Ghosh AA (2011) Compliance of scored tablet halves produced by Palestinian Pharmaceutical Companies with the new European Pharmacopoeia requirements. Arch Pharm Res 34:1183–1189
Zaid AN, Abu Ghosh A, Kittana N (2010) Weight uniformity of scored tablet halves manufactured by Palestinian pharmaceutical companies. Intern J Pharm Compd 14:257–260
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zaid, A.N., Al-Ramahi, R., Ghoush, A.A. et al. Influence of physical factors on tablet splitting, weight and content uniformity of atenolol tablets. Journal of Pharmaceutical Investigation 42, 229–234 (2012). https://doi.org/10.1007/s40005-012-0030-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40005-012-0030-7