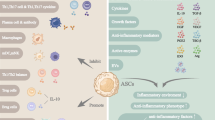
Abstract
Background:
Adipose-derived stem cells (ADSCs) exert immunomodulatory effects in the treatment of transplant rejection. This study aimed to evaluate the effects of ADSCs on the skin graft survival in a human-to-rat xenograft transplantation model and to compare single and multiple injections of ADSCs.
Methods:
Full-thickness human skin xenografts were transplanted into the backs of Sprague–Dawley rats. The rats were injected subcutaneously on postoperative days 0, 3, and 5. The injections were as follows: triple injections of phosphate-buffered saline (PBS group), a single injection of ADSCs and double injections of PBS (ADSC × 1 group), and triple injections of ADSCs (ADSC × 3 group). The immunomodulatory effects of ADSCs on human skin xenografts were assessed.
Results:
Triple injections of ADSCs considerably delayed cell-mediated xenograft rejection compared with the PBS and ADSC × 1 groups. The vascularization and collagen type 1–3 ratios in the ADSC × 3 group were significantly higher than those in the other groups. In addition, intragraft infiltration of CD3-, CD4-, CD8-, and CD68-positive cells was reduced in the ADSC × 3 group. Furthermore, in the ADSC × 3 group, the expression levels of proinflammatory cytokine interferon-gamma (IFN-γ) were decreased and immunosuppressive prostaglandin E synthase (PGES) was increased in the xenograft and lymph node samples.
Conclusion:
This study presented that triple injections of ADSCs appeared to be superior to a single injection in suppressing cell-mediated xenograft rejection. The immunomodulatory effects of ADSCs are associated with the downregulation of IFN-γ and upregulation of PGES in skin xenografts and lymph nodes.







Similar content being viewed by others
References
Carrier AN, Verma A, Mohiuddin M, Pascual M, Muller YD, Longchamp A, et al. Xenotransplantation: a new era. Front Immunol. 2022;13:900594.
Siemionow M, Nasir S. Immunologic responses in vascularized and nonvascularized skin allografts. J Reconstr Microsurg. 2008;24:497–505.
Simeonovic CJ, Townsend MJ, Karupiah G, Wilson JD, Zarb JC, Mann DA, et al. Analysis of the Th1/Th2 paradigm in transplantation: interferon-gamma deficiency converts Th1-type proislet allograft rejection to a Th2-type xenograft-like response. Cell Transplant. 1999;8:365–73.
Zhao Y, Xiong W, Yang T, Prall A, Baxter BT, Langnas AN. Xenogeneic skin graft rejection in M-CSF/macrophage deficient osteopetrotic mice. Xenotransplantation. 2003;10:232–9.
Yamamoto T, Iwase H, King TW, Hara H, Cooper DKC. Skin xenotransplantation: historical review and clinical potential. Burns. 2018;44:1738–49.
Tomita Y, Uchida T, Zhang QW, Shimizu I, Iwai T, Yoshikawa M, et al. Human skin xenograft rejection in CD45 exon-6 knockout mice: the implication of involvement of a direct pathway. Surg Today. 2000;30:816–20.
Sykes M. Immunobiology of transplantation. FASEB J. 1996;10:721–30.
Gardner CR. The pharmacology of immunosuppressant drugs in skin transplant rejection in mice and other rodents. Gen Pharmacol. 1995;26:245–71.
Yañez R, Lamana ML, Garcia-Castro J, Colmenero I, Ramirez M, Bueren JA. Adipose tissue-derived mesenchymal stem cells have in vivo immunosuppressive properties applicable for the control of the graft-versus-host disease. Stem Cells. 2006;24:2582–91.
Castro-Manrreza ME, Montesinos JJ. Immunoregulation by mesenchymal stem cells: biological aspects and clinical applications. J Immunol Res. 2015;2015:394917.
Puissant B, Barreau C, Bourin P, Clavel C, Corre J, Bousquet C, et al. Immunomodulatory effect of human adipose tissue-derived adult stem cells: comparison with bone marrow mesenchymal stem cells. Br J Haematol. 2005;129:118–29.
Glennie S, Soeiro I, Dyson PJ, Lam EW, Dazzi F. Bone marrow mesenchymal stem cells induce division arrest anergy of activated T cells. Blood. 2005;105:2821–7.
Corcione A, Benvenuto F, Ferretti E, Giunti D, Cappiello V, Cazzanti F, et al. Human mesenchymal stem cells modulate B-cell functions. Blood. 2006;107:367–72.
Chen CC, Chen RF, Shao JS, Li YT, Wang YC, Brandacher G, et al. Adipose-derived stromal cells modulating composite allotransplant survival is correlated with B cell regulation in a rodent hind-limb allotransplantation model. Stem Cell Res Ther. 2020;11:478.
Zomer HD, Jeremias TDS, Ratner B, Trentin AG. Mesenchymal stromal cells from dermal and adipose tissues induce macrophage polarization to a pro-repair phenotype and improve skin wound healing. Cytotherapy. 2020;22:247–60.
Burchell JT, Strickland DH, Stumbles PA. The role of dendritic cells and regulatory T cells in the regulation of allergic asthma. Pharmacol Ther. 2010;125:1–10.
Leto Barone AA, Khalifian S, Lee WP, Brandacher G. Immunomodulatory effects of adipose-derived stem cells: Fact or fiction? Biomed Res Int. 2013;2013:383685.
Cui L, Yin S, Liu W, Li N, Zhang W, Cao Y. Expanded adipose-derived stem cells suppress mixed lymphocyte reaction by secretion of prostaglandin E2. Tissue Eng. 2007;13:1185–95.
An JH, Song WJ, Li Q, Kim SM, Yang JI, Ryu MO, et al. Prostaglandin E2 secreted from feline adipose tissue-derived mesenchymal stem cells alleviate DSS-induced colitis by increasing regulatory T cells in mice. BMC Vet Res. 2018;14:354.
DelaRosa O, Lombardo E, Beraza A, Mancheño-Corvo P, Ramirez C, Menta R, et al. Requirement of IFN-gamma-mediated indoleamine 2,3-dioxygenase expression in the modulation of lymphocyte proliferation by human adipose-derived stem cells. Tissue Eng Part A. 2009;15:2795–806.
Ren G, Zhang L, Zhao X, Xu G, Zhang Y, Roberts AI, et al. Mesenchymal stem cell-mediated immunosuppression occurs via concerted action of chemokines and nitric oxide. Cell Stem Cell. 2008;2:141–50.
Melief SM, Zwaginga JJ, Fibbe WE, Roelofs H. Adipose tissue-derived multipotent stromal cells have a higher immunomodulatory capacity than their bone marrow-derived counterparts. Stem Cell Transl Med. 2013;2:455–63.
Hoogduijn MJ, Betjes MG, Baan CC. Mesenchymal stromal cells for organ transplantation: Different sources and unique characteristics? Curr Opin Organ Transplant. 2014;19:41–6.
Lee SM, Lee SC, Kim SJ. Contribution of human adipose tissue-derived stem cells and the secretome to the skin allograft survival in mice. J Surg Res. 2014;188:280–9.
Larocca RA, Moraes-Vieira PM, Bassi EJ, Semedo P, de Almeida DC, da Silva MB, et al. Adipose tissue-derived mesenchymal stem cells increase skin allograft survival and inhibit Th-17 immune response. PLoS One. 2013;8:e76396.
Davis TA, Anam K, Lazdun Y, Gimble JM, Elster EA. Adipose-derived stromal cells promote allograft tolerance induction. Stem Cells Transl Med. 2014;3:1444–50.
Rosenberg AS, Singer A. Cellular basis of skin allograft rejection: an in vivo model of immune-mediated tissue destruction. Annu Rev Immunol. 1992;10:333–58.
Halloran PF, Einecke G, Sikosana MLN, Madill-Thomsen K. The biology and molecular basis of organ transplant rejection. Handb Exp Pharmacol. 2022;272:1–26.
Zhou J, He W, Luo G, Wu J. Fundamental immunology of skin transplantation and key strategies for tolerance induction. Arch Immunol Ther Exp (Warsz). 2013;61:397–405.
Wang J, Hao H, Huang H, Chen D, Han Y, Han W. The Effect of Adipose-Derived Stem Cells on Full-Thickness Skin Grafts. Biomed Res Int. 2016;2016:1464725.
Zografou A, Tsigris C, Papadopoulos O, Kavantzas N, Patsouris E, Donta I, et al. Improvement of skin-graft survival after autologous transplantation of adipose-derived stem cells in rats. J Plast Reconstr Aesthet Surg. 2011;64:1647–56.
Vidor SB, Terraciano PB, Valente FS, Rolim VM, Kuhl CP, Ayres LS, et al. Adipose-derived stem cells improve full-thickness skin grafts in a rat model. Res Vet Sci. 2018;118:336–44.
Jeon S, Ha JH, Kim I, Bae J, Kim SW. The immunomodulatory effect of adipose-derived stem cells in xenograft transplantation model. Transplant Proc. 2022;54:2388–95.
Duijvestein M, Vos AC, Roelofs H, Wildenberg ME, Wendrich BB, Verspaget HW, et al. Autologous bone marrow-derived mesenchymal stromal cell treatment for refractory luminal Crohn’s disease: results of a phase I study. Gut. 2010;59:1662–9.
Gao F, Chiu SM, Motan DA, Zhang Z, Chen L, Ji HL, et al. Mesenchymal stem cells and immunomodulation: current status and future prospects. Cell Death Dis. 2016;7:e2062.
Keating A. Mesenchymal stromal cells: new directions. Cell Stem Cell. 2012;10:709–16.
Yang C, Wang G, Ma F, Yu B, Chen F, Yang J, et al. Repeated injections of human umbilical cord blood-derived mesenchymal stem cells significantly promotes functional recovery in rabbits with spinal cord injury of two noncontinuous segments. Stem Cell Res Ther. 2018;9:136.
Mahmoud EE, Adachi N, Mawas AS, Deie M, Ochi M. Multiple intra-articular injections of allogeneic bone marrow-derived stem cells potentially improve knee lesions resulting from surgically induced osteoarthritis: an animal study. Bone Joint J. 2019;101-B:824–31.
Plock JA, Schnider JT, Schweizer R, Zhang W, Tsuji W, Waldner M, et al. The influence of timing and frequency of adipose-derived mesenchymal stem cell therapy on immunomodulation outcomes after vascularized composite allotransplantation. Transplantation. 2017;101:e1–11.
Zhou X, Ning K, Ling B, Chen X, Cheng H, Lu B, et al. Multiple injections of autologous adipose-derived stem cells accelerate the burn wound healing process and promote blood vessel regeneration in a rat model. Stem Cells Dev. 2019;28:1463–72.
Kim H, Hyun MR, Kim SW. The effect of adipose-derived stem cells on wound healing: comparison of methods of application. Stem Cells Int. 2019;2019:2745640.
Cendales LC, Kanitakis J, Schneeberger S, Burns C, Ruiz P, Landin L, et al. The Banff 2007 working classification of skin-containing composite tissue allograft pathology. Am J Transplant. 2008;8:1396–400.
Luo Y, Yi X, Liang T, Jiang S, He R, Hu Y, et al. Autograft microskin combined with adipose-derived stem cell enhances wound healing in a full-thickness skin defect mouse model. Stem Cell Res Ther. 2019;10:279.
Benichou G, Yamada Y, Yun SH, Lin C, Fray M, Tocco G. Immune recognition and rejection of allogeneic skin grafts. Immunotherapy. 2011;3:757–70.
Fairchild RL. The Yin and Yang of IFN-gamma in allograft rejection. Am J Transplant. 2003;3:913–4.
Hidalgo LG, Halloran PF. Role of IFN-gamma in allograft rejection. Crit Rev Immunol. 2002;22:317–49.
Moss MI, Pauli M, Moreau JM, Cohen JN, Rosenblum MD, Lowe MM. Xenograft skin model to manipulate human immune responses in vivo. J Vis Exp. 2022. https://doi.org/10.3791/64040
Manning DD, Reed ND, Shaffer CF. Maintenance of skin xenografts of widely divergent phylogenetic origin of congenitally athymic (nude) mice. J Exp Med. 1973;138:488–94.
Al-Ghadban S, Bunnell BA. Adipose tissue-derived stem cells: immunomodulatory effects and therapeutic potential. Physiology (Bethesda). 2020;35:125–33.
Wolbank S, Peterbauer A, Fahrner M, Hennerbichler S, van Griensven M, Stadler G, et al. Dose-dependent immunomodulatory effect of human stem cells from amniotic membrane: a comparison with human mesenchymal stem cells from adipose tissue. Tissue Eng. 2007;13:1173–83.
Hu JL, Kim BJ, Yu NH, Kwon ST. Impact of injection frequency of adipose-derived stem cells on allogeneic skin graft survival outcomes in mice. Cell Transplant. 2021;30:9636897211041966.
Lorenzi W, Goncalves FDC, Schneider N, Silva EF, Visioli F, Paz AH, et al. Repeated systemic administration of adipose tissue-derived mesenchymal stem cells prevents tracheal obliteration in a murine model of bronchiolitis obliterans. Biotechnol Lett. 2017;39:1269–77.
Famulski KS, Sis B, Billesberger L, Halloran PF. Interferon-gamma and donor MHC class I control alternative macrophage activation and activin expression in rejecting kidney allografts: a shift in the Th1–Th2 paradigm. Am J Transplant. 2008;8:547–56.
Zhang YJ, Wang YC, Wu GF, Zhang W, Wang XJ, Cai WX, et al. Prolonged skin grafts survival time by IFN-gamma in allogeneic skin transplantation model during acute rejection through IFN-gamma/STAT3/IDO pathway in epidermal layer. Biochem Biophys Res Commun. 2018;496:436–42.
Wang H, DeVries ME, Deng S, Khandaker MH, Pickering JG, Chow LH, et al. The axis of interleukin 12 and gamma interferon regulates acute vascular xenogeneic rejection. Nat Med. 2000;6:549–55.
Sultan P, Murray AG, McNiff JM, Lorber MI, Askenase PW, Bothwell AL, et al. Pig but not human interferon-gamma initiates human cell-mediated rejection of pig tissue in vivo. Proc Natl Acad Sci U S A. 1997;94:8767–72.
Bui TM, Wiesolek HL, Sumagin R. ICAM-1: a master regulator of cellular responses in inflammation, injury resolution, and tumorigenesis. J Leukoc Biol. 2020;108:787–99.
Short SM, Paasch BD, Turner JH, Weiner N, Daugherty AL, Mrsny RJ. Percutaneous absorption of biologically-active interferon-gamma in a human skin graft-nude mouse model. Pharm Res. 1996;13:1020–7.
Zafranskaya M, Nizheharodava D, Yurkevich M, Ivanchik G, Demidchik Y, Kozhukh H, et al. PGE2 contributes to in vitro MSC-mediated inhibition of non-specific and antigen-specific T cell proliferation in MS patients. Scand J Immunol. 2013;78:455–62.
Yanez R, Oviedo A, Aldea M, Bueren JA, Lamana ML. Prostaglandin E2 plays a key role in the immunosuppressive properties of adipose and bone marrow tissue-derived mesenchymal stromal cells. Exp Cell Res. 2010;316:3109–23.
Ghannam S, Pene J, Moquet-Torcy G, Jorgensen C, Yssel H. Mesenchymal stem cells inhibit human Th17 cell differentiation and function and induce a T regulatory cell phenotype. J Immunol. 2010;185:302–12.
Bassi EJ, Moraes-Vieira PM, Moreira-Sa CS, Almeida DC, Vieira LM, Cunha CS, et al. Immune regulatory properties of allogeneic adipose-derived mesenchymal stem cells in the treatment of experimental autoimmune diabetes. Diabetes. 2012;61:2534–45.
Ma S, Xie N, Li W, Yuan B, Shi Y, Wang Y. Immunobiology of mesenchymal stem cells. Cell Death Differ. 2014;21:216–25.
Acknowledgements
This research was supported and funded by Seoul National University Hospital (Grant Number: 0420203100).
Author information
Authors and Affiliations
Contributions
S.W.K. and J.H.C. supervised the study; S.J. designed and performed the experiments with assistance from I.K., Y.R.N, K.Y.H, H.C., S.H.K., and Y.J.J.; S.J. and I.K. wrote the manuscript; and all authors commented on the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest. None of the authors have a financial interest in any of the products, devices, or drugs mentioned in this manuscript.
Ethical statement
All animal experiments were approved by the Institutional Animal Care and Use Committee of the Seoul National University Hospital (SNUH-IACUC no. 21-0143-S1A0). Informed consent was waived after approval by the Institutional Review Board of Seoul National University Hospital (no. 2021-3273).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
13770_2023_552_MOESM2_ESM.pdf
Supplementary Figure S2. Characterization of adipose-derived stem cells. A Representative images of cell morphology under light microscope. Lower (left) and higher (right) magnification. Scale bar, 100 µm. C Flow cytometric analysis for mesenchymal (CD73 and CD90) and hematopoietic (CD11b and CD34) cell markers (PDF 707 KB)
13770_2023_552_MOESM3_ESM.pdf
Supplementary Figure S3. Long-term results of the skin xenografts until postoperative 8 weeks. A Representative images of the xenografts on POD 21, 28 and 56. Scale bar, 1 mm. B The human skin xenografts firmly attached to the rat tissue as dry scabs 8 weeks post operation (PDF 359 KB)
13770_2023_552_MOESM4_ESM.pdf
Supplementary Figure S4. Long-term histological results of the skin xenografts until 8 weeks. A Representative images of hematoxylin and eosin (H&E) and Masson’s trichome (MT) staining in low-power field (LPF) 8 weeks post operation. There were few noticeable differences between the PBS, ADSC × 1, and ADSC × 3 groups at the 8-week time point. Scale bar, 1 mm. B Representative images showing the contact layer between the human xenograft and rat tissue 8 weeks post operation. Regeneration of the rat-derived epithelium was limited to the peripheral rim of the xenografts, which suggests that the grafts did not fully integrate with the host tissue. Scale bar, 3 mm and 1 mm (for upper and lower row, respectively) (PDF 3755 KB)
13770_2023_552_MOESM5_ESM.pdf
Supplementary Figure S5. Immunohistochemistry staining for proliferating cell nuclear antigen (PCNA) in the epithelium of the skin xenografts on POD 10 and 14. PCNA-positive cells decreased with the advancement of rejection, accompanied by disorganization of the epidermis, in the PBS group followed by the ADSC × 1 and ADSC × 3 groups. Scale bar, 50 μm. PBS, phosphate-buffered saline; ADSC, adipose-derived stem cell; POD, postoperative day (PDF 328 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jeon, S., Kim, I., Na, Y.R. et al. Multiple Injections of Adipose-Derived Stem Cells Improve Graft Survival in Human-to-Rat Skin Xenotransplantation through Immune Modulation. Tissue Eng Regen Med 20, 905–919 (2023). https://doi.org/10.1007/s13770-023-00552-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13770-023-00552-x