Abstract
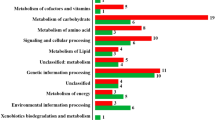
Leptospirosis, caused by the pathogenic Leptospira, is an emerging zoonotic disease affecting over one million people annually. The existence of a large number of serovars, different reservoir hosts, and common disease symptoms account for the difficulty in early diagnosis, prophylaxis, and treatment. Post-translational modification plays a significant regulatory role in both eukaryotic and prokaryotic organisms. Therefore, the study of post-translational modification may help in better understanding the pathogenesis of the bacterial disease. Acetylation at lysine residue was found to be involved in regulating bacterial pathogenesis. This study aims to identify protein lysine acetylation patterns among groups of pathogenic and saprophytic species of Leptospira, by screening the leptospiral proteome using a robust proteomics approach. In this study, a total of 15, 78,796 acetylated proteins with 83, 65,945 acetylation sites were identified among 469 strains of Leptospira to predict the pathogenesis pattern and signature peptide sequence, which was conserved among pathogenic Leptospira species, that can be used as a novel vaccine candidate. A similar pattern of acetylation was observed among the pathogenic and intermediate groups while different in the saprophytic group of Leptospira. Consequently, a common signature peptide was observed among pathogenic strains of Leptospira. Acetylated proteins were found to be primarily involved in metabolic processes. As a result, this is the first study to analyze proteome-wise strain specific lysine acetylation of Leptospiral proteins which may constitute a valuable resource for in-depth studies of the impact of lysine acetylation in the pathogenesis of Leptospira.



Similar content being viewed by others
Data availability statement
Not available.
References
Adler B, Lo M, Seemann T, Murray GL (2011) Pathogenesis of leptospirosis: The influence of genomics. Vet Microbiol 153(1–2):73–81. https://doi.org/10.1016/j.vetmic.2011.02.055
Arratia R, Goldstein L, Gordon L (2010) Poisson approximation and the Chen-Stein method. Statistics (ber) 5(4):403–424
Barbosa AS, Isaac L (2020) Strategies used by Leptospira spirochetes to evade the host complement system. FEBS Lett 594(16):2633–2644. https://doi.org/10.1002/1873-3468.13768
Beltrao P, Bork P, Krogan NJ, van Noort V (2013) Evolution and functional cross-talk of protein post-translational modifications. MsbEmbopressOrg 2013:1–13. https://doi.org/10.1093/ckj/sft027
Beriwal S, Padhiyar N, Bhatt D et al (2018) LeptoDB: an integrated database of genomics and proteomics resource of Leptospira. Database 2018(2018):1–10. https://doi.org/10.1093/database/bay057
Bharti AR, Nally JE, Ricaldi JN et al (2003) Leptospirosis: a zoonotic disease of global importance. Lancet Infect Dis 3:757–771. https://doi.org/10.1016/S1473-3099(03)00830-2
Bonham-Carter O, Thapa I, From S, Bastola D (2017) A study of bias and increasing organismal complexity from their post-translational modifications and reaction site interplays. Brief Bioinform 18(1):69–84. https://doi.org/10.1093/bib/bbv111
Boulanger MJ, Eshghi A, Witchell TD et al (2014) Post-translational Modification of LipL32 during Leptospira interrogans Infection. PLoS Negl Trop Dis 8(10):e3280. https://doi.org/10.1371/journal.pntd.0003280
Bulach DM, Haake DA, Adler B, Lo M, Matsunaga J, Zuerner RL (2007) Response of leptospira interrogans to physiologic osmolarity: relevance in signaling the environment-to-host transition. Infect Immun 75(6):2864–2874. https://doi.org/10.1128/iai.01619-06
Caimano MJ, Grassmann AA, Nally JE et al (2014) A Model system for studying the transcriptomic and physiological changes associated with mammalian host-adaptation by Leptospira interrogans Serovar Copenhageni. PLoS Pathog 10(3):e1004004. https://doi.org/10.1371/journal.ppat.1004004
Cain JA, Solis N, Cordwell SJ (2014) Beyond gene expression: the impact of protein post-translational modifications in bacteria. J Proteomics 97:265–286. https://doi.org/10.1016/j.jprot.2013.08.012
Choudhary C, Kumar C, Gnad F et al (2012) Lysine acetylation targets protein complexes and co-regulates major cellular functions. Science 325:834–840. https://doi.org/10.1126/science.1175371
Costa F, Hagan JE, Calcagno J et al (2015) Global morbidity and mortality of leptospirosis: a systematic review. PLoS Negl Trop Dis 9(9):e0003898. https://doi.org/10.1371/journal.pntd.0003898
Crosby HA, Pelletier DA, Hurst GB, Escalante-Semerena JC (2012) System-wide studies of N-lysine acetylation in Rhodopseudomonas palustris reveal substrate specificity of protein acetyltransferases. J Biol Chem 287(19):15590–15601. https://doi.org/10.1074/jbc.M112.352104
De Lose Reyes JO, Bragg SL, Plikaytis B et al (2017) Asymptomatic infection and risk factors for leptospirosis in Nicaragua. Am J Trop Med Hyg 63(5):249–254. https://doi.org/10.4269/ajtmh.2000.63.249
Deng W, Wang C, Zhang Y et al (2016) GPS-PAIL: prediction of lysine acetyltransferase-specific modification sites from protein sequences. Sci Rep 6:1–10. https://doi.org/10.1038/srep39787
Dey P (2014) Stein-Chen method for poisson approximation. vol. 414, pp. 2013–2014
Eshghi A, Cullen PA, Cowen L, Zuerner RL, Cameron CE (2009) Global proteome analysis of Leptospira interrogans. J Proteome Res 8(10):4564–4578. https://doi.org/10.1021/pr9004597
Eshghi A, Pinne M, Haake DA, Zuerner RL, Frank A, Cameron CE (2012) Methylation and in vivo expression of the surfaceexposed Leptospira interrogans outer-membrane protein OmpL32. Microbiology 158(3):622–635. https://doi.org/10.1099/mic.0.054767-0
Grangeasse C, Stülke J, Mijakovic I (2015) Regulatory potential of post-translational modifications in bacteria. Front Microbiol 6:2014–2015. https://doi.org/10.3389/fmicb.2015.00500
Hou T et al (2014) LAceP: lysine acetylation site prediction using logistic regression classifiers. PLoS ONE 9:e89575
Huang DW, Sherman BT, Lempicki RA (2009a) Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc 4(1):44–57. https://doi.org/10.1038/nprot.2008.211
Huang DW, Sherman BT, Lempicki RA (2009b) Bioinformatics enrichment tools: Paths toward the comprehensive functional analysis of large gene lists. Nucleic Acids Res 37(1):1–13. https://doi.org/10.1093/nar/gkn923
Huang L, Zhu W-G, Li T et al (2011) Characterization and prediction of lysine (K)-acetyl-transferase specific acetylation sites. Mol Cell Proteomics 11(1):M111.011080. https://doi.org/10.1074/mcp.m111.011080
Jean Beltran PM, Federspiel JD, Sheng X, Cristea IM (2017) Proteomics and integrative omic approaches for understanding host–pathogen interactions and infectious diseases. Mol Syst Biol 13(3):922. https://doi.org/10.15252/msb.20167062
Jers C, Ravikumar V, Lezyk M et al (2018) The global acetylome of the human pathogen Vibrio cholerae V52 reveals lysine acetylation of major transcriptional regulators. Front Cell Infect Microbiol 7:1–13. https://doi.org/10.3389/fcimb.2017.00537
Karve TM, Cheema AK (2011) Small changes huge impact: the role of protein posttranslational modifications in cellular homeostasis and disease. J Amino Acids 2011:1–13. https://doi.org/10.4061/2011/207691
Ko AI, Galvão Reis M, Ribeiro Dourado CM, Johnson WD, Riley LW (1999) Urban epidemic of severe leptospirosis in Brazil Salvador Leptospirosis Study Group. Lancet (london, England) 354(9181):820–825
Kumar S, Lata KS, Sharma P, Bhairappanavar SB, Soni S, Das J (2019) Inferring pathogen-host interactions between Leptospira interrogans and Homo sapiens using network theory. Sci Rep 9(1):1–17. https://doi.org/10.1038/s41598-018-38329-1
Lata KS, Vaghasia V, Bhairappanavar SB et al (2020) Whole genome sequencing and de novo assembly of three virulent Indian isolates of Leptospira. Infect Genet Evol 85:104579. https://doi.org/10.1016/j.meegid.2020.104579
Lata KS, Kumar S, Vindal V, Patel S, Das J (2022) A core and pan gene map of Leptospira genus and its interactions with human host. Microb Pathog 162:105347. https://doi.org/10.1016/j.micpath.2021.105347
Lee KK, Workman JL (2007) Histone acetyltransferase complexes : one size doesn’t fit all. Nat Rev Mol Cell Biol 8:284–295. https://doi.org/10.1038/nrm2145
Lee TY et al (2010) N-Ace: using solvent accessibility and physicochemical properties to identify protein N-acetylation sites. J Comput Chem 31:2759–2771
Lehmann J, Matthias M, Vinetz J, Fouts D (2014) Leptospiral Pathogenomics. Pathogens 3(2):280–308. https://doi.org/10.3390/pathogens3020280
Levett PN (2001) Leptospirosis. Clin Microbiol 14(2):296–326. https://doi.org/10.1128/CMR.14.2.296
Li S et al (2009) Improved prediction of lysine acetylation by support vector machines. Protein Pept Lett 16:977–983
Li T et al (2012) Characterization and prediction of lysine (K)-acetyl-transferase specific acetylation sites. Mol Cell Proteom 11:M111011080
Li Y et al (2014) Accurate in silico identification of species-specific acetylation sites by integrating protein sequence-derived and functional features. Sci Reports 4:5765
Lu CT, Lee TY, Chen YJ (2014) An intelligent system for identifying acetylated lysine on histones and nonhistone proteins. BioMed Res Int 2014:528650
Luu J, Carabetta VJ (2021) Contribution of N ε -lysine acetylation towards regulation of bacterial pathogenesis. mSystems. https://doi.org/10.1128/msystems.00422-21
O’Shea JP, Chou MF, Quader SA, Ryan JK, Church GM, Schwartz D (2013) PLogo: a probabilistic approach to visualizing sequence motifs. Nat Methods 10(12):1211–1212. https://doi.org/10.1038/nmeth.2646
Okada AK, Teranishi K, Ambroso MR et al (2021) Lysine acetylation regulates the interaction between proteins and membranes. Nat Commun 12(1):1–12
Okanishi H, Kim K, Masui R, Kuramitsu S (2013) Acetylome with structural mapping reveals the significance of lysine acetylation in Thermus thermophilus. J Proteome Res 12(9):3952–3968. https://doi.org/10.1021/pr400245k
Ouidir T, Cosette P, Jouenne T, Hardouin J (2015) Proteomic profiling of lysine acetylation in Pseudomonas aeruginosa reveals the diversity of acetylated proteins. Proteomics 15(13):2152–2157. https://doi.org/10.1002/pmic.201500056
Ouidir T, Kentache T, Hardouin J (2016) Protein lysine acetylation in bacteria: current state of the art. Proteomics 16(2):301–309. https://doi.org/10.1002/pmic.201500258
Pejaver V, Hsu WL, Xin F, Dunker AK, Uversky VN, Radivojac P (2014) The structural and functional signatures of proteins that undergo multiple events of post-translational modification. Protein Sci 23(8):1077–1093. https://doi.org/10.1002/pro.2494
Picardeau M (2015) Leptospira and Leptospirosis. Springer, Berlin
Plank R, Dean D (2000) Overview of the epidemiology, microbiology, and pathogenesis of Leptospira spp. in humans. Microbes Infect 2(10):1265–1276. https://doi.org/10.1016/S1286-4579(00)01280-6
Qiu WR, Sun BQ, Xiao X, Xu ZC, Chou KC (2016) iPTM-mLys: identifying multiple lysine PTM sites and their different types. Bioinformatics 32(20):3116–3123
Ravikumar V, Jers C, Mijakovic I (2015) Elucidating host-pathogen interactions based on post-translational modifications using proteomics approaches. Front Microbiol 6:1–7. https://doi.org/10.3389/fmicb.2015.01312
Rosa PA, Stewart PE, Sturdevant DE, Carroll JA, Olano LR (2015) Multiple posttranslational modifications of leptospira biflexa proteins as revealed by proteomic analysis. Appl Environ Microbiol 82(4):1183–1195. https://doi.org/10.1128/aem.03056-15
Shao J et al (2012) Systematic analysis of human lysine acetylation proteins and accurate prediction of human lysine acetylation through bi-relative adapted binomial score Bayes feature representation. Mol bioSystems 8:2964–2973
Sheng Q-H, Dai J, Li Y-X et al (2009) High-coverage proteome analysis reveals the first insight of protein modification systems in the pathogenic spirochete Leptospira interrogans. Cell Res 20(2):197–210. https://doi.org/10.1038/cr.2009.127
Shi SP et al (2012a) PLMLA: prediction of lysine methylation and lysine acetylation by combining multiple features. Mol bioSystems 8:1520–1527
Shi SP et al (2012b) A method to distinguish between lysine acetylation and lysine methylation from protein sequences. J Theor Biol 310:223–230
Soo ZMP, Khan NA, Siddiqui R (2020) Leptospirosis: Increasing importance in developing countries. Acta Trop. https://doi.org/10.1016/j.actatropica.2019.105183
Suo SB et al (2012) Position-specific analysis and prediction for protein lysine acetylation based on multiple features. PLoS ONE 7:e49108
Suo SB et al (2013) Proteome-wide analysis of amino acid variations that influence protein lysine acetylation. J Proteome Res 12:949–958
Vaghasia V, Lata KS, Das J et al (2018) Exploring Leptospiral proteomes to identify potential candidates for vaccine design against Leptospirosis using an immunoinformatics approach. Sci Rep. https://doi.org/10.1038/s41598-018-25281-3
Walukiewicz HE, Shukla AK, Cort JR et al (2017) Ancient regulatory role of lysine acetylation in central metabolism. Mbio 8(6):1–12. https://doi.org/10.1128/mbio.01894-17
Wang L, Du Y, Lu M, Li T (2012) ASEB: a web server for KAT-specific acetylation site prediction. Nucleic Acids Res 40:W376-379
Wang ZK, Cai Q, Liu J, Ying SH, Feng MG (2017) Global insight into lysine acetylation events and their links to biological aspects in beauveria bassiana, a fungal insect pathogen. Sci Rep 7:1–12. https://doi.org/10.1038/srep44360
Wang Y, Chen Y, Chen Q et al (2019) The role of acetylation sites in the regulation of p53 activity. Mol Biol Rep. https://doi.org/10.1007/s11033-019-05141-7
Wuyun Q, Zheng W, Zhang Y, Ruan J, Hu G (2016) Improved species-specific lysine acetylation site prediction based on a large variety of features set. PLoS ONE 11:e0155370
Xu Y, Wang XB, Ding J, Wu LY, Deng NY (2010) Lysine acetylation sites prediction using an ensemble of support vector machine classifiers. J Theor Biol 264:130–135
Xue Y, Deng W, Xu Y et al (2016) GPS-PAIL: prediction of lysine acetyltransferase-specific modification sites from protein sequences. Sci Rep 6(1):1–10. https://doi.org/10.1038/srep39787
Yang XJ, Seto E (2008) Lysine acetylation: codified crosstalk with other posttranslational modifications. Mol Cell 31(4):449–461. https://doi.org/10.1016/j.molcel.2008.07.002
Zhou S, Yang Q, Yin C, Liu L, Liang W (2016) Systematic analysis of the lysine acetylome in Fusarium graminearum. BMC Genomics. https://doi.org/10.1186/s12864-016-3361-3
Acknowledgements
The authors would like to thank the Department of Botany, Bioinformatics and Climate Change, Gujarat University for providing all the computational facilities to carry out the research. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
V.V., and J.D., conceived and designed the experiments. V.V. and K.S.L. performed the experiments and analyzed the data. V.V., and K.S.L. wrote the manuscript. J.D., and S.P. provided valuable insight and advice from time to time throughout the study. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
This article does not contain any studies with human or animal participants performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vaghasia, V., Lata, K.S., Patel, S. et al. Deciphering the lysine acetylation pattern of leptospiral strains by in silico approach. Netw Model Anal Health Inform Bioinforma 12, 15 (2023). https://doi.org/10.1007/s13721-023-00411-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13721-023-00411-8