Abstract
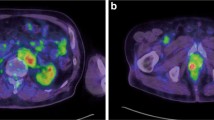
Pazopanib is an oral multi-targeted tyrosine kinase inhibitor and has been approved for metastatic renal cell carcinoma and advanced soft tissue sarcoma. To the best of our knowledge, pazopanib-induced thyrotoxicosis has never been reported. The patient was a 68-year-old woman with renal cell carcinoma and multiple metastases. Three weeks after pazopanib medication, she felt fatigue and palpitation. She was admitted because of liver chemistry abnormalities [AST 723 IU/L (normal range 7–37 IU/L); ALT 953 IU/L (normal range 3–33 IU/L)]. She showed abnormal thyroid function tests with suppressed TSH of 0.0585 μIU/mL, and elevated fT4 of 3.38 ng/dL. Ten days after admission, tachycardia appeared and fT3 and fT4 were elevated (fT3 27.85 pg/mL, fT4 2.58 ng/dL), and TSH was suppressed (0.0414 μIU/mL). Her thyroid ultrasound showed hypervascular thyroid gland without nodules. We diagnosed pazopanib-induced thyrotoxicosis. Because the fT3/fT4 ratio was high (10.8) and the thyroid was enlarged, we thought that her thyrotoxicosis might get worse. She was treated with medication of beta-blockers, hydrocortisone, and potassium iodide and thyrotoxicosis resolved. This report highlights that Pazopanib could induce thyrotoxicosis, and therefore, periodic surveillance of thyroid function is required during pazopanib therapy.

Similar content being viewed by others
References
Sternberg CN, Davis ID, Mardiak J et al (2010) Pazopanib in locally advanced or metastatic renal cell carcinoma: results of a randomized phase III trial. J Clin Oncol 28:1061–1068
Desai J, Yassa L, Marqusee E et al (2006) Hypothyroidism after sunitinib treatment for patients with gastrointestinal stromal tumors. Ann Intern Med 145:660–664
Motzer RJ, Hutson TE, Cella D et al (2013) Pazopanib versus sunitinib in metastatic renal-cell carcinoma. N Engl J Med 369:722–731
Haraldsdottir S, Li Q, Villalona-Calero MA et al (2013) Case of sorafenib-induced thyroid storm. J Clin Oncol 31:e262–e264
Sakurai K, Fukazawa H, Arihara Z et al (2010) Sunitinib-induced thyrotoxicosis followed by persistent hypothyroidism with shrinkage of thyroid volume. Tohoku J Exp Med 222:39–44
Jazvić M, Prpić M, Jukić T et al (2015) Sunitinib-induced thyrotoxicosis - a not so rare entity. Anticancer Res 35:481–485
Daimon M, Kato T, Kaino W et al (2012) Thyroid dysfunction in patients treated with tyrosine kinase inhibitors, sunitinib, sorafenib and axitinib, for metastatic renal cell carcinoma. Jpn J Clin Oncol 42:742–747
Grossmann M, Premaratne E, Desai J et al (2008) Thyrotoxicosis during sunitinib treatment for renal cell carcinoma. Clin Endocrinol (Oxf) 69:669–672
Mannavola D, Coco P, Vannucchi G et al (2007) A novel tyrosine-kinase selective inhibitor, sunitinib, induces transient hypothyroidism by blocking iodine uptake. J Clin Endocrinol Metab 92:3531–3534
Alexandrescu DT, Popoveniuc G, Farzanmehr H et al (2008) Sunitinib-associated lymphocytic thyroiditis without circulating antithyroid antibodies. Thyroid 18:809–812
Kamba T, McDonald DM (2007) Mechanisms of adverse effects of anti-VEGF therapy for cancer. Br J Cancer 96:1788–1795
Izumi Y, Hidaka Y, Tada H et al (2002) Simple and practical parameters for differentiation between destruction-induced thyrotoxicosis and Graves’ thyrotoxicosis. Clin Endocrinol (Oxf) 57:51–58
Sriphrapradang C, Bhasipol A (2016) Differentiating Graves’ disease from subacute thyroiditis using ratio of serum free triiodothyronine to free thyroxine. Ann Med Surg (Lond) 10:69–72
Rogiers A, Wolter P, Op de Beeck K (2010) Shrinkage of thyroid volume in sunitinib-treated patients with renal-cell carcinoma: a potential marker of irreversible thyroid dysfunction? Thyroid 20:317–322
Powles T, Bracarda S, Chen M et al (2015) Characterisation of liver chemistry abnormalities associated with pazopanib monotherapy: a systematic review and meta-analysis of clinical trials in advanced cancer patients. Eur J Cancer 51:1293–1302
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Research involving human participants and/or animals
For this type of study, formal consent is not required.
Informed consent
Informed consent was obtained from the patient including this report.
About this article
Cite this article
Kuribayashi, S., Takao, T., Okuda, Y. et al. Case of pazopanib-induced thyrotoxicosis in a patient with metastatic renal cell carcinoma. Int Canc Conf J 6, 118–120 (2017). https://doi.org/10.1007/s13691-017-0288-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13691-017-0288-8