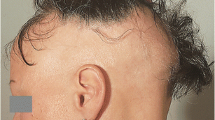
Abstract
Alopecia areata (AA) and Androgenic alopecia (AGA) are the most common multifactorial hair loss disorders that have a serious psychological impact on the affected individuals, while frontal fibrosing alopecia (FFA) is comparatively less common. However, due to the unknown etiology and the effect of many adverse factors, the prognosis of these conditions is challenging to predict. Moreover, no approved therapy has been available to date to prevent or treat these disorders. MicroRNAs (miRNAs) are a group of evolutionary conserved small non-coding RNA molecules with significant roles in the posttranscriptional gene regulation either through mRNA degradation or translational repression. A number of biological processes are controlled by these molecules, including cell growth and differentiation, proliferation, inflammation, immune responses, and apoptosis. Recently, a handful of studies have demonstrated the impact of miRNAs on common hair loss-related disorders; however, the exhaustive molecular mechanisms are still unclear. In this review, we discussed the functional implications of miRNAs in common hair loss-related disorders and addressed their efficacy to be used for theranostic purposes shortly.



Similar content being viewed by others
Abbreviations
- AA:
-
Alopecia areata
- AGA:
-
Androgenic alopecia
- AGO2:
-
Argonaute 2
- APP:
-
Amyloid-beta precursor protein
- ASK1:
-
Apoptosis signal-regulating kinase 1
- BAK1:
-
Brassinosteroid insensitive 1–associated kinase 1
- BCL2L10:
-
B-cell lymphoma/leukemia 2-like protein 10
- BNIP2:
-
B-cell lymphoma 2 interacting protein 2
- CTLA4:
-
Cytotoxic T lymphocyte antigen 4
- CXCL11:
-
C-X-C motif chemokine 11
- DTH:
-
Delayed-type hypersensitivity
- DYRK1A:
-
Dual‐specificity tyrosine phosphorylation‐regulated kinase 1A
- FFA:
-
Frontal fibrosing alopecia
- FOXO1:
-
Forkhead transcription factor 1
- FPHL:
-
Female pattern hair loss
- HF:
-
Hair follicle
- HDPC:
-
Human dermal papilla cells
- HS:
-
Hair shaft
- ICOS:
-
Inducible co-stimulatory protein
- IFNG:
-
Interferon-gamma
- IL2RA:
-
Interleukin-2 receptor alpha
- JAK/STAT:
-
Janus kinase/signal transducers and activators of transcription
- MAPK:
-
Mitogen-activated protein kinase
- miRNA:
-
MicroRNA
- MPB:
-
Male pattern baldness
- NFAT:
-
Nuclear factor of activated T cell
- p27/kip1:
-
Cyclin-dependent kinase inhibitor 1B
- p57/kip2:
-
Cyclin-dependent kinase inhibitor 1C
- pre-miRNA:
-
Precursor microRNA
- pri-miRNA:
-
Primary microRNA
- RISC:
-
RNA-induced silencing complex
- SCF/c-kit:
-
Stem cell factor/tyrosine-protein kinase KIT
- SFRP1:
-
Secreted frizzled-related protein 1
- STAT1:
-
Signal transducer and activator of transcription 1
- STX17:
-
Syntaxin 17
- TAP:
-
Transporter associated with antigen processing
- TAP2:
-
Transporter associated with antigen processing 2
- TGF:
-
Transforming growth factor
- TNKS2:
-
Tankyrase-2
- TNXB:
-
Tenascin XB
- UTR:
-
Untranslated region
- VDR:
-
Vitamin D receptor
- XPO5:
-
Exportin 5
References
Brunner MAT, Jagannathan V, Waluk DP, et al. Novel insights into the pathways regulating the canine hair cycle and their deregulation in alopecia X. PLoS ONE. 2017;12(10):e0186469. https://doi.org/10.1371/journal.pone.0186469.
Purba TS, Brunken L, Hawkshaw NJ, Peake M, Hardman J, Paus R. A primer for studying cell cycle dynamics of the human hair follicle. ExpDermatol. 2016;25(9):663–8. https://doi.org/10.1111/exd.13046.
Andl T, Botchkareva NV. MicroRNAs (miRNAs) in the control of HF development and cycling: the next frontiers in hair research. ExpDermatol. 2015;24(11):821–6. https://doi.org/10.1111/exd.12785.
Alonso L, Fuchs E. The hair cycle. J Cell Sci. 2006;119(3):391–3. https://doi.org/10.1242/jcs.02793.
Vasserot AP, Geyfman M, Poloso NJ. Androgenetic alopecia: combing the hair follicle signaling pathways for new therapeutic targets and more effective treatment options. Expert OpinTher Targets. 2019;23(9):755–71. https://doi.org/10.1080/14728222.2019.1659779.
Babalola O, Mamalis A, Lev-Tov H, Jagdeo J. The role of microRNAs in skin fibrosis. Arch Dermatol Res. 2013;305(9):763–76. https://doi.org/10.1007/s00403-013-1410-1.
Zhu L, Liu J, Cheng G. Role of microRNAs in schistosomes and schistosomiasis. Front Cell Infect Microbiol. 2014. https://doi.org/10.3389/fcimb.2014.00165.
Paul S, de la Fuente-Jiménez JL, Manriquez CG, Sharma A. Identification, characterization and expression analysis of passion fruit (Passiflora edulis) microRNAs. 3 Biotech. 2020;10(1):25. https://doi.org/10.1007/s13205-019-2000-5.
Paul S, Ruiz-Manriquez LM, Serrano-Cano FI, Estrada-Meza C, Solorio-Diaz KA, Srivastava A. Human microRNAs in host–parasite interaction: a review. 3 Biotech. 2020;10(12):510. https://doi.org/10.1007/s13205-020-02498-6.
Paul S, Bravo Vázquez LA, Pérez Uribe S, Roxana Reyes-Pérez P, Sharma A. Current status of microRNA-based therapeutic approaches in neurodegenerative disorders. Cells. 2020;9(7):1698. https://doi.org/10.3390/cells9071698.
Paul S, Reyes PR, Garza BS, Sharma A. MicroRNAs and child neuropsychiatric disorders: a brief review. Neurochem Res. 2020;45(2):232–40. https://doi.org/10.1007/s11064-019-02917-y.
Paul S, Ruiz-Manriquez LM, Ledesma-Pacheco SJ, et al. Roles of microRNAs in chronic pediatric diseases and their use as potential biomarkers: a review. Arch BiochemBiophys. 2021;699:108763. https://doi.org/10.1016/j.abb.2021.108763.
de la Fuente Jiménez JL, Sharma A, Paul S. Characterization of miRNAs from sardine (Sardina pilchardusWalbaum, 1792) and their tissue-specific expression analysis in brain and liver. 3 Biotech. 2020;10(7):318. https://doi.org/10.1007/s13205-020-02298-y.
Sharma A, Ruiz-Manriquez LM, Serrano-Cano FI, et al. Identification of microRNAs and their expression in leaf tissues of guava (Psidium guajava L) under salinity stress. Agronomy. 2020;10(12):1920. https://doi.org/10.3390/agronomy10121920.
Wolska-Gawron K, Bartosińska J, Krasowska D. MicroRNA in localized scleroderma: a review of literature. Arch Dermatol Res. 2020;312(5):317–24. https://doi.org/10.1007/s00403-019-01991-0.
Witek Ł, Janikowski T, Gabriel I, et al. Analysis of microRNA regulating cell cycle-related tumor suppressor genes in endometrial cancer patients. Hum Cell. 2020. https://doi.org/10.1007/s13577-020-00451-6.
Masalha M, Sidi Y, Avni D. The contribution of feedback loops between miRNAs, cytokines and growth factors to the pathogenesis of psoriasis. ExpDermatol. 2018;27(6):603–10. https://doi.org/10.1111/exd.13520.
Friedman RC, Farh KKH, Burge CB, Bartel DP. Most mammalian mRNAs are conserved targets of microRNAs. Genome Res. 2009;19(1):92–105. https://doi.org/10.1101/gr.082701.108.
Yang Y, Huang Q, Luo C, Wen Y, Liu R, Sun H, Tang L. MicroRNAs in acute pancreatitis: from pathogenesis to novel diagnosis and therapy. J Cell Physiol. 2020;235(3):1948–61. https://doi.org/10.1002/jcp.29212.
O’Brien J, Hayder H, Zayed Y, Peng C. Overview of MicroRNA biogenesis, mechanisms of actions, and circulation. Front Endocrinol. 2018. https://doi.org/10.3389/fendo.2018.00402.
Macfarlane LA, Murphy PR. MicroRNA: biogenesis, function and role in cancer. Curr Genomics. 2010;11(7):537–61. https://doi.org/10.2174/138920210793175895.
Shivdasani RA. MicroRNAs: regulators of gene expression and cell differentiation. Blood. 2006;108(12):3646–53. https://doi.org/10.1182/blood-2006-01-030015.
Song X, Ning W, Niu J, et al. CBX8 acts as an independent RNA-binding protein to regulate the maturation of miR-378a-3p in colon cancer cells. Hum Cell. 2021. https://doi.org/10.1007/s13577-020-00477-w.
Kotyla PJ, Islam MA. MicroRNA (miRNA): a new dimension in the pathogenesis of antiphospholipid syndrome (APS). Int J Mol Sci. 2020;21(6):2076. https://doi.org/10.3390/ijms21062076.
Aksenenko M, Palkina N, Komina A, Ruksha T. MiR-92a-1-5p and miR-328-3p are up-regulated in skin of female pattern hair loss patients. Ann Dermatol. 2019;31(2):256–9. https://doi.org/10.5021/ad.2019.31.2.256.
Goodarzi HR, Abbasi A, Saffari M, Tabei MB, NooriDaloii MR. MicroRNAs take part in pathophysiology and pathogenesis of male pattern baldness. MolBiol Rep. 2010;37(6):2959–65. https://doi.org/10.1007/s11033-009-9862-2.
Wang EHC, DeStefano GM, Patel AV, et al. Identification of differentially expressed miRNAs in alopecia areata that target immune-regulatory pathways. Genes Immun. 2017;18(2):100–4. https://doi.org/10.1038/gene.2017.4.
Bi Y, Liu G, Yang R. MicroRNAs: novel regulators during the immune response. J Cell Physiol. 2009;218(3):467–72. https://doi.org/10.1002/jcp.21639.
Engin B, Oba MÇ, Tüzün Y. Alopecia Areata. Hair Scalp Disorders. 2017. https://doi.org/10.5772/66594.
Pratt CH, King LE Jr, Messenger AG, Christiano AM, Sundberg JP. Alopecia areata. Nat Rev Dis Primers. 2017;3:17011. https://doi.org/10.1038/nrdp.2017.11.
Harries MJ, Sun J, Paus R, King LE Jr. Management of alopecia areata. BMJ. 2010;341:c3671. https://doi.org/10.1136/bmj.c3671.
Gilhar A, Schrum AG, Etzioni A, Waldmann H, Paus R. Alopecia areata: Animal models illuminate autoimmune pathogenesis and novel immunotherapeutic strategies. Autoimmun Rev. 2016;15(7):726–35. https://doi.org/10.1016/j.autrev.2016.03.008.
Pauley KM, Cha S, Chan EK. MicroRNA in autoimmunity and autoimmune diseases. J Autoimmun. 2009;32(3–4):189–94. https://doi.org/10.1016/j.jaut.2009.02.012.
Wang EHC, Yu M, Breitkopf T, et al. Identification of autoantigen epitopes in alopecia areata. J Invest Dermatol. 2016;136(8):1617–26. https://doi.org/10.1016/j.jid.2016.04.004.
Wang E, Chong K, Yu M, et al. Development of autoimmune hair loss disease alopecia areata is associated with cardiac dysfunction in C3H/HeJ mice. PLoS ONE. 2013;8(4):e62935. https://doi.org/10.1371/journal.pone.0062935.
Zhang L, Ke F, Liu Z, et al. MicroRNA-31 negatively regulates peripherally derived regulatory T-cell generation by repressing retinoic acid-inducible protein 3. Nat Commun. 2015;6(1):7639. https://doi.org/10.1038/ncomms8639.
Sheng Y, Qi S, Hu R, et al. Identification of blood microRNA alterations in patients with severe active alopecia areata. J Cell Biochem. 2019;120(9):14421–30. https://doi.org/10.1002/jcb.28700.
Sánchez-Jiménez C, Carrascoso I, Barrero J, Izquierdo JM. Identification of a set of miRNAs differentially expressed in transiently TIA-depleted HeLa cells by genome-wide profiling. BMC MolBiol. 2013;14(1):4. https://doi.org/10.1186/1471-2199-14-4.
Tafazzoli A, Forstner AJ, Broadley D, et al. Genome-wide MicroRNA analysis implicates miR-30b/d in the etiology of alopecia areata. J Invest Dermatol. 2018;138(3):549–56. https://doi.org/10.1016/j.jid.2017.09.046.
Gratz IK, Truong HA, Yang SH, et al. Cutting Edge: memory regulatory t cells require IL-7 and not IL-2 for their maintenance in peripheral tissues. J Immunol. 2013;190(9):4483–7. https://doi.org/10.4049/jimmunol.1300212.
Erjavec S, Abdelaziz A, Patel A, Petukhova L, Christiano A. 827 Role of the autophagy protein, Syntaxin 17 (STX17), in melanogenesis and alopecia areata. J Invest Dermatol. 2018;138(5):S140. https://doi.org/10.1016/j.jid.2018.03.837.
Petukhova L, Duvic M, Hordinsky M, et al. Genome-wide association study in alopecia areata implicates both innate and adaptive immunity. Nature. 2010;466(7302):113–7. https://doi.org/10.1038/nature09114.
Lee YH, Bae S-C, Choi SJ, Ji JD, Song GG. Genome-wide pathway analysis of genome-wide association studies on systemic lupus erythematosus and rheumatoid arthritis. MolBiol Rep. 2012;39(12):10627–35. https://doi.org/10.1007/s11033-012-1952-x.
Gulati N, Løvendorf MB, Zibert JR, et al. Unique microRNAs appear at different times during the course of a delayed-type hypersensitivity reaction in human skin. ExpDermatol. 2015;24(12):953–7. https://doi.org/10.1111/exd.12813.
Smith NL, Wissink EM, Grimson A, Rudd BD. miR-150 regulates differentiation and cytolytic effector function in CD8+ T cells. Sci Rep. 2015;5(1):16399. https://doi.org/10.1038/srep16399.
Huang B, Zhao J, Lei Z, et al. miR-142-3p restricts cAMP production in CD4+CD25- T cells and CD4+CD25+ TREG cells by targeting AC9 mRNA. EMBO Rep. 2009;10(2):180–5. https://doi.org/10.1038/embor.2008.224.
Ding S, Liang Y, Zhao M, et al. Decreased microRNA-142-3p/5p expression causes CD4+ T cell activation and B cell hyperstimulation in systemic lupus erythematosus. Arthritis Rheum. 2012;64(9):2953–63. https://doi.org/10.1002/art.34505.
Heilmann-Heimbach S, Herold C, Hochfeld LM, et al. Meta-analysis identifies novel risk loci and yields systematic insights into the biology of male-pattern baldness. Nat Commun. 2017;8:14694. https://doi.org/10.1038/ncomms14694.
Mysore V, Parthasaradhi A, Kharkar RD, et al. Expert consensus on the management of androgenetic alopecia in India. Int J Trichology. 2019;11(3):101–6. https://doi.org/10.4103/ijt.ijt_24_19.
Ning MS, Andl T. Control by a hair’s breadth: the role of microRNAs in the skin. Cell Mol Life Sci. 2013;70(7):1149–69. https://doi.org/10.1007/s00018-012-1117-z.
Deng W, Hu T, Han L, et al. miRNA microarray profiling in patients with androgenic alopecia and the effects of miR-133b on hair growth. ExpMolPathol. 2021;118:104589. https://doi.org/10.1016/j.yexmp.2020.104589.
Mohammadi P, Nilforoushzadeh MA, Youssef KK, et al. Defining microRNA signatures of hair follicular stem and progenitor cells in healthy and androgenic alopecia patients. J DermatolSci. 2021;101(1):49–57. https://doi.org/10.1016/j.jdermsci.2020.11.002.
Fabbrocini G, Cantelli M, Masarà A, Annunziata MC, Marasca C, Cacciapuoti S. Female pattern hair loss: a clinical, pathophysiologic, and therapeutic review. Int J WomensDermatol. 2018;4(4):203–11. https://doi.org/10.1016/j.ijwd.2018.05.001.
Tziotzios C, Ainali C, Holmes S, et al. Tissue and Circulating MicroRNA Co-expression Analysis Shows Potential Involvement of miRNAs in the Pathobiology of Frontal Fibrosing Alopecia. J Invest Dermatol. 2017;137(11):2440–3. https://doi.org/10.1016/j.jid.2017.06.030.
Tziotzios C, Stefanato CM, Fenton DA, Simpson MA, McGrath JA. Frontal fibrosing alopecia: reflections and hypotheses on aetiology and pathogenesis. ExpDermatol. 2016;25(11):847–52. https://doi.org/10.1111/exd.13071.
Ideozu JE, Zhang X, Rangaraj V, McColley S, Levy H. Microarray profiling identifies extracellular circulating miRNAs dysregulated in cystic fibrosis. Sci Rep. 2019;9(1):15483. https://doi.org/10.1038/s41598-019-51890-7.
Ichihara A, Wang Z, Jinnin M, et al. Upregulation of miR-18a-5p contributes to epidermal necrolysis in severe drug eruptions. J Allergy ClinImmunol. 2014;133(4):1065–74. https://doi.org/10.1016/j.jaci.2013.09.019.
Kim O-Y, Cha HJ, Ahn KJ, An I-S, An S, Bae S. Identification of microRNAs involved in growth arrest and cell death in hydrogen peroxide-treated human dermal papilla cells. Mol Med Rep. 2014;10(1):145–54. https://doi.org/10.3892/mmr.2014.2158.
Lu Y, Li Z, Xie B, Song Y, Ye X, Liu P. hsa-miR-20a-5p attenuates allergic inflammation in HMC-1 cells by targeting HDAC4. MolImmunol. 2019;107:84–90. https://doi.org/10.1016/j.molimm.2019.01.010.
Jinnin M. Recent progress in studies of miRNA and skin diseases. J Dermatol. 2015;42(6):551–8. https://doi.org/10.1111/1346-8138.12904.
Gohary YM, Abdel Fattah DS. Detection of tumor necrosis factor-alpha in nonlesional tissues of alopecia areata patients: a prove for a systemic disease. Int J Trichology. 2017;9(4):154–9. https://doi.org/10.4103/ijt.ijt_47_17.
Hassan AM, Neinaa YME-H, El-Bendary AS, Zakaria SS. MicroRNA-146a and Forkhead box protein 3 expressions in nonsegmental vitiligo: an insight into disease pathogenesis. J WomensDermatolSoc. 2019;16(2):105. https://doi.org/10.4103/JEWD.JEWD_19_19.
Lu MC, Yu CL, Chen HC, Yu HC, Huang HB, Lai NS. Increased miR-223 expression in T cells from patients with rheumatoid arthritis leads to decreased insulin-like growth factor-1-mediated interleukin-10 production. ClinExpImmunol. 2014;177(3):641–51. https://doi.org/10.1111/cei.12374.
Khor B, Gagnon JD, Goel G, et al. The kinase DYRK1A reciprocally regulates the differentiation of Th17 and regulatory T cells. Elife. 2015;4:e05920. https://doi.org/10.7554/eLife.05920.
Rehage N, Davydova E, Conrad C, et al. Binding of NUFIP2 to Roquin promotes recognition and regulation of ICOS mRNA. Nat Commun. 2018;9(1):299. https://doi.org/10.1038/s41467-017-02582-1.
Sánchez-Díaz R, Blanco-Dominguez R, Lasarte S, et al. Thymus-derived regulatory T cell development is regulated by C-Type lectin-mediated BIC/MicroRNA 155 expression. Mol Cell Biol. 2017;37(9):e00341-e416. https://doi.org/10.1128/MCB.00341-16.
Yuan S, Li F, Meng Q, et al. Posttranscriptional regulation of keratinocyte progenitor cell expansion, differentiation and hair follicle regression by miR-22. PLoS Genet. 2015;11(5):e1005253. https://doi.org/10.1371/journal.pgen.1005253.
Malik A, Pal R, Gupta SK. EGF-mediated reduced miR-92a-1-5p controls HTR-8/SVneo cell invasion through activation of MAPK8 and FAS which in turn increase MMP-2/-9 expression. Sci Rep. 2020;10(1):12274. https://doi.org/10.1038/s41598-020-68966-4.
Koga T, Ichinose K, Mizui M, Crispín JC, Tsokos GC. Calcium/calmodulin-dependent protein kinase IV suppresses IL-2 production and regulatory T cell activity in lupus. J Immunol. 2012;189(7):3490–6. https://doi.org/10.4049/jimmunol.1201785.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that there are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Paul, S., Licona-Vázquez, I., Serrano-Cano, F.I. et al. Current insight into the functions of microRNAs in common human hair loss disorders: a mini review. Human Cell 34, 1040–1050 (2021). https://doi.org/10.1007/s13577-021-00540-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13577-021-00540-0