Abstract
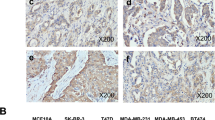
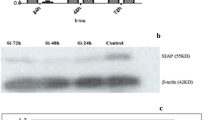
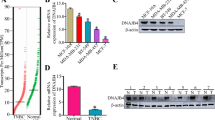
Breast cancer (BC) threatened the life health of a tremendous amount of the population, and the estimated number of death is still rising nowadays. We found that stress-induced phosphoprotein 1 (STIP1) is overexpressed in BC tissues compared to non-tumorous breast tissues. Our study is to validate the prognostic value of STIP1 and investigate its biological role in BC. We verified the upregulation of STIP1 in multiple databases, proved that STIP1 is upregulated in BC tissues and cell lines using real-time quantitative PCR (qRT-PCR). We used small interfering RNA to examine the function of STIP1 in BC cell lines (BT-549, MDA-MB-231, Hs-578 T) and explored the mechanism of function of STIP1 in BC cells using Western blotting and qRT-PCR. Analyses of multiple databases indicated that high STIP1 expression is a marker that effectively distinguishes BC patients from healthy control and predicts worse clinical outcomes in BC. The loss-of-function experiments showed that STIP1 silencing results in inhibition of cell proliferation and migration, inducing cell apoptosis, and S-phase arrest in vitro. Our study also showed that STIP1 downregulation inhibited the JAK2/STAT3 pathway and epithelial-mesenchymal transition process. Rescue experiments demonstrated that the oncogenic effect of STIP1 is partially dependent on mediating JAK2 expression. This study verified that STIP1 is an oncogenic gene that promotes BC progression and serves as a valuable diagnostic and outcome-related marker of BC.







Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70(1):7–30. https://doi.org/10.3322/caac.21590.
Howlader N, Altekruse SF, Li CI, Chen VW, Clarke CA, Ries LA, et al. US incidence of breast cancer subtypes defined by joint hormone receptor and HER2 status. J Natl Cancer Inst. 2014;106:5. https://doi.org/10.1093/jnci/dju055.
The Cancer Genome Atlas Network. Comprehensive molecular portraits of human breast tumours. Nature 490(7418):61–70 https://doi.org/10.1038/nature11412.
Moo TA, Sanford R, Dang C, Morrow M. Overview of breast cancer therapy. PET Clin. 2018;13(3):339–54. https://doi.org/10.1016/j.cpet.2018.02.006.
Krug D, Baumann R, Budach W, Dunst J, Feyer P, Fietkau R, et al. Individualization of post-mastectomy radiotherapy and regional nodal irradiation based on treatment response after neoadjuvant chemotherapy for breast cancer : a systematic review. Strahlenther Onkol Organ Deutschen Rontgengesellschaft. 2018;194(7):607–18. https://doi.org/10.1007/s00066-018-1270-x.
Freedman RA. Treatment of breast cancer in the elderly. Curr Oncol Rep. 2015;17(11):51. https://doi.org/10.1007/s11912-015-0475-8.
Harbeck N, Gnant M. Breast cancer. Lancet (Lond Engl). 2017;389(10074):1134–50. https://doi.org/10.1016/S0140-6736(16)31891-8.
Noone AM HN, Krapcho M, Miller D, Brest A, Yu M, Ruhl J, Tatalovich Z, Mariotto A, Lewis DR, Chen HS, Feuer EJ, Cronin KA (eds) SEER cancer statistics review, 1975–2015, National Cancer Institute. Bethesda, MD, https://seer.cancer.gov/csr/1975_2015/, based on November 2017 SEER data submission, posted to the SEER web site, April 2018. Accessed 26 July 2020.
Bhandari A, Zheng C, Sindan N, Quan R, Xia E, Thapa Y, et al. COPB2 is up-regulated in breast cancer and plays a vital role in the metastasis via N-cadherin and Vimentin. J Cell Mol Med. 2019;23(8):5235–45. https://doi.org/10.1111/jcmm.14398.
Guan Y, Bhandari A, Xia E, Yang F, Xiang J, Wang O. lncRNA FOXD3-AS1 is associated with clinical progression and regulates cell migration and invasion in breast cancer. Cell Biochem Funct. 2019;37(4):239–44. https://doi.org/10.1002/cbf.3393.
Song Y, Masison DC. Independent regulation of Hsp70 and Hsp90 chaperones by Hsp70/Hsp90-organizing protein Sti1 (Hop1). J Biol Chem. 2005;280(40):34178–85. https://doi.org/10.1074/jbc.M505420200.
Odunuga OO, Longshaw VM, Blatch GL. Hop: more than an Hsp70/Hsp90 adaptor protein. BioEssays. 2004;26(10):1058–68. https://doi.org/10.1002/bies.20107.
Muller P, Ruckova E, Halada P, Coates PJ, Hrstka R, Lane DP, et al. C-terminal phosphorylation of Hsp70 and Hsp90 regulates alternate binding to co-chaperones CHIP and HOP to determine cellular protein folding/degradation balances. Oncogene. 2013;32(25):3101–10. https://doi.org/10.1038/onc.2012.314.
Jing Y, Liang W, Liu J, Zhang L, Wei J, Zhu Y, et al. Stress-induced phosphoprotein 1 promotes pancreatic cancer progression through activation of the FAK/AKT/MMP signaling axis. Pathol Res Pract. 2019;215(11):152564. https://doi.org/10.1016/j.prp.2019.152564.
Sun X, Cao N, Mu L, Cao W. Stress induced phosphoprotein 1 promotes tumor growth and metastasis of melanoma via modulating JAK2/STAT3 pathway. Biomed Pharmacother. 2019;116:108962. https://doi.org/10.1016/j.biopha.2019.108962.
Wang TH, Chao A, Tsai CL, Chang CL, Chen SH, Lee YS, et al. Stress-induced phosphoprotein 1 as a secreted biomarker for human ovarian cancer promotes cancer cell proliferation. Mol Cell Proteomics. 2010;9(9):1873–84. https://doi.org/10.1074/mcp.M110.000802.
Tsai CL, Tsai CN, Lin CY, Chen HW, Lee YS, Chao A, et al. Secreted stress-induced phosphoprotein 1 activates the ALK2-SMAD signaling pathways and promotes cell proliferation of ovarian cancer cells. Cell Rep. 2012;2(2):283–93. https://doi.org/10.1016/j.celrep.2012.07.002.
Huang L, Zhai E, Cai S, Lin Y, Liao J, Jin H, et al. Stress-inducible Protein-1 promotes metastasis of gastric cancer via Wnt/beta-catenin signaling pathway. J Exp Clin Cancer Res. 2018;37(1):6. https://doi.org/10.1186/s13046-018-0676-8.
Wu R, Liu F, Peng P, Qiu H, Xiong H, Yu S, et al. Tumor stress-induced phosphoprotein 1 as a prognostic biomarker for breast cancer. Ann Transl Med. 2018;6(15):302. https://doi.org/10.21037/atm.2018.06.46.
Li T, Fan J, Wang B, Traugh N, Chen Q, Liu JS, et al. TIMER: a web server for comprehensive analysis of tumor-infiltrating immune cells. Cancer Res. 2017;77(21):e108–10. https://doi.org/10.1158/0008-5472.CAN-17-0307.
Ivshina AV, George J, Senko O, Mow B, Putti TC, Smeds J, et al. Genetic reclassification of histologic grade delineates new clinical subtypes of breast cancer. Cancer Res. 2006;66(21):10292–301. https://doi.org/10.1158/0008-5472.CAN-05-4414.
Pawitan Y, Bjohle J, Amler L, Borg AL, Egyhazi S, Hall P, et al. Gene expression profiling spares early breast cancer patients from adjuvant therapy: derived and validated in two population-based cohorts. Breast Cancer Res. 2005;7(6):R953–64. https://doi.org/10.1186/bcr1325.
Lance D, Miller JS, George J, Vega VB, Vergara L, Ploner A, Pawitan Y, Hall P, Klaar S, Liu ET, Bergh J. An expression signature for p53 status in human breast cancer predicts mutation status, transcriptional effects, and patient survival. PNAS. 2005;102(38):13550–5.
Nagy A, Lanczky A, Menyhart O, Gyorffy B. Validation of miRNA prognostic power in hepatocellular carcinoma using expression data of independent datasets. Sci Rep. 2018;8(1):9227. https://doi.org/10.1038/s41598-018-27521-y.
Gyorffy B, Lanczky A, Eklund AC, Denkert C, Budczies J, Li Q, et al. An online survival analysis tool to rapidly assess the effect of 22,277 genes on breast cancer prognosis using microarray data of 1809 patients. Breast Cancer Res Treat. 2010;123(3):725–31. https://doi.org/10.1007/s10549-009-0674-9.
Curtis C, Shah SP, Chin SF, Turashvili G, Rueda OM, Dunning MJ, et al. The genomic and transcriptomic architecture of 2,000 breast tumours reveals novel subgroups. Nature. 2012;486(7403):346–52. https://doi.org/10.1038/nature10983.
Stefan GluCk JSR, Royce M, Edward F, Jr. McKenna, Perou CM, Avisar E, Lin W. TP53 genomics predict higher clinical and pathologic tumor response in operable early-stage breast cancer treated with docetaxel-capecitabine—trastuzumab. Breast Cancer Res Treat. 2012;132:781–91. https://doi.org/10.1007/s10549-011-1412-7.
Chandrashekar D, Bashel B, Balasubramanya S, Creighton C, Ponce-Rodriguez I, Chakravarthi B, et al. UALCAN: a portal for facilitating tumor subgroup gene expression and survival analyses. Neoplasia (New York, NY). 2017;19(8):649–58. https://doi.org/10.1016/j.neo.2017.05.002.
Guo X, Yan Z, Zhang G, Wang X, Pan Y, Huang M. STIP1 regulates proliferation and migration of lung adenocarcinoma through JAK2/STAT3 signaling pathway. Cancer Manag Res. 2019;11:10061–72. https://doi.org/10.2147/CMAR.S233758.
Jin W. Role of JAK/STAT3 signaling in the regulation of metastasis, the transition of cancer stem cells, and chemoresistance of cancer by epithelial-mesenchymal transition. Cells. 2020;9:1. https://doi.org/10.3390/cells9010217.
Tsai CL, Chao A, Jung SM, Tsai CN, Lin CY, Chen SH, et al. Stress-induced phosphoprotein-1 maintains the stability of JAK2 in cancer cells. Oncotarget. 2016;7(31):50548–63. https://doi.org/10.18632/oncotarget.10500.
Lehmann BD, Pietenpol JA. Clinical implications of molecular heterogeneity in triple negative breast cancer. Breast. 2015;24(Suppl 2):S36-40. https://doi.org/10.1016/j.breast.2015.07.009.
Baindur-Hudson S, Edkins AL, Blatch GL. Hsp70/Hsp90 organising protein (hop): beyond interactions with chaperones and prion proteins. Subcell Biochem. 2015;78:69–90. https://doi.org/10.1007/978-3-319-11731-7_3.
Beraldo FH, Soares IN, Goncalves DF, Fan J, Thomas AA, Santos TG, et al. Stress-inducible phosphoprotein 1 has unique cochaperone activity during development and regulates cellular response to ischemia via the prion protein. FASEB J. 2013;27(9):3594–607. https://doi.org/10.1096/fj.13-232280.
Yin H, Deng Z, Li X, Li Y, Yin W, Zhao G, et al. Down-regulation of STIP1 regulate apoptosis and invasion of glioma cells via TRAP1/AKT signaling pathway. Cancer Genet. 2019;237:1–9. https://doi.org/10.1016/j.cancergen.2019.05.006.
Luo X, Liu Y, Ma S, Liu L, Xie R, Li M, et al. STIP1 is over-expressed in hepatocellular carcinoma and promotes the growth and migration of cancer cells. Gene. 2018;662:110–7. https://doi.org/10.1016/j.gene.2018.03.076.
Krafft U, Tschirdewahn S, Hess J, Harke NN, Hadaschik BA, Nyirady P, et al. STIP1 tissue expression is associated with survival in chemotherapy-treated bladder cancer patients. Pathol Oncol Res. 2020;26(2):1243–9. https://doi.org/10.1007/s12253-019-00689-y.
Jiang YZ, Ma D, Suo C, Shi J, Xue M, Hu X, et al. Genomic and transcriptomic landscape of triple-negative breast cancers: subtypes and treatment strategies. Cancer Cell. 2019;35(3):428–40. https://doi.org/10.1016/j.ccell.2019.02.001.
Miklossy G, Hilliard TS, Turkson J. Therapeutic modulators of STAT signalling for human diseases. Nat Rev Drug Discov. 2013;12(8):611–29. https://doi.org/10.1038/nrd4088.
Chang R, Song L, Xu Y, Wu Y, Dai C, Wang X, et al. Loss of Wwox drives metastasis in triple-negative breast cancer by JAK2/STAT3 axis. Nat Commun. 2018;9(1):3486. https://doi.org/10.1038/s41467-018-05852-8.
Lin WH, Dai WG, Xu XD, Yu QH, Zhang B, Li J, et al. Downregulation of DPF3 promotes the proliferation and motility of breast cancer cells through activating JAK2/STAT3 signaling. Biochem Biophys Res Commun. 2019;514(3):639–44. https://doi.org/10.1016/j.bbrc.2019.04.170.
Groner B, von Manstein V. Jak Stat signaling and cancer: Opportunities, benefits and side effects of targeted inhibition. Mol Cell Endocrinol. 2017;451:1–14. https://doi.org/10.1016/j.mce.2017.05.033.
Cho KH, Jeong KJ, Shin SC, Kang J, Park CG, Lee HY. STAT3 mediates TGF-beta1-induced TWIST1 expression and prostate cancer invasion. Cancer Lett. 2013;336(1):167–73. https://doi.org/10.1016/j.canlet.2013.04.024.
Steelman LS, Pohnert SC, Shelton JG, Franklin RA, Bertrand FE, McCubrey JA. JAK/STAT, Raf/MEK/ERK, PI3K/Akt and BCR-ABL in cell cycle progression and leukemogenesis. Leukemia. 2004;18(2):189–218. https://doi.org/10.1038/sj.leu.2403241.
Wu Y, Sarkissyan M, Vadgama JV. Epithelial-mesenchymal transition and breast cancer. J Clin Med. 2016;5:2. https://doi.org/10.3390/jcm5020013.
Chen Z, Xu L, Su T, Ke Z, Peng Z, Zhang N, et al. Autocrine STIP1 signaling promotes tumor growth and is associated with disease outcome in hepatocellular carcinoma. Biochem Biophys Res Commun. 2017;493(1):365–72. https://doi.org/10.1016/j.bbrc.2017.09.016.
Tsai C, Tsai C, Lin C, Chen H, Lee Y, Chao A, et al. Secreted stress-induced phosphoprotein 1 activates the ALK2-SMAD signaling pathways and promotes cell proliferation of ovarian cancer cells. Cell reports. 2012;2(2):283–93. https://doi.org/10.1016/j.celrep.2012.07.002.
Wang J, Gong C, Guo F, Zhou X, Zhang M, Qiu H, et al. Knockdown of STIP1 inhibits the invasion of CD133-positive cancer stem-like cells of the osteosarcoma MG63 cell line via the PI3K/Akt and ERK1/2 pathways. Int J Mol Med. 2020;46(6):2251–9. https://doi.org/10.3892/ijmm.2020.4764.
Wang T, Chao A, Tsai C, Chang C, Chen S, Lee Y, et al. Stress-induced phosphoprotein 1 as a secreted biomarker for human ovarian cancer promotes cancer cell proliferation. Mol Cell Proteom MCP. 2010;9(9):1873–84. https://doi.org/10.1074/mcp.M110.000802.
Funding
This study was funded by the National Natural Science Foundation of China (No. 81572291), the Natural Science Foundation of Zhejiang Province (LGF18H160031, LY18H160053), and the Science And Technology Project of Wenzhou (Y20190204, Y20170740).
Author information
Authors and Affiliations
Contributions
LL and JW wrote the manuscript. JW and BL did the main experiments. EX and CZ analyzed the raw data. YW and LY revised the manuscript. YC and OW designed and funded the whole work.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
The ethics approval of this study was acquired from the Institutional Review Board of First Affiliated Hospital of Wenzhou Medical University (Approval no. 2012‐57).
Informed consent
All patients provided written informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lin, L., Wen, J., Lin, B. et al. Stress-induced phosphoprotein 1 facilitates breast cancer cell progression and indicates poor prognosis for breast cancer patients. Human Cell 34, 901–917 (2021). https://doi.org/10.1007/s13577-021-00507-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13577-021-00507-1