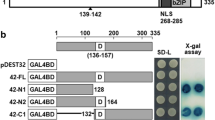
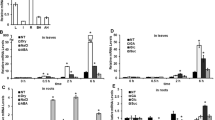
Abstract
Plants exposed to adverse environmental conditions are invariably compromised in their growth and development. The bZIP class of transcription factors (TF) form a large group among stress signalling components that regulate plant responses towards stress. We identified bZIP TF encoding genes that are expressed differentially in indica rice under stress and here we functionally characterize one such gene, OsbZIP16. Although, OsbZIP16 forms a clade with its orthologous monocot protein sequences, we find in our study that it can impart tolerance to abiotic stress in Arabidopsis. OsbZIP16 is expressed strongly upon dehydration, salt and ABA treatment in Oryza sativa cv. IR64 seedlings. It localizes in the cell nucleus and the gene product is capable of transcriptional activation, thus providing evidence for its capability as a functional TF. Upon overexpression in Arabidopsis, OsbZIP16ox plants show wild type morphology, however, these plants showed tolerance when subjected to drought stress at vegetative stage and set healthy seeds on recovery. The OsbZIP16ox seedlings showed reduced sensitivity to mannitol, ABA and sodium chloride during germination and also reduced ROS accumulation upon H2O2 exposure. Thus, OsbZIP16 regulates abiotic stress responses and is also a good candidate gene that can be utilised to impart tolerance in plants under water deficit conditions.





Similar content being viewed by others
Abbreviations
- OsbZIP:
-
Oryza sativa basic leucine zipper
- ABA:
-
Abscisic acid
- ROS:
-
Reactive oxygen species
- TFs:
-
Transcription factors
References
Amir Hossain M, Lee Y, Cho J-I et al (2010) The bZIP transcription factor OsABF1 is an ABA responsive element binding factor that enhances abiotic stress signaling in rice. Plant Mol Biol 72:557–566. https://doi.org/10.1007/s11103-009-9592-9
Bensmihen S, Giraudat J, Parcy F (2005) Characterization of three homologous basic leucine zipper transcription factors (bZIP) of the ABI5 family during Arabidopsis thaliana embryo maturation. J Exp Bot 56:597–603. https://doi.org/10.1093/jxb/eri050
Borah P, Sharma E, Kaur A et al (2017) Analysis of drought-responsive signalling network in two contrasting rice cultivars using transcriptome-based approach. Sci Rep 7:42131. https://doi.org/10.1038/srep42131
Burman N, Bhatnagar A, Khurana JP (2017) OsbZIP48, a HY5 transcription factor ortholog, exerts pleiotropic effects in light-regulated development. Plant Physiol. https://doi.org/10.1104/pp.17.00478
Chen H, Chen W, Zhou J et al (2012) Basic leucine zipper transcription factor OsbZIP16 positively regulates drought resistance in rice. Plant Sci 193–194:8–17. https://doi.org/10.1016/j.plantsci.2012.05.003
Chern MS, Eiben HG, Bustos MM (1996) The developmentally regulated bZIP factor ROM1 modulates transcription from lectin and storage protein genes in bean embryos. Plant J 10:135–148
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
Finkelstein R (2013) Abscisic acid synthesis and response. Arabidopsis Book 11:e0166. https://doi.org/10.1199/tab.0166
Guo M, Chen Y, Du Y et al (2011) The bZIP transcription factor MoAP1 mediates the oxidative stress response and is critical for pathogenicity of the rice blast fungus magnaporthe oryzae. PLoS Pathog. https://doi.org/10.1371/journal.ppat.1001302
Huang XS, Liu JH, Chen XJ (2010) Overexpression of PtrABF gene, a bZIP transcription factor isolated from Poncirus trifoliata, enhances dehydration and drought tolerance in tobacco via scavenging ROS and modulating expression of stress-responsive genes. BMC Plant Biol. https://doi.org/10.1186/1471-2229-10-230
Hurst HC (1995) Transcription factors 1: bZIP proteins. Protein Profile 2:101–168
Jabs T, Dietrich RA, Dangl JL (1996) Initiation of runaway cell death in an Arabidopsis mutant by extracellular superoxide. Science 273:1853–1856. https://doi.org/10.1126/science.273.5283.1853
Jain M, Nijhawan A, Tyagi AK, Khurana JP (2006a) Validation of housekeeping genes as internal control for studying gene expression in rice by quantitative real-time PCR. Biochem Biophys Res Commun 345:646–651. https://doi.org/10.1016/j.bbrc.2006.04.140
Jain M, Tyagi AK, Khurana JP (2006b) Overexpression of putative topoisomerase 6 genes from rice confers stress tolerance in transgenic Arabidopsis plants. FEBS J 273:5245–5260. https://doi.org/10.1111/j.1742-4658.2006.05518.x
Jain M, Tyagi AK, Khurana JP (2008) Constitutive expression of a meiotic recombination protein gene homolog, OsTOP6A1, from rice confers abiotic stress tolerance in transgenic Arabidopsis plants. Plant Cell Rep 27:767–778. https://doi.org/10.1007/s00299-007-0491-8
Jakoby M, Weisshaar B, Dröge-Laser W et al (2002) bZIP transcription factors in Arabidopsis. Trends Plant Sci 7:106–11
Jin J, Tian F, Yang DC et al (2017) PlantTFDB 4.0: toward a central hub for transcription factors and regulatory interactions in plants. Nucleic Acids Res 45:D1040–D1045. https://doi.org/10.1093/nar/gkw982
Liu C, Wu Y, Wang X (2012) BZIP transcription factor OsbZIP52/RISBZ5: a potential negative regulator of cold and drought stress response in rice. Planta 235:1157–1169. https://doi.org/10.1007/s00425-011-1564-z
Liu C, Mao B, Ou S et al (2014) OsbZIP71, a bZIP transcription factor, confers salinity and drought tolerance in rice. Plant Mol Biol 84:19–36. https://doi.org/10.1007/s11103-013-0115-3
Lu G, Gao C, Zheng X, Han B (2009) Identification of OsbZIP72 as a positive regulator of ABA response and drought tolerance in rice. Planta 229:605–615. https://doi.org/10.1007/s00425-008-0857-3
Meng X-B, Zhao W-S, Lin R-M et al (2005) Identification of a novel rice bZIP-type transcription factor gene, OsbZIP1, involved in response to infection ofMagnaporthe grisea. Plant Mol Biol Report 23:301–302. https://doi.org/10.1007/BF02772762
Mittler R, Vanderauwera S, Suzuki N et al (2011) ROS signaling: the new wave? Trends Plant Sci 16:300–309. https://doi.org/10.1016/j.tplants.2011.03.007
Mukherjee K, Choudhury A, Gupta B et al (2006) An ABRE-binding factor, OSBZ8, is highly expressed in salt tolerant cultivars than in salt sensitive cultivars of indica rice. BMC Plant Biol 6:18. https://doi.org/10.1186/1471-2229-6-18
Nakagawa H, Ohmiya K, Hattori T (1996) A rice bZIP protein, designated OSBZ8, is rapidly induced by abscisic acid. Plant J 9:217–227. https://doi.org/10.1046/J.1365-313x.1996.09020217.X
Nijhawan A, Jain M, Tyagi AK, Khurana JP (2008) Genomic survey and gene expression analysis of the basic leucine zipper transcription factor family in rice. Plant Physiol 146:333–350. https://doi.org/10.1104/pp.107.112821
Noctor G, Mhamdi A, Foyer CH (2014) The roles of reactive oxygen metabolism in drought: not so cut and dried. Plant Physiol 164:1636–1648. https://doi.org/10.1104/pp.113.233478
Osakabe Y, Osakabe K, Shinozaki K, Tran L-SP (2014) Response of plants to water stress. Front Plant Sci 5:86. https://doi.org/10.3389/fpls.2014.00086
Pandey AS (2015) Functional characterization of some rice bZIP transcription factor genes involved in stress tolerance and seed development (PhD thesis). University of Delhi
Riechmann JL, Heard J, Martin G et al (2000) Arabidopsis transcription factors: genome-wide comparative analysis among eukaryotes. Science 290:2105–2110. https://doi.org/10.1126/science.290.5499.2105
Schraudner M, Moeder W, Wiese C et al (1998) Ozone-induced oxidative burst in the ozone biomonitor plant, tobacco Bel W3. Plant J 16:235–245. https://doi.org/10.1046/j.1365-313X.1998.00294.x
Sharma E, Sharma R, Borah P et al (2015) Emerging roles of auxin in abiotic stress responses. In: Pandey GK (ed) Elucidation abiotic stress signal plants funct genomics perspect. Springer, New York. https://doi.org/10.1007/978-1-4939-2211-6_11
Tang N, Zhang H, Li X et al (2012) Constitutive activation of transcription factor OsbZIP46 improves drought tolerance in rice. Plant Physiol 158:1755–1768. https://doi.org/10.1104/pp.111.190389
Todaka D, Shinozaki K, Yamaguchi-Shinozaki K (2015) Recent advances in the dissection of drought-stress regulatory networks and strategies for development of drought-tolerant transgenic rice plants. Front Plant Sci 6:84. https://doi.org/10.3389/fpls.2015.00084
Wohlgemuth H, Mittelstrass K, Kschieschan S et al (2002) Activation of an oxidative burst is a general feature of sensitive plants exposed to the air pollutant ozone. Plant Cell Environ 25:717–726. https://doi.org/10.1046/j.1365-3040.2002.00859.x
Xiang Y, Tang N, Du H et al (2008) Characterization of OsbZIP23 as a key player of the basic leucine zipper transcription factor family for conferring abscisic acid sensitivity and salinity and drought tolerance in rice. Plant Physiol 148:1938–1952. https://doi.org/10.1104/pp.108.128199
Zou M, Guan Y, Ren H et al (2007) Characterization of alternative splicing products of bZIP transcription factors OsABI5. Biochem Biophys Res Commun 360:307–313. https://doi.org/10.1016/j.bbrc.2007.05.226
Zou M, Guan Y, Ren H et al (2008) A bZIP transcription factor, OsABI5, is involved in rice fertility and stress tolerance. Plant Mol Biol 66:675–683. https://doi.org/10.1007/s11103-008-9298-4
Acknowledgements
This work was financially supported by a grant (BT/PR12394/AGIII/103/891/2014) from the Department of Biotechnology, Government of India. ASP, ES and NB are grateful to CSIR and NJ to UGC for the award of research fellowships. The infrastructure support provided by the Department of Science and Technology, Government of India, and the University Grants Commission, New Delhi, is also acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no competing financial conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pandey, A.S., Sharma, E., Jain, N. et al. A rice bZIP transcription factor, OsbZIP16, regulates abiotic stress tolerance when over-expressed in Arabidopsis. J. Plant Biochem. Biotechnol. 27, 393–400 (2018). https://doi.org/10.1007/s13562-018-0448-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13562-018-0448-8