Abstract
The skin microbiome consists of the microorganisms populating the human skin. Cutibacterium acnes (C. acnes, formerly named Propionibacterium acnes) is recognized as a key factor in acne development, regulating inflammatory and immune pathways. Dysbiosis has been described as the imbalance in skin microbiome homeostasis and may play a role in acne pathogenesis. Microbial interference has been shown to be a contributor to healthy skin homeostasis and staphylococcal strains may exclude acne-associated C. acnes phylotypes. In this review we present an update on the skin microbiome in acne and discuss how current acne treatments such as benzoyl peroxide, orally administered isotretinoin, and antibiotics may affect the skin microbiome homeostasis. We highlight the collateral damage of acne antibiotics on the skin microbiome, including the risk of antimicrobial resistance and the dysregulation of the microbiome equilibrium that may occur even with short-term antibiotic courses. Consequently, the interest is shifting towards new non-antibiotic pharmacological acne treatments. Orally administered spironolactone is an emerging off-label treatment for adult female patients and topical peroxisome proliferator-activated receptor gamma (PPARγ) modulation is being studied for patients with acne. The potential application of topical or oral probiotics, bacteriotherapy, and phage therapy for acne are further promising areas of future research.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
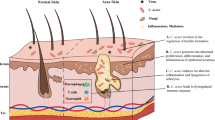
C. acnes represents a paradigm of a skin commensal bacterium residing in all humans’ healthy skin that can also be etiologically related to acne, e.g., a non-infectious, chronic inflammatory skin disease. |
The interaction of C. acnes with other microorganisms including Staphylococcus epidermidis may also be implicated in acne and the term dysbiosis describes changes in the skin microbiota equilibrium. |
The increasing risk of antimicrobial resistance and the collateral damage to the steady-state microbiome with antibiotics raise important concerns about limiting their use for acne. |
Orally administered spironolactone has been used as an off-label non-antibiotic treatment for female acne owing to its anti-androgenic properties. In 2023, a pragmatic, multicenter, phase 3, double-blind randomized controlled trial on spironolactone for adult female acne was reported. |
Other non-antibiotic treatments under study for acne include PPARγ modulators, probiotics, bacteriotherapy, and C. acnes phage therapy. |
Introduction
The human skin microbiota defines the microorganisms colonizing the skin, including bacteria, fungi, viruses, and mites [1]. The term microbiome regards the composition of all microbial genes in a community [1]. The skin microenvironments are grouped into three categories: sebaceous/oily (such as forehead, scalp, chest, back), moist/humid (such as armpit, antecubital fossa, inguinal crease, popliteal fossa, umbilicus, gluteal crease, plantar heel, interdigital space), and dry (forearms, back of the elbow, buttock, and front of the knee/legs) [2]. In healthy skin, the microbial communities consist of four major phyla, namely Actinobacteria, Firmicutes, Proteobacteria, and Bacteroidetes. Their proportions are dependent on age, sex, lifestyle, and on body area-specific topographical characteristics including pH, temperature, and sebum content [1,2,3,4,5,6,7]. Culture-based and 16S rRNA methods have shown that in healthy, sebaceous-rich body areas, the skin microflora consists primarily of Cutibacterium, followed by Staphylococcus and Malassezia species [2, 8, 9].
The commensal bacterium Cutibacterium acnes (C. acnes, previously termed Propionibacterium acnes) has a key role in the pathogenesis of acne vulgaris (called acne hereafter). C. acnes acts in interplay with three other major pathogenetic factors, namely androgen-dependent hyperseborrhea, follicular keratinocyte hyperproliferation, and inflammation [10, 11]. Acne preferentially affects body areas rich in sebaceous glands, such as the face and trunk [10]. In sebaceous body sites, the follicular microenvironments are dominated by Cutibacterium species, particularly C. acnes [1]. A study described that there is population fragmentation of C. acnes within each pore on the face and that is dominated by a population of C. acnes typically differing by fewer than one mutation [12].
Staphylococcus epidermidis (S. epidermidis) is the most abundant skin-colonizing coagulase-negative staphylococcus (CoNS). It is a permanent inhabitant of human skin and colonizes predominantly the axillae, head, and nares. Some exciting roles for this commensal are being described. Commensal staphylococci are important to regulate C. acnes homeostasis in healthy human skin. Microbial interference has been shown to be a contributor to healthy skin homeostasis and staphylococcal strains may exhibit anti-C. acnes activities [13]. Also, S. epidermidis may exert probiotic-like function by preventing colonization by pathogenic bacteria such as Staphylococcus aureus [14].
In this review, we provide an update on the skin microbiome in acne and highlight the implications for acne treatments. We describe how current approved acne treatments may affect the skin microbiome and highlight the risk of antimicrobial resistance and the dysregulation of the microbiome equilibrium with the use of antibiotics for acne. Furthermore, we discuss new, off-label and emerging non-antibiotic pharmacological acne treatments with the aim to maintain the skin microbiome balance.
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Cutibacterium acnes in Acne Pathogenesis
The human skin commensal bacterium C. acnes is a Gram-positive, non-spore-forming, anaerobic bacillus that resides mainly in the microaerophilic deeper portions of healthy follicles, in contact with follicular keratinocytes and cells in the sebaceous duct [15]. C. acnes was first implicated as a direct cause of acne in 1896. However, in the early 1960s, it was identified as part of the normal human skin flora and in similar concentrations between patients with acne and individuals without acne [16, 17]. In this context, C. acnes represents a paradigm of a skin commensal bacterium residing in the human healthy skin that can also be etiologically related to acne (e.g., a non-infectious, chronic inflammatory skin disease).
Several non-infectious, inflammatory and immunomodulatory properties of C. acnes in acne pathophysiology have been elucidated. C. acnes may increase local inflammation by inducing innate immune cells to secrete pro-inflammatory cytokines including tumor necrosis factor-α (TNFα), interleukin (IL)-6, IL-8, IL-12, in part through Toll-like receptor 2 (TLR2) signaling [16, 18]. It can induce IL-1β secretion in human monocytes through the nucleotide oligomerization domain (NOD)-like receptor signaling, and in particular the NLRP3 inflammasome [19]. C. acnes can promote Th17/Th1 responses from T cells and trigger the production of IL-17A and interferon-γ (INFγ) in vitro [20, 21]. The treatment of keratinocytes with lipoteichoic acid and peptidoglycan, which are components of the coat of gram-positive bacteria, stimulates the production of neutrophil chemoattractant cytokines TNFα and IL-8, via TLR2 activation [22, 23]. C. acnes may modulate the differentiation of keratinocytes and regulate lipogenesis and sebum production from sebocytes, and induce reactive adipogenesis in dermal fibroblasts [16, 21, 24,25,26]. Reactive adipogenesis—a process in which skin fibroblasts can undergo localized proliferation and differentiation into a preadipocyte lineage in response to bacteria stimuli—was shown in human acne lesions, while C. acnes induced reactive adipogenesis via TLR2 in mice. Adipocytes, in turn, mount an innate immune defense response that may contribute to acne pathophysiology [26].
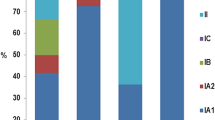
There are six phylotypes of C. acnes: IA1, IA2, IB, IC, II, and III. Multi-locus sequence typing (MLST) and single-locus sequence typing (SLST) identified further subgroups among phylotypes, called clonal complexes (CCs) [27,28,29]. Distinct “acnegenic” C. acnes phylotypes and a loss of C. acnes phylotype diversity are associated with acne [17, 27, 30, 31]. A case–control study reported loss of C. acnes phylotype diversity in patients with severe inflammatory acne, with a predominance of phylotype IA1 and SLST-type A1, compared to healthy controls [31]. On the other hand, a small study in 29 patients with mild acne compared to 34 patients with severe acne showed that the phylotype IA1 SLST type A1 was the predominant type in both groups [32]. Similarly, Guo et al. did not detect differences in species diversity between healthy individuals and those with mild or severe acne in a total of 34 college students in China. In that study, 16S and 18S rRNA gene sequencing was used to investigate Propionibacterium, Staphylococcus, Corynebacterium, and Malassezia genera [33]. Studies in Japanese patients have reported the phylotype IA2 as strongly acne-associated, suggesting geographic differences between Europe and Asia [34, 35]. The results on strain comparisons should be interpreted with caution, as they may depend on different sampling methods and the use of culture-based methods or amplicon-based sequencing and shotgun metagenomics [1, 22, 36].
The coexistence of C. acnes with other microorganisms may also be implicated in acne and the term dysbiosis was introduced to describe changes in the skin microbiota equilibrium [1]. C. acnes possesses diverse strategies of follicular niche competition, including bacteriocins acnecin and cutimycin, and propionic acid [37]. The interaction of C. acnes with Staphylococcus species is discussed in the following section.
Staphylococcus Species in Acne Pathogenesis
The effect of the dysbiosis between C. acnes and S. epidermidis was studied by Dagnelie et al. In that study, different bacterial ratios, favoring either C. acnes or S. epidermidis, or neutral ratios (1:1), were cultured with human skin explants. A higher inflammatory response was detected in the presence of dysbiosis (unbalance) favoring either C. acnes or S. epidermidis, compared to the neutral state. In addition, S. epidermidis showed a higher induction of IL-6, an innate immunity marker with pro-Inflammatory effects [38]. In another study, a strain of Staphylococcus capitis (S. capitis E12) was found to selectively inhibit the growth of C. acnes with potency greater than antibiotics. Phenol soluble modulins were secreted from S. capitis E12 and acted as antimicrobial peptides selectively against C. acnes on pig skin and on mice, without killing other commensal skin bacteria [39].
Beyond their interaction with C. acnes, some strains of skin commensal CoNS can produce bacteriocins and selectively inhibit nonresident pathogenic bacteria such as S. aureus, group A streptococci, and Escherichia coli on the skin [40].
Approved Acne Treatments: Effects on the Microbiome
Can currently approved acne treatments affect the microbiome? Topical acne treatments recommended in the current European and UK National Institute for Health and Care Excellence (NICE) guidelines include azelaic acid and topical fixed dose combinations of an antibiotic with a retinoid, or an antibiotic with benzoyl peroxide (BPO) or combination of adapalene with BPO [41, 42]. In both guidelines, oral acne treatments include antibiotics and isotretinoin, and in the European guidelines hormonal anti-androgens are recommended for female patients as an alternative treatment with a low strength of recommendation [41, 42]. Among these acne treatments, an effect on the skin microbiome has been shown for BPO, orally administered isotretinoin, and, as expected, for oral and topical antibiotics. Also, weak antimicrobial efficacy has been suggested for azelaic acid [43,44,45]. Consequently, there is an unmet need for additional non-antibiotic treatments for moderate and severe acne [46,47,48].
BPO does not induce bacterial resistance and shows a well-established bactericidal non-antibiotic action. In addition, BPO has been shown to reduce antibiotic-resistant C. acnes strains. BPO 5% gel treatment in patients with acne significantly reduced the surface and follicular C. acnes after 2 days of treatment, suggesting usefulness of short-course treatment to reduce the carriage of antibiotic-resistant C. acnes [49]. BPO had a bactericidal effect in vitro against both antibiotic-resistant and antibiotic-susceptible C. acnes. The minimum contact time needed in vitro was 60 min, 15 min, and 30 s, with concentrations of 1.25%, 2.5%, and 5%, respectively. The median minimum inhibitory concentration (MIC) of BPO did not significantly differ between antibiotic-resistant and nonresistant C. acnes [50]. It has been recommended to consider a course of topical BPO, of at least 5 to 7 days, between antibiotic courses with the aim to reduce the emergence of cutaneous resistant strains [43]. Another study in 33 patients with acne compared to 19 healthy controls investigated microbial diversity with a high-throughput sequencing targeting the V3-V4 region of 16S RNA genes. This study reported that BPO 5% gel for 12 weeks resulted in reduction of microbial diversity and reduction of the prevalence of the genus Cutibacterium [51].
Orally administered isotretinoin does not have direct antimicrobial actions, but has indirect effects by reducing sebum production. As early as 1981, while orally administered 13-cis-retinoic acid (isotretinoin) was being evaluated for acne in clinical trials, Weissmann et al. reported its effect on bacterial skin flora in five patients. Samples were collected by the “detergent scrub” technique and bacteria were identified by colony morphology, Gram stain, and coagulase test. There was a decrease in the density of propionibacteria and aerobic cocci in vivo during treatment. There was no direct inhibition of bacterial growth by 13-cis-retinoic acid in vitro, as assessed by a MIC assay. The authors attributed the decrease in bacterial counts to the isotretinoin-induced change in sebum production [52]. In 1982, King et al. showed that the decrease in sebum was the first to occur with orally administered isotretinoin, followed by a secondary decrease in propionibacteria [53]. Further studies showed that C. acnes declined through oral treatment with isotretinoin, and that isotretinoin significantly increased alpha- and beta-diversity [54]. Interestingly, the effects of orally administered isotretinoin on C. acnes were reported to be associated with the clinical response. Nolan et al. showed that the decrease of C. acnes was significantly higher in responders compared to those with minimal improvement (31% versus no decrease, respectively) after 5 months of orally administered isotretinoin therapy [55]. Also, specific shifts in C. acnes strain composition in the pilosebaceous follicle were correlated with the clinical response after 5 months of orally administered isotretinoin [55].
The place of antibiotics for acne treatment and their effects on the microbiome will be discussed separately in the following sections.
Use and Overuse of Antibiotics for Acne Treatment
The impact of oral antibiotics on the risk of antimicrobial resistance has put into question whether antibiotics could be replaced by non-antibiotic treatments. Current European guidelines (version 2016) on the management of acne recommend topical antibiotics as fixed-dose combinations, with different strengths of recommendation, for all types of acne vulgaris (mild to moderate papulopustular, severe papulopustular, moderate nodular, severe nodular acne), except for comedonal acne [41]. Also, systemic antibiotics, and in particular orally administered doxycycline or lymecycline, have a place in the treatment of all types of acne, except for comedonal acne [41]. In addition, the 2021 NICE guidelines recommend topical clindamycin in a topical fixed dose combination as first-line treatment for mild to severe acne vulgaris, and in combination with oral treatments for moderate or severe acne [42]. Orally administered doxycycline or lymecycline were the antibiotics recommended as first-line systemic treatment for moderate and severe acne, underscoring a central place for antibiotics in the treatment of acne [42]. In addition to the guideline recommendations, real-world data from clinical practice shows the extensive use of oral antibiotics as a treatment for acne. A large UK study used primary care electronic health record data (2004–2019) on the use of long-term oral antibiotics, tetracyclines, macrolides, or trimethoprim for acne vulgaris. Long-term use was defined as a course duration of 28 days or longer. Out of 217,410 people with an acne diagnosis within the previous year, 96,703 patients (45%) received a long-term antibiotic. The majority of patients (55%) were treated for a duration of 42–90 days. However, a considerable proportion (26.1%) were treated for longer than 90 days during their first course of oral antibiotic, of whom 7.3% were treated for longer than 6 months. Most (80%) of first antibiotic prescriptions for acne were with a tetracycline. Almost 60% of patients subsequently had a repeat oral antibiotic course with a median of four long-term antibiotic courses during a median follow-up of 5.3 years [56].
In an effort to reduce the risk of antimicrobial resistance, sarecycline, a third-generation tetracycline-class antibiotic with a structural modification at hydrocarbon C7, was developed. Sarecycline was approved by the US Food and Drug Administration (FDA) in 2018 for the treatment of inflammatory lesions of non-nodular moderate to severe acne vulgaris in patients aged 9 years and older. It has the advantage of a narrow spectrum of action; it is active against Gram-positive bacterial while showing reduced activity against Gram-negative bacteria [57, 58].
The aforementioned findings show that antibiotics still have a central role in the treatment of acne in guidelines and in clinical practice. However, antibiotics have considerable effects on the microbiome which will be discussed in the following section.
Antibiotics for Acne Treatment: Effects on the Microbiome
Antimicrobial resistance is an increasing public health problem worldwide. Ross et al., in a European study in six countries, showed that 66% of patients with acne had resistant propionibacteria. Combined resistance to erythromycin and clindamycin was much more common than resistance to tetracyclines. Also, 64% of dermatologists were colonized on the face with resistant propionibacteria, including all those who specialized in treating acne. In contrast, none of 27 physicians working in other outpatient departments harbored resistant propionibacteria [59]. C. acnes resistance to antibiotics has been described in various countries over the years and a detailed overview is outside the scope of this article and has been reviewed elsewhere [15, 60].
Even short-term oral or topical antibiotic therapy may have considerable effects on other skin microbes, apart from C. acnes. A 4-week orally administered minocycline treatment in four patients with acne investigated changes in the skin microbiota with 16S ribosomal RNA gene sequencing. After the 4-week course, there was reduction in C. acnes and Lactobacillus species, and increase in the relative abundance of Pseudomonas species and Streptococcus species. After a follow-up of 8 weeks, the changes in relative abundance of Streptococcus and Lactobacillus species persisted despite the discontinuation of minocycline [61]. In a randomized study in 208 patients with acne, the application of topical 2% erythromycin gel for 12 weeks (versus vehicle) resulted in an increased prevalence of erythromycin-resistant coagulase-negative staphylococci on the face which persistent up to 24 weeks, even after treatment was discontinued [62].
Beyond the skin, antibiotics used for acne may affect non-skin microbiota and may be associated with upper respiratory tract infections. A UK retrospective cohort study reported that 71.7% of 118,496 patients with acne received a topical or oral antibiotic for more than 6 weeks. The risk of upper respiratory tract infections developing within the first year of observation was 2.15 times higher among antibiotic users versus non-users [63].
New Approved Non-antibiotic Pharmacological Treatments for Acne
The increasing risk of antimicrobial resistance and the collateral damage to the steady-state microbiome with antibiotics raise important concerns about limiting their use for acne [41]. In thise context, new topical non-antibiotic pharmacological treatments have been approved for acne (Table 1). Trifarotene is a selective retinoic acid receptor (RARγ) agonist, with comedolytic, anti-inflammatory, and antipigmenting properties. Two double-blind randomized controlled trials of 12-week trifarotene 0.005% cream for moderate acne showed efficacy in reducing inflammatory and non-inflammatory acne lesions [64]. The long-term safety and increasing efficacy were shown with trifarotene application for up to 1 year [65]. Trifarotene 0.005% cream was approved by the European Medicines Agency (EMA) in 2020 for the treatment of facial and truncal acne in patients aged 9 years and older. Clascoterone is a first-in-class topical anti-androgen for acne. It competes with androgens, especially dihydrotestosterone, for androgen-receptor binding. The efficacy and safety of clascoterone 1% cream were investigated in two phase 3 randomized clinical trials [66]. Also, the long-term safety of 9-month application of clascoterone cream was reported by Eichenfield et al. Topical adverse events were mainly mild and included erythema, scaling, and itching. There were no reported adverse events in vital signs or laboratory results, or any systemic adverse events, such as reduced libido or feminization in male patients [67]. Clascoterone 1% cream was FDA approved in 2020, for acne in patients aged 12 years and older [66].
Emerging Non-antibiotic Pharmacological Treatments Used Off-Label for Acne
Spironolactone antagonizes aldosterone action on the mineralocorticoid/aldosterone receptor. It was first used as a potassium-sparing diuretic for arterial hypertension and it is also an androgen receptor antagonist [68] (Table 1). Orally administered spironolactone has been used as an off-label treatment for acne in female adolescents and adults, mostly without a medical history of polycystic ovary syndrome [69,70,71,72]. It causes gynecomastia in male individuals.
Spironolactone in vitro and ex vivo induces the specific rapid proteolytic degradation of xeroderma pigmentosum group B (XPB) protein, a DNA translocase. XPB is part of the larger protein complex transcription factor II-H (TFIIH) that plays an important role in the removal of UV photoproducts from DNA and in the initiation of transcription [73]. In vitro studies with keratinocytes and ex vivo studies with skin explants showed that spironolactone can deplete these cells and tissues of XPB, inhibit UV photoproduct removal, and increase mutagenesis [74, 75]. Notably, orally administered spironolactone is rapidly metabolized by the liver into compounds, including canrenone and 7α-thiomethylspironolactone, that did not affect XPB protein levels in the study by Kemp et al. [74, 76]. A systematic review of clinical trials of topically administered spironolactone for acne identified five clinical trials with a total of 195 female and male patients. Concentrations of 1% to 5% spironolactone were applied for a treatment duration ranging from 8 to 24 weeks. Effectiveness in acne improvement was shown, but each trial included a small number of patients, had considerable risk of bias, and a long-term follow-up was not available [77, 78].
There is an official FDA warning regarding possible tumorigenicity of spironolactone that is based mainly on animal studies using oral doses up to 150 times greater than human doses and reporting the development of hepatic, thyroid, testicular, and breast adenomas [79]. The systematic review and meta-analysis by Bommareddy et al. in 2022 included studies in 4,528,332 patients treated with spironolactone and reported prostate, breast, ovarian, kidney, gastric, and esophageal cancers. In that meta-analysis, there was no statistically significant association between spironolactone use and breast, ovarian, bladder, kidney, gastric, or esophageal cancers, while there was a decreased risk of prostate cancer [80].
Santer et al. performed a large pragmatic, multicenter, phase 3, double-blind randomized controlled trial on orally administered spironolactone for acne [81]. That trial included 410 women with facial acne warranting oral antibiotics, and patients received spironolactone or placebo during a blinded period of 24 weeks. The dose of spironolactone was 50 mg/day until week 6 and then increased to 100 mg/day until week 24. In this pragmatic trial, after the 12-week phase, participants in both groups could receive usual care, such as oral antibiotics, hormonal treatment, or isotretinoin, if judged necessary. In both groups, spironolactone or placebo was stopped at 24 weeks, treatment was unblinded, and patients could seek any treatment they wished afterwards [81].
In that study, the primary endpoint, of at least 2 points difference between groups in the mean difference of the acne-specific quality of life, was not met at 12 weeks, but it was achieved at 24 weeks (end of treatment). Regarding secondary endpoints, the PGA (participant’s global assessment) success was not significantly different with spironolactone compared to placebo at 12 weeks, but it became significantly higher at 24 weeks. The investigator’s global assessment (IGA) success score was significantly higher with spironolactone compared to placebo at 12 weeks. Adverse events were collected with questionnaire up to 24 weeks; most were mild, and there was significantly higher frequency of at least one adverse reaction with spironolactone versus placebo (64% versus 51%), driven mainly by dizziness/vertigo/light headedness (19% versus 12%) and headache (20% versus 12%) [81].
Non-Antibiotic Pharmacological Treatments Under Study for Acne
Other non-antibiotic pharmacological treatments under study for acne include PPARγ modulators, probiotics, bacteriotherapy and phage therapy.
Targeting PPARγ
Peroxisome proliferator-activated receptors (PPARs) are members of the nuclear hormone receptor family and regulate the transcription of genes involved in lipid metabolism in adipose tissue, liver, and skin. PPARγs are expressed in an immortalized human sebocyte cell line (SZ95), in isolated human sebaceous glands, in human skin (in epidermis, hair follicles, sebaceous ducts, and sebaceous glands), and in SEB-1 sebocytes [82]. PPARγ activators resulted in differentiation of keratinocytes and inhibition of cutaneous inflammation in keratinocytes in mice [83].
The PPARγ modulator N-acetyl-GED0507-LEVO (NAC-GED) was reported by Ottaviani et al. to induce PPARγ expression and promote sebocyte differentiation in vitro [84]. It modified and improved sebum composition in vitro, and resulted in lower IL-1α level, decreased lipid peroxidation, and lower levels of sapienic acid [84]. In vivo treatment of patients with acne with NAC-GED 1% gel reduced the inflammatory response [84]. Picardo et al., in a recent multicenter phase 2B randomized double-bind, vehicle-controlled trial, showed the efficacy and safety of NAC-GED 5% gel for patients with moderate to severe facial acne vulgaris [85]. In that study, there was a significantly higher reduction in total acne lesion counts with NAC-GED 5% gel once daily versus vehicle (− 57% versus − 33.9%) at week 12. The proportion of patients who achieved an IGA score of clear or almost clear skin with at least a 2-point reduction at week 12 was significantly higher with NAC-GED gel versus vehicle (45% versus 24%) [85] (Table 1).
Probiotics, Bacteriotherapy, and Phage Therapy
Oral probiotics are defined as live microorganisms with a potential to correct dysbiosis. They include Lactobacillus, Bifidobacterium, Lactococcus, and yeast species Saccharomyces boulardii. The small number of studies and small number of included patients with acne treated with oral probiotics do not allow for robust conclusions on their effectiveness for acne at present [86,87,88,89]. Additional considerations regard the quality of probiotics used, as they are sensitive to temperature and humidity [90].
The concept of bacteriotherapy was used by Nakatsuji et al. in patients with atopic dermatitis [91, 92]. In a phase 1 randomized clinical trial, Staphylococcus hominis A9 (ShA9), a bacterium isolated from healthy human skin, was used as a topical therapy for atopic dermatitis. ShA9 was applied for 8 days on the forearm skin of 54 patients with S. aureus-positive atopic dermatitis and resulted in a significant decrease in S. aureus, compared to vehicle [91]. Dagnelie et al. showed that the restoration of phylotype diversity of C. acnes decreased the inflammatory response in healthy skin explants in vitro (IL-1β, TLR2, IL-17, IL-6, IL-8, IL-10) [93]. The potential use of topical “healthy skin”-associated C. acnes phylotypes for maintaining and restoring the cutaneous diversity of C. acnes phylotypes may be promising research.
Bacteriophages (also known as phages) are viruses that can infect and kill bacteria. C. acnes phages are part of the microbial ecosystem on the skin and are highly conserved across individuals, showing limited genetic diversity [94]. The ratio between C. acnes phages and C. acnes was 1:20 in pilosebaceous units in healthy skin samples [17]. A future perspective could include the potential topical application of acne phage therapy; candidate phages should be lytic, non-lysogenic, and free of antibiotic resistance genes [95]. Moreover, it has been suggested that phages may be used to restore sensitivity of C. acnes strains to antibiotics, as they can exert selection pressure to resistant bacteria that present multidrug efflux pumps that in turn act as receptors for some phages [96]. Liu et al. isolated C. acnes phages from follicle samples from the skin of patients with acne and healthy individuals [97]. They showed that the bacterium-phage interactions are C. acnes lineage dependent and that almost all type I C. acnes strains were susceptible to the tested phages [97]. C. acnes phages had efficacy in treating a mouse model of C. acnes-induced acne [98] and as topical application in an acne-like mouse model in vivo [99]. In addition, Kim et al. isolated C. acnes-specific phages from clinical samples from patients with acne vulgaris and characterized the genotypic and phenotypic characteristics, including their hydrophobic properties [95].
Conclusions
The skin microbiome consists of microbes populating the human skin. The dysbiosis of the skin microbiome and a loss of diversity of C. acnes have been implicated in acne pathogenesis. Furthermore, interactions exist between cutaneous microbial species, and microorganisms can act in competition or in synergy. An imbalance between C. acnes and S. epidermidis has been described in acne. Currently approved acne treatments such as benzoyl peroxide, orally administered isotretinoin, and antibiotics may affect the skin microbiome. Antibiotics cause considerable collateral damage, inducing antimicrobial resistance and dysregulation of the microbiome equilibrium. Consequently, the interest is shifting towards new non-antibiotic pharmacological acne treatments. Spironolactone is an emerging off-label treatment for adult female patients and topical PPARγ modulation is being studied for patients with acne. The potential application of topical or oral probiotics, bacteriotherapy, and phage therapy for acne are further promising areas of future research. Understanding how the microbiome may regulate specific key pathways in the pathophysiology of acne may open the way for the design and development of new treatments for patients with acne.
References
Byrd AL, Belkaid Y, Segre JA. The human skin microbiome. Nat Rev Microbiol. 2018;16:143–55.
Grice EA, Kong HH, Conlan S, et al. Topographical and temporal diversity of the human skin microbiome. Science. 2009;324:1190–2.
Proksch E. pH in nature, humans and skin. J Dermatol. 2018;45:1044–52.
Grice EA, Kong HH, Renaud G, et al. A diversity profile of the human skin microbiota. Genome Res. 2008;18:1043–50.
Costello EK, Lauber CL, Hamady M, et al. Bacterial community variation in human body habitats across space and time. Science. 2009;326:1694–7.
Carmona-Cruz S, Orozco-Covarrubias L, Saez-de-Ocariz M. The human skin microbiome in selected cutaneous diseases. Front Cell Infect Microbiol. 2022;12:834135.
Robert C, Cascella F, Mellai M, et al. Influence of sex on the microbiota of the human face. Microorganisms. 2022;10(12):2470.
Till AE, Goulden V, Cunliffe WJ, et al. The cutaneous microflora of adolescent, persistent and late-onset acne patients does not differ. Br J Dermatol. 2000;142:885–92.
Kelhala HL, Aho VTE, Fyhrquist N, et al. Isotretinoin and lymecycline treatments modify the skin microbiota in acne. Exp Dermatol. 2018;27:30–6.
Moradi Tuchayi S, Makrantonaki E, Ganceviciene R, Dessinioti C, Feldman SR, Zouboulis CC. Acne vulgaris. Nat Rev Dis Primers. 2015;1:15029.
Dessinioti C, Tzanetakou V, Zisimou C, et al. A study of androgenic signs and disorders in Greek female patients with acne. J Eur Acad Dermatol Venereol. 2018;32:e279–82.
Conwill A, Kuan AC, Damerla R, et al. Anatomy promotes neutral coexistence of strains in the human skin microbiome. Cell Host Microbe. 2022;30:171–182.e7.
Ahle CM, Stodkilde K, Poehlein A, et al. Interference and co-existence of staphylococci and Cutibacterium acnes within the healthy human skin microbiome. Commun Biol. 2022;5:923.
Otto M. Staphylococcus epidermidis–the ‘accidental’ pathogen. Nat Rev Microbiol. 2009;7:555–67.
Dessinioti C, Katsambas A. Propionibacterium acnes and antimicrobial resistance in acne. Clin Dermatol. 2017;35:163–7.
Dessinioti C, Katsambas AD. The role of Propionibacterium acnes in acne pathogenesis: facts and controversies. Clin Dermatol. 2010;28:2–7.
Fitz-Gibbon S, Tomida S, Chiu BH, et al. Propionibacterium acnes strain populations in the human skin microbiome associated with acne. J Invest Dermatol. 2013;133:2152–60.
Kim J, Ochoa MT, Krutzik SR, et al. Activation of toll-like receptor 2 in acne triggers inflammatory cytokine responses. J Immunol. 2002;169:1535–41.
Qin M, Pirouz A, Kim MH, et al. Propionibacterium acnes induces IL-1beta secretion via the NLRP3 inflammasome in human monocytes. J Invest Dermatol. 2014;134:381–8.
Agak GW, Qin M, Nobe J, et al. Propionibacterium acnes induces an IL-17 response in acne vulgaris that is regulated by vitamin A and vitamin D. J Invest Dermatol. 2014;134:366–73.
Kistowska M, Meier B, Proust T, et al. Propionibacterium acnes promotes Th17 and Th17/Th1 responses in acne patients. J Invest Dermatol. 2015;135:110–8.
O’Neill AM, Gallo RL. Host-microbiome interactions and recent progress into understanding the biology of acne vulgaris. Microbiome. 2018;6:177.
Takeuchi O, Hoshino K, Kawai T, et al. Differential roles of TLR2 and TLR4 in recognition of gram-negative and gram-positive bacterial cell wall components. Immunity. 1999;11:443–51.
Isard O, Knol AC, Aries MF, et al. Propionibacterium acnes activates the IGF-1/IGF-1R system in the epidermis and induces keratinocyte proliferation. J Invest Dermatol. 2011;131:59–66.
Kistowska M, Gehrke S, Jankovic D, et al. IL-1beta drives inflammatory responses to propionibacterium acnes in vitro and in vivo. J Invest Dermatol. 2014;134:677–85.
O'Neill AM, Liggins MC, Seidman JS, et al. Antimicrobial production by perifollicular dermal preadipocytes is essential to the pathophysiology of acne. Sci Transl Med. 2022;14:eabh1478.
McDowell A, Gao A, Barnard E, et al. A novel multilocus sequence typing scheme for the opportunistic pathogen Propionibacterium acnes and characterization of type I cell surface-associated antigens. Microbiology. 2011;157:1990–2003.
McLaughlin J, Watterson S, Layton AM, et al. Propionibacterium acnes and acne vulgaris: new insights from the integration of population genetic, multi-omic, biochemical and host-microbe studies. Microorganisms. 2019;7:128.
Dekio I, McDowell A, Sakamoto M, et al. Proposal of new combination, Cutibacterium acnes subsp. elongatum comb. nov., and emended descriptions of the genus Cutibacterium, Cutibacterium acnes subsp. acnes and Cutibacterium acnes subsp. defendens. Int J Syst Evol Microbiol. 2019;69:1087–1092.
Platsidaki E, Dessinioti C. Recent advances in understanding Propionibacterium acnes (Cutibacterium acnes) in acne. F1000Res. 2018;7:1953. https://doi.org/10.12688/f1000research.15659.1.
Dagnelie MA, Corvec S, Saint-Jean M, et al. Decrease in diversity of Propionibacterium acnes phylotypes in patients with severe acne on the back. Acta Derm Venereol. 2018;98:262–7.
Paugam C, Corvec S, Saint-Jean M, et al. Propionibacterium acnes phylotypes and acne severity: an observational prospective study. J Eur Acad Dermatol Venereol. 2017;31:e398–9.
Guo Z, Yang Y, Wu Q, et al. New insights into the characteristic skin microorganisms in different grades of acne and different acne sites. Front Microbiol. 2023;14:1167923.
Nakase K, Hayashi N, Akiyama Y, et al. Antimicrobial susceptibility and phylogenetic analysis of Propionibacterium acnes isolated from acne patients in Japan between 2013 and 2015. J Dermatol. 2017;44:1248–54.
Nakase K, Okamoto Y, Aoki S, et al. Long-term administration of oral macrolides for acne treatment increases macrolide-resistant Propionibacterium acnes. J Dermatol. 2018;45:340–3.
Grice EA, Segre JA. The skin microbiome. Nat Rev Microbiol. 2011;9:244–53.
Bruggemann H, Salar-Vidal L, Gollnick HPM, et al. A Janus-faced bacterium: host-beneficial and -detrimental roles of Cutibacterium acnes. Front Microbiol. 2021;12:673845.
Dagnelie MA, Corvec S, Timon-David E, et al. Cutibacterium acnes and Staphylococcus epidermidis: the unmissable modulators of skin inflammatory response. Exp Dermatol. 2022;31:406–12.
O'Neill AM, Nakatsuji T, Hayachi A, et al. Identification of a human skin commensal bacterium that selectively kills Cutibacterium acnes. J Invest Dermatol. 2020;140(8):1619–28.e2.
Cogen AL, Yamasaki K, Sanchez KM, et al. Selective antimicrobial action is provided by phenol-soluble modulins derived from Staphylococcus epidermidis, a normal resident of the skin. J Invest Dermatol. 2010;130:192–200.
Nast A, Dreno B, Bettoli V, et al. European evidence-based (S3) guideline for the treatment of acne - update 2016 - short version. J Eur Acad Dermatol Venereol. 2016;30:1261–8.
Xu J, Mavranezouli I, Kuznetsov L, et al. Management of acne vulgaris: summary of NICE guidance. BMJ. 2021;374:n1800.
Gollnick H, Cunliffe W, Berson D, et al. Management of acne: a report from a global alliance to improve outcomes in acne. J Am Acad Dermatol. 2003;49:S1–37.
Bojar RA, Holland KT, Cunliffe WJ. The in-vitro antimicrobial effects of azelaic acid upon Propionibacterium acnes strain P37. J Antimicrob Chemother. 1991;28:843–53.
Cunliffe WJ, Holland KT. Clinical and laboratory studies on treatment with 20% azelaic acid cream for acne. Acta Derm Venereol Suppl (Stockh). 1989;143:31–4.
Dessinioti C, Zouboulis CC, Bettoli V, et al. Comparison of guidelines and consensus articles on the management of patients with acne with oral isotretinoin. J Eur Acad Dermatol Venereol. 2020;34(10):2229–40.
Katsambas AD, Dessinioti C. Hormonal therapy for acne: why not as first line therapy? facts and controversies. Clin Dermatol. 2010;28:17–23.
Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016;74:945–73.e33.
Bojar RA, Cunliffe WJ, Holland KT. The short-term treatment of acne vulgaris with benzoyl peroxide: effects on the surface and follicular cutaneous microflora. Br J Dermatol. 1995;132:204–8.
Boonchaya P, Rojhirunsakool S, Kamanamool N, et al. Minimum contact time of 1.25%, 2.5%, 5%, and 10% benzoyl peroxide for a bactericidal effect against Cutibacterium acnes. Clin Cosmet Investig Dermatol. 2022;15:403–9.
Zhou L, Chen L, Liu X, et al. The influence of benzoyl peroxide on skin microbiota and the epidermal barrier for acne vulgaris. Dermatol Ther. 2022;35:e15288.
Weissmann A, Wagner A, Plewig G. Reduction of bacterial skin flora during oral treatment of severe acne with 13-cis retinoic acid. Arch Dermatol Res. 1981;270:179–83.
King K, Jones DH, Daltrey DC, et al. A double-blind study of the effects of 13-cis-retinoic acid on acne, sebum excretion rate and microbial population. Br J Dermatol. 1982;107:583–90.
McCoy WH 4th, Otchere E, Rosa BA, et al. Skin ecology during sebaceous drought—how skin microbes respond to isotretinoin. J Invest Dermatol. 2019;139:732–5.
Nolan ZT, Banerjee K, Cong Z, et al. Treatment response to isotretinoin correlates with specific shifts in Cutibacterium acnes strain composition within the follicular microbiome. Exp Dermatol. 2023;32(7):955–64.
Bhate K, Mansfield KE, Sinnott SJ, et al. Long-term oral antibiotic use in people with acne vulgaris in UK primary care: a drug utilization study. Br J Dermatol. 2023;188:361–71.
Grada A, Ghannoum MA, Bunick CG. Sarecycline demonstrates clinical effectiveness against Staphylococcal infections and inflammatory dermatoses: evidence for improving antibiotic stewardship in dermatology. Antibiotics (Basel). 2022;11(6):722.
Moore A, Green LJ, Bruce S, et al. Once-daily oral sarecycline 1.5 mg/kg/day is effective for moderate to severe acne vulgaris: results from two identically designed, phase 3, randomized, double-blind clinical trials. J Drugs Dermatol. 2018;17:987–96.
Ross JI, Snelling AM, Carnegie E, et al. Antibiotic-resistant acne: lessons from Europe. Br J Dermatol. 2003;148:467–78.
Dessinioti C, Katsambas A. Antibiotics and antimicrobial resistance in acne: epidemiological trends and clinical practice considerations. Yale J Biol Med. 2022;95:429–43.
Chien AL, Tsai J, Leung S, et al. Association of systemic antibiotic treatment of acne with skin microbiota characteristics. JAMA Dermatol. 2019;155:425–34.
Mills O Jr, Thornsberry C, Cardin CW, et al. Bacterial resistance and therapeutic outcome following three months of topical acne therapy with 2% erythromycin gel versus its vehicle. Acta Derm Venereol. 2002;82:260–5.
Margolis DJ, Bowe WP, Hoffstad O, et al. Antibiotic treatment of acne may be associated with upper respiratory tract infections. Arch Dermatol. 2005;141:1132–6.
Tan J, Thiboutot D, Popp G, et al. Randomized phase 3 evaluation of trifarotene 50 mug/g cream treatment of moderate facial and truncal acne. J Am Acad Dermatol. 2019;80:1691–9.
Blume-Peytavi U, Fowler J, Kemeny L, et al. Long-term safety and efficacy of trifarotene 50 mug/g cream, a first-in-class RAR-gamma selective topical retinoid, in patients with moderate facial and truncal acne. J Eur Acad Dermatol Venereol. 2020;34:166–73.
Hebert A, Thiboutot D, Stein Gold L, et al. Efficacy and safety of topical clascoterone cream, 1%, for treatment in patients with facial acne: two phase 3 randomized clinical trials. JAMA Dermatol. 2020;156(6):621–30.
Eichenfield L, Hebert A, Gold LS, et al. Open-label, long-term extension study to evaluate the safety of clascoterone (CB-03-01) cream, 1% twice daily, in patients with acne vulgaris. J Am Acad Dermatol. 2020;83:477–85.
Corvol P, Michaud A, Menard J, et al. Antiandrogenic effect of spirolactones: mechanism of action. Endocrinology. 1975;97:52–8.
Berman HS, Cheng CE, Hogeling M. Spironolactone in the treatment of adolescent acne: a retrospective review. J Am Acad Dermatol. 2021;85:269–71.
Grandhi R, Alikhan A. Spironolactone for the treatment of acne: a 4-year retrospective study. Dermatology. 2017;233:141–4.
Garg V, Choi JK, James WD, et al. Long-term use of spironolactone for acne in women: a case series of 403 patients. J Am Acad Dermatol. 2021;84:1348–55.
Isvy-Joubert A, Nguyen JM, Gaultier A, et al. Adult female acne treated with spironolactone: a retrospective data review of 70 cases. Eur J Dermatol. 2017;27:393–8.
Alekseev S, Ayadi M, Brino L, et al. A small molecule screen identifies an inhibitor of DNA repair inducing the degradation of TFIIH and the chemosensitization of tumor cells to platinum. Chem Biol. 2014;21:398–407.
Kemp MG, Krishnamurthy S, Kent MN, et al. Spironolactone depletes the XPB protein and inhibits DNA damage responses in UVB-irradiated human skin. J Invest Dermatol. 2019;139:448–54.
Choi JH, Han S, Kemp MG. Detection of the small oligonucleotide products of nucleotide excision repair in UVB-irradiated human skin. DNA Repair (Amst). 2020;86:102766.
Ueda M, Matsuura K, Kawai H, et al. Spironolactone-induced XPB degradation depends on CDK7 kinase and SCF(FBXL18) E3 ligase. Genes Cells. 2019;24:284–96.
Afzali BM, Yaghoobi E, Yaghoobi R, et al. Comparison of the efficacy of 5% topical spironolactone gel and placebo in the treatment of mild and moderate acne vulgaris: a randomized controlled trial. J Dermatolog Treat. 2012;23:21–5.
Rehan ST, Khan Z, Abbas S, et al. Role of topical spironolactone in the treatment of acne: a systematic review of clinical trials—Does this therapy open a path towards favorable outcomes? J Dermatol. 2023;50:166–74.
Aldactone. Summary of product characteristics. Food and Drug Administration (FDA). https://www.accessdata.fda.gov/drugsatfda_docs/label/2008/012151s062lbl.pdf . Accessed Oct 7, 2023.
Bommareddy K, Hamade H, Lopez-Olivo MA, et al. Association of spironolactone use with risk of cancer: a systematic review and meta-analysis. JAMA Dermatol. 2022;158:275–82.
Santer M, Lawrence M, Renz S, et al. Effectiveness of spironolactone for women with acne vulgaris (SAFA) in England and wales: pragmatic, multicentre, phase 3, double blind, randomised controlled trial. BMJ. 2023;381:e074349.
Trivedi NR, Cong Z, Nelson AM, et al. Peroxisome proliferator-activated receptors increase human sebum production. J Invest Dermatol. 2006;126:2002–9.
Mao-Qiang M, Fowler AJ, Schmuth M, et al. Peroxisome-proliferator-activated receptor (PPAR)-gamma activation stimulates keratinocyte differentiation. J Invest Dermatol. 2004;123:305–12.
Ottaviani M, Flori E, Mastrofrancesco A, et al. Sebocyte differentiation as a new target for acne therapy: an in vivo experience. J Eur Acad Dermatol Venereol. 2020;34:1803–14.
Picardo M, Cardinali C, La Placa M, et al. Efficacy and safety of N-acetyl-GED-0507–34-LEVO gel in patients with moderate-to severe facial acne vulgaris: a phase 2B randomised double-blind, vehicle-controlled trial. Br J Dermatol. 2022;187(4):507–14.
Jung GW, Tse JE, Guiha I, et al. Prospective, randomized, open-label trial comparing the safety, efficacy, and tolerability of an acne treatment regimen with and without a probiotic supplement and minocycline in subjects with mild to moderate acne. J Cutan Med Surg. 2013;17:114–22.
Kim J, Ko Y, Park YK, et al. Dietary effect of lactoferrin-enriched fermented milk on skin surface lipid and clinical improvement of acne vulgaris. Nutrition. 2010;26:902–9.
Fabbrocini G, Bertona M, Picazo O, et al. Supplementation with lactobacillus rhamnosus SP1 normalises skin expression of genes implicated in insulin signalling and improves adult acne. Benef Microbes. 2016;7:625–30.
Dessinioti C, Dreno B. Acne treatments: future trajectories. Clin Exp Dermatol. 2020;45:955–61.
Salminen S, Collado MC, Endo A, et al. The International Scientific Association of Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of postbiotics. Nat Rev Gastroenterol Hepatol. 2021;18:649–67.
Nakatsuji T, Hata TR, Tong Y, et al. Development of a human skin commensal microbe for bacteriotherapy of atopic dermatitis and use in a phase 1 randomized clinical trial. Nat Med. 2021;27:700–9.
Nakatsuji T, Gallo RL, Shafiq F, et al. Use of autologous bacteriotherapy to treat Staphylococcus aureus in patients with atopic dermatitis: a randomized double-blind clinical trial. JAMA Dermatol. 2021;157:978–82.
Dagnelie MA, Corvec S, Saint-Jean M, et al. Cutibacterium acnes phylotypes diversity loss: a trigger for skin inflammatory process. J Eur Acad Dermatol Venereol. 2019;33:2340–8.
Farrar MD, Howson KM, Bojar RA, et al. Genome sequence and analysis of a Propionibacterium acnes bacteriophage. J Bacteriol. 2007;189:4161–7.
Kim S, Song H, Jin JS, et al. Genomic and phenotypic characterization of Cutibacterium acnes bacteriophages isolated from acne patients. Antibiotics (Basel). 2022;11:1041.
Farfan J, Gonzalez JM, Vives M. The immunomodulatory potential of phage therapy to treat acne: a review on bacterial lysis and immunomodulation. PeerJ. 2022;10:e13553.
Liu J, Yan R, Zhong Q, et al. The diversity and host interactions of Propionibacterium acnes bacteriophages on human skin. ISME J. 2015;9:2078–93.
Kim MJ, Eun DH, Kim SM, et al. Efficacy of bacteriophages in Propionibacterium acnes-induced inflammation in mice. Ann Dermatol. 2019;31:22–8.
Rimon A, Rakov C, Lerer V, et al. Topical phage therapy in a mouse model of Cutibacterium acnes-induced acne-like lesions. Nat Commun. 2023;14:1005.
Medical Writing/Editorial Assistance
This article did not use medical writing or editorial assistance.
Funding
No funding or sponsorship was received for this publication of this article.
Author information
Authors and Affiliations
Contributions
Clio Dessinioti: Conceptualization, writing of the draft manuscript, revision and approval of the final manuscript. Andreas Katsambas: writing of the draft manuscript, supervision, revision and approval of the final manuscript.
Corresponding author
Ethics declarations
Ethical Approval
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Conflict of Interest
Clio Dessinioti reports travel support for attendance of congresses by Pfizer, La Roche Posay, Bioderma NAOS, Galderma, outside the submitted work. Andreas Katsambas has nothing to disclose.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Dessinioti, C., Katsambas, A. The Microbiome and Acne: Perspectives for Treatment. Dermatol Ther (Heidelb) 14, 31–44 (2024). https://doi.org/10.1007/s13555-023-01079-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13555-023-01079-8