Abstract
Purpose
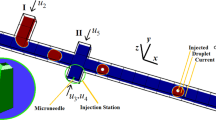
Injection is a useful technology for delivery the solution like drug in medical science, because injection amount can be simply controlled by adjusting the injection duration and pressure. With advances in fabrication technology, the small size of syringe can be made from cm to nm scale. Therefore, its application model has been also decreased to a single cell. However, in femto-scale, the precise volume measurement and the theoretical estimation by the time and the pressure becomes very challengeable, because of the dramatic change of inertial forces. In order to estimate the quantitative value of injected volume in femtoliter scale, empirical equation was established as injection pressure, duration and viscosity.
Methods
We observed the experimental results of the injected volume in femtoliter scale as a function of viscosity, duration, and pressure. Based on these results, we design and experimentally verify a theoretical equation for the quantitative control of the volume in femto-scale. The quantitative deliveries of the injection solution in a living cell were investigated.
Results
When the external material has viscosity between 80 to 12 mPa·s, injection volume is observed to vary between 22 to 46 fL. At same conditions, it was observed similarities between theoretical values and experimental results. Furthermore, fluorescent material was quantitatively injected into a living single cell. The fluorescent intensity is linearly increased with the injection volume.
Conclusions
It is possible to quantitatively control the injection volume using the empirical equation in the scale of femtoliter without any process on coating an injection solution.
Similar content being viewed by others
References
Yan R, Park JH, Choi Y, Heo CJ, Yang SM, Lee LP, Yang P. Nanowire-based single-cell endoscopy. Nat Nanotechnol. 2011; 7(3):191–6.
Yoo SM, Kang M, Kang T, Kim DM, Lee SY, Kim B. Electrotriggered, spatioselective, quantitative gene delivery into a single cell nucleus by Au nanowire nanoinjector. Nano Lett. 2013; 13(6):2431–5.
Chen X, Kis A, Zettl A, Bertozzi CR. A cell nanoinjector based on carbon nanotubes. Proc Natl Acad Sci USA. 2007; 104(20):8218–22.
Chiu DT, Lorenz RM. Chemistry and biology in femtoliter and picoliter volume droplets. Acc Chem Res. 2009; 42(5):649–58.
Grossier R, Hammadi Z, Morin R, Magnaldo A, Veesler S. Generating nanoliter to femtoliter microdroplets with ease. Appl Phys Lett. 2011; 98(9):091916.
Kwon H, Park HS, Yu J, Hong S, Choi Y. Spatio-temporally controlled transfection by quantitative injection into a single cell. Biomaterials. 2015; 67:225–31.
He M, Edgar JS, Jeffries GDM, Lorenz RM, Shelby JP, Chiu DT. Selective encapsulation of single cells and subcellular organelles into picoliter-and femtoliter-volume droplets. Anal Chem. 2005; 77(6):1539–44.
Matsuoka H, Shimoda S, Ozaki M, Mizukami H, Shibusawa M, Yamada Y, Saito M. Semi-quantitative expression and knockdown of a target gene in single-cell mouse embryonic stem cells by high performance microinjection. Biotechnol Lett. 2007; 29(3):341–50.
Funabashi H, Sugimoto Y, Saito M, Matsuoka H. A femtoinjection technique for dynamic analysis of protein function in living embryonic stem cells. Biotechnol Lett. 2012; 34(7):1257–62.
Guillaume-Gentil O, Potthoff E, Ossola D, Dörig P, Zambelli T, Vorholt JA. Force-controlled fluidic injection into single cell nuclei. Small. 2013; 9(11):1904–7.
Wefers B, Ortiz O, Wurst W, Kühn R. Generation of targeted mouse mutants by embryo microinjection of TALENs. Methods. 2014; 69(1):94–101.
Yum K, Na S, Xiang Y, Wang N, Yu M-F. Mechanochemical delivery and dynamic tracking of fluorescent quantum dots in the cytoplasm and nucleus of living cells. Nano Lett. 2009; 9(5):2193–8.
Dunkel J, Zaid IM. Noisy swimming at low Reynolds numbers. Phys Rev E. 2009; 80(2):021903.
Brody JP, Yager P, Goldstein RE, Austin RH. Biotechnology at low Reynolds numbers. Biophys J. 1996; 71(6):3430–41.
Hong J, Kim YK, Kang KH, Oh JM, Kang IS. Effects of drop size and viscosity on spreading dynamics in DC electrowetting. Langmuir. 2013; 29(29):9118–25.
Matsuoka H, Komazaki T, Mukai Y, Shibusawa M, Akane H, Chaki A, Uetake N, Saito M. High throughput easy microinjection with a single-cell manipulation supporting robot. J Biotechnol. 2005; 116(2):185–94.
Mulderry PK. Intracellular microinjection: a powerful tool in the study of neuronal gene expression and function. Methods. 1996; 10(3):292–300.
Sanger JM, Danowski BA, Sanger JW. Microinjection of fluorescently labeled alpha-actinin into living cells. Methods Mol Biol. 2000; 137:449–56.
Pfitzner J. Poiseuille and his law. Anaesthesia. 1976; 31(2):273–5.
Backer JA, Lowe CP, Hoefsloot HCJ, Iedema PD. Poiseuille flow to measure the viscosity of particle model fluids. J Chem Phys. 2005; 122(15):154503.
Chen L, Zhao Y, Zhang X-R. Poiseuille rayleigh-benard convective flow and compressible boundary effects of nearcritical fluid in microchannels. J Nanosci Nanotechnol. 2015; 15(4):3035–42.
Negishi M, Seto H, Hase M, Yoshikawa K. How does the mobility of phospholipid molecules at a water/oil interface reflect the viscosity of the surrounding oil? Langmuir. 2008; 24(16):8431–4.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Yu, J., Kwon, H., Park, Hs. et al. Femtoliter scale quantitative injection control by experimental and theoretical modeling. Biomed. Eng. Lett. 6, 250–255 (2016). https://doi.org/10.1007/s13534-016-0228-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13534-016-0228-0