Abstract
Purpose
Diabetic retinopathy (DR) is still a leading cause of blindness. The role of miRNAs in diabetic retinopathy still needs more research. We aimed at validating the role of circulating miRNAs: 21,181C and 1179 in early detection, dynamic monitoring, and management of DR.
Methods
Whole blood samples were collected from 180 diabetic patients and 60 normal individuals as control. The diabetic patients were subdivided into 60 subjects without retinopathy, 60 with non-proliferative diabetic retinopathy (NPDR), and 60 with proliferative diabetic retinopathy (PDR). Gene expression of miR-21, miR-181c, and miR-1179 were estimated in each sample using two-step reverse transcription quantitative real-time polymerase chain reaction (RT-qPCR).
Results
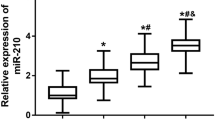
MicroRNA 181c and miRNA 1179 were significantly higher among PDR group compared to NPDR,W DM and control groups, while no significant difference was detected regarding miRNA 21. The areas under the receiver operating characteristic (ROC) curves of the validated two-serum miRNAs were 0.983 and 0.927 respectively. Combination of miRNA 1179 and miRNA 21 improved the accuracy rate to 90%. Combination of miR-181c and miR-1179 possessed high ability to discriminate between PDR and NPDR with an accuracy rate of 100%.
Conclusion
MicroRNAs play a role in pathogenesis of diabetic retinopathy. In our study, miR-181c and miR-1179 were significantly high in PDR patients compared to NPDR and controls hence can be used to anticipate and follow up the progression. MicroRNA antagonists or mimics can be tried as new medications to modify DR by reducing the rate of progression, and subsequent blindness.






Similar content being viewed by others
References
Wang W, ACY l. Diabetic retinopathy: pathophysiology and treatments. International journal of molecular sciences. 2018;19(6):1816.
Duh EJ, Sun JK, Stitt AW. Diabetic retinopathy: current understanding, mechanisms, and treatment strategies. JCI Insight. 2017;2(14):e93751–1:13.
Aiello LP, Avery RL, Arrigg PG, Keyt BA, Jampel HD, Shah ST, Pasquale LR, Thieme H, Iwamoto MA, Park JE, Nguyen HV, Aiello LM, Ferrara N, King GL. Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. N Engl J Med. 1994;331(22):1480–7.
Mace KA, Yu DH, Paydar KZ, Boudreau N, Young DM. Sustained expression of Hif-1alpha in the diabetic environment promotes angiogenesis and cutaneous wound repair. Wound Repair Regen. 2007;15:636–45. https://doi.org/10.1111/j.1524-475X.2007.00278.x.
Lee RC, Feinbaum RL, Ambros V. The C. elegans heterochronic gene Lin-4 encodes small RNAs with antisense complementarity to Lin-14. Cell. 1993;75:843–54. https://doi.org/10.1016/0092-8674(93)90529-y.
Dunmire JJ, Lagouros E, Bouhenni RA, Jones M, Edward DP. MicroRNA in aqueous humor from patients with cataract. Exp Eye Res. 2013;108:68–71.
Qing S , Yuan S, Yun C, Hui H, Mao P, Wen F, Ding Y, Liu Q (2014): Serum miRNA biomarkers serve as a fingerprint for proliferative diabetic retinopathy .;34(5):1733-1740.
Hourigan ST, Solly EL, Nankivell VA, Ridiandries A, Weimann BM, Henriquez R, Tepper ER, Zhang JQJ, Tsatralis T, Clayton ZE, Vanags LZ, Robertson S, Nicholls SJ, Ng MKC, Bursill CA, Tan JTM. The regulation of miRNAs by reconstituted high-density lipoproteins in diabetes-impaired angiogenesis. Sci. Rep. 2018;8:13596. https://doi.org/10.1038/s41598-018-32016-x.
Li Y, Sun R, Zou J, Ying Y, Luo Z. MiR-21 induced angiogenesis through AKT and ERK activation and HIF-1α expression. Cells. 19. 2019;8(7):752. https://doi.org/10.3390/cells8070752.
Martins B, Amorim M, Reis F, Ambrósio AF, Fernandes R. Extracellular vesicles and microRNA: putative role in diagnosis and treatment of diabetic retinopathy. Antioxidants (Basel). 2020;9(8):705.
Tang W, Li H, Tang J, Wu W, Qin J, Lei H, Cai P, Huo W, Li B, Rehan V, Xu X, Geng Q, Zhang H, Xia Y. Specific serum microrna profile in the molecular diagnosis of Hirschsprung’s disease. J Cell Mol Med. 2014;18(8):1580–7.
Jiang Q, Lyu X, Yuan Y, Wang L. Plasma miR-21 expression: an indicator for the severity of type 2 diabetes with diabetic retinopathy. Biosci Rep. 2017;27-37(2):BSR20160589. https://doi.org/10.1042/BSR20160589.
Alberti KG, Zimmet PZ. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet Med. 1998;1(15):539–53.
Salz DA and Witkin AJ (2015): Diabetic retinopathy update ; imaging in diabetic retinopathy. New England Eye Center, Tufts Medical Center, Boston; 22(2):145-150.
Yin ZL, Wang YL, Ge SF, Guo TT, Wang L, Zheng XM, Liu J. Reduced expression of miR-503 is associated with poor prognosis in cervical cancer. Eur Rev Med Pharmacol Sci. 2015;19:4081–5.
Shin Y, Kim DY, Ko JY, Woo YM, Park JH. Regulation of KLF12 by microRNA-20b and microRNA-106a in cystogenesis. FASEB J. 2018;32:3574–82. https://doi.org/10.1096/fj.201700923R.
Wang Y, Toh HC, Chow P, Chung AYF, Meyers DJ, Cole PA, Ooi LLPJ, Lee CGL. MicroRNA-224 is up regulated in hepatocellular carcinoma through epigenetic mechanisms. The FASEB Journal. 2012;26(7):3032–41.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real- time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25(4):402–8.
Karoli R, Fatima J, Shukla V, Garg P, Ali A. Predictors of diabetic retinopathy in patients with type 2 diabetes who have normoalbuminuria. Ann Med Health Sci Res. 2013;3(4):536–40.
Martinez B, Peplow P. MicroRNAs as biomarkers of diabetic retinopathy and disease progression. Neural Regen Res. 2019;14(11):1858–69.
Deng Y, Li S, Li S, Yu C, Huang D, Chen H, Yin X. CircPDE4B inhibits retinal pathological angiogenesis via promoting degradation of HIF-1α though targeting miR-181c. IUBMB Life. 2020;72:1920–9. https://doi.org/10.1002/iub.2307.
Usui-Ouchi A, Ouchi Y, Kiyokawa M, Sakuma T, Itol R, Ebihara N. Upregulation of Mir-21 Levels in the vitreous humor is associated with development of proliferative vitreoretinal disease. PLoS ONE. 2016;11(6):e0158043. https://doi.org/10.1371/journal.pone.0158043.
Ma J, Wang J, Liu Y, Wang C, DuanD LN, Wang K, Zhang L, Gu K, Chen S, Zhang T, You D, Han L. Comparisons of serum miRNA expression profiles in patients with diabetic retinopathy and type 2 diabetes mellitus. Clinics. 2017;72(2):111–5.
Raum P, Lamparter J, Ponto KA, Peto T, Hoehn R, Schulz A, Schneider A, Wild PS, Pfeiffer N, Mirshahi A. Correction: Prevalence and cardiovascular associations of diabetic retinopathy and maculopathy: results from the Gutenberg Health Study. PloS one. 2015;10(9):e0139527.
Availability of data and materials
All relevant raw data will be freely available to any scientist wishing to use them for non-commercial purposes without breaching participant confidentiality.
Funding
This work was financially supported by the Molecular Biology Unit, Egypt as well as authors’ contributions.
Author information
Authors and Affiliations
Contributions
Tamer Ibrahiem Salem: collecting samples and clinical data, drafted the manuscript and revision of the manuscript
Nashwa Badr Eldin: collecting samples and clinical data, drafted the manuscript and revision of the manuscript
Naglaa Fathy Alhusseini: molecular biology technique
Omnia Alsaied Abdullah: conceived and designed the research, molecular biology technique, analyzed and interpreted the data, critical revision of the manuscript and corresponding author
Nashwa Elsayed Ahmed: rewrote the parts previously been commented on by the reviewers and needed to be corrected in the manuscript
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interest.
Animal research
This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health (NIH publications 85-23, revised 2011). All protocols were approved by the institutional review board for animal experiments of the Faculty of Medicine, Benha University, Egypt.
Consent to participate and consent to publish
The research protocol was accepted by the Medical Faculty Ethical Committee, Benha University. Before participation, written informed consents were received from all patients at the study initiation, both consent to participate in the study and consent to have the data published.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Salem, T.I., Eldin, N.B., Alhusseini, N.F. et al. Expression profile of microRNAs may be promising in diagnosis of proliferative diabetic retinopathy: an Egyptian study. Int J Diabetes Dev Ctries 43, 36–44 (2023). https://doi.org/10.1007/s13410-022-01044-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13410-022-01044-9