Abstract
An alternative to mitigate the environmental impact of food waste is the separation of valuable components. Due to the difficulty and heterogeneity of such wastes, the study of model systems allowing a regular and predictable production and composition was used. A mixture of pre- and post-consumption kitchen wastes from a hospital with an automated organic matter segregation and drying system (HKW) has been collected to obtain representative samples of the different diets prepared daily. The chemical characterization of streams revealed a variation in composition of the wastes depending on the menus. Those from dinner + breakfast (D + B) presented protein content in the range 42–54% of the dry weight and those form lunch + snack (L + S) in the range 19–33%, whereas the carbohydrate content was 52–65% in D + B and 47–53% in L + S waste streams. These values suggested the possibility of proposing a different valorization scheme for each type of stream. In this study, a green extraction process with pressurized hot water under microwave heating has been explored for the solubilization of the most abundant and valuable components, protein, and carbohydrates. The starch fractions recovered from the crude extract were proposed for the formulation of anti-freezing hydrogels suitable for 3D printing, and the possibilities of valorizing other fractions such as proteins as plant biostimulants are also discussed.
Graphical abstract

Similar content being viewed by others
1 Introduction
The increasing amounts of food losses and wastes are having a negative environmental impact [13, 23, 35], and their sustainable management represents a challenge from economic and ecological points of view. Different strategies can be proposed, including prevention, mitigation, and post-valorization [8, 13, 14].
Kitchen wastes, representing one part of food wastes, are worldwide relevant and due to their concentration in some big restaurants, would serve as an excellent source of value added products owing to their organic content. However, the high heterogeneity and moisture content are challenging the further efficient utilization [35]. Kitchen waste is often mixed with municipal solid waste streams for energetic valorization; the most usual are based on thermal (gasification, pyrolysis, and incineration), chemical or biochemical (composting, anaerobic digestion) transformations [22], but different final products can be obtained either for bioconversion processes, from isolation of constituents or for the formulation of materials [26, 36, 43]. A biorefinery scheme has also been proposed to maximize both recovered nutrients and generation of high value-added products Carmona-Cabello et al. [6]. This change from the current linear economy model towards a circular economy based on the continuous valorization of waste generated would have a positive impact on the environment and also builds long-term resilience, new technologies, and jobs [5, 28, 34]. Furthermore, the transition towards bioeconomy can reduce the need of fossil fuels and contribute to the United Nations Sustainable Development Goals [6, 21].
The reduction in chemicals, time and energy, and the integral valorization of the raw materials in the framework of biorefineries are major concerns in the development of environmentally friendly processes. Hydrothermal treatment using pressurized hot water as solvent is an efficient green, scalable technology, useful both for extraction and for hydrolysis of different components. Water is found in the subcritical state at 100–374 °C and pressures up to 22 MPa, presenting unique properties, such as lower viscosity, density and dielectric constant, whereas the increased ionic product favors catalytic processes allowing shorter times and higher rates than conventional extraction [4, 32]. This technology has been proposed to enhance depolymerization of polysaccharides to oligomer and monomeric units, and for the generation of soluble protein fractions, peptides, and amino acids [2, 49]. An additional advantage is the possibility of avoiding the drying stages. This technology has also been proposed as the first stage in biorefineries of lignocellulosics [44, 47], food wastes [41], or micro and macroalgae [49]. The careful tuning is needed to maximize the extraction yield and to avoid degradation into monomeric units and decomposition products.
The whole domestic waste valorization is frequently studied using wastes from large scale kitchen, for example from restaurants. In the present study, the possibility of obtaining more representative, standardized, concentrated waste, and segregated streams corresponding to specific menus was considered a great advantage allowing a rational design of fractionation and valorization strategies. Therefore, a model hospital kitchen with a waste segregation system was selected for the collection of a mixture of the pre- and post-consumption kitchen waste samples representative of the different diets prepared. The samples have been characterized for the major components, and a green extraction technology was further evaluated for the solubilization of valuable fractions. Under conditions leading to maximal yield and concentration of protein and carbohydrate-rich fractions in the liquid extract, a potential application was tried, based on the formulation of anti-freezing hydrogels suitable for 3D printing.
2 Materials and methods
2.1 Materials
Samples from mixed pre- and post-consumption waste streams generated in the Complexo Hospitalario Universitario de Ourense (Spain) hospital kitchen were collected during the first week of January 2021. The automatic segregation system removes paper, metallic and, non-organic elements before grinding and dewatering. Each sample corresponded to different daily diets and two mixed menus, lunch and afternoon snack (L + S), and dinner and breakfast (D + B) were collected. Samples (500 g) of well-homogenized hospital kitchen wastes (HKW) with an initial moisture content of 76.86 ± 1.36% were packed in closed bags and were cold stored at − 18 °C. The defrosted samples were freeze-dried, homogenized, and milled in a Moulinex mill (MC300, 180 W). The moisture content after freeze-drying was 2.4–4.2%. Samples were further stored in hermetic plastic bags in darkness at room temperature before use.
2.2 Microwave-assisted pressurized hot water extraction
The freeze-dried and ground hospital kitchen-waste samples were mixed with distilled water at a high liquid:solid ratio 30:1 (w/w) in this initial approach. The suspensions were placed in the vials of a microwave (Monowave 450, Anton Paar, Austria), equipped with an autosampler MAS24, and heated until the selected target temperatures (140, 160, 180, and 200 °C). The operation mode consisted on a non-isothermal processing during the heating period (800 rpm, 850 W), with severity values (log R0) in the range 1.88–3.64, where \({R}_{0}=t\cdot \mathrm{exp}\left[\frac{T-100}{14.75}\right]\), being t, the reaction time (min) and T, the temperature (°C).
When the target temperature was reached, the microwave device was cooled down, and the liquid and solid phases were separated by vacuum filtration. The liquid fraction was cold stored overnight prior to separate the precipitated starch fraction [42]. Both dried starch (air convective oven, 40 °C, 24 h) and remaining liquid fractions were further analyzed. Extracts containing soluble components were concentrated to a final volume concentration ratio 3 in a 5-kDa ultrafiltration membrane (Merk-Millipore, Germany).
2.3 Analytical methodology
2.3.1 Chemical characterization
Moisture and ash content were determined gravimetrically. For moisture content determination, samples were placed in an air convective oven at 105 °C for 48 h, whereas for the ash content analysis, a muffle was used at 575 °C for 6 h. Protein content was estimated from the total nitrogen content value (Kjeldahl method) using the universal factor 6.25. Soluble protein was measured incubating samples with Bradford reagent (Sigma, Spain) for 20 min at room temperature and reading absorbance at 595 nm in relation to a standard curve prepared with bovine serum albumin (BSA) (Sigma, Spain). Mineral content was determined on the microwave (Marsxpress, CEM, USA)-assisted acid digestion of ashes (HNO3 and H2O2 at 1600 W, 15 min and 200 °C, 10 min). The contents of Na and K were obtained by atomic emission spectrophotometry, whereas atomic absorption spectroscopy was used for Ca, Fe, B, Cu, and Mg employing a 220 Fast sequential spectrophotometer (Varian, Palo Alto, CA). Cd and Pb contents were determined by inductively coupled plasma mass spectrometry (ICP-MS) (X Series, Thermo Scientific, USA). Hg was obtained by cold vapor atomic absoprtion spectroscopy (CVAAS).
The saccharidic composition of the samples was determined after an acid digestion (72% sulfuric acid, 30 °C, 60 min). The sample hydrolysis continued in an autoclave with sulfuric acid at 4% (121 °C, 60 min). The liquid phase was separated by filtration (0.45 μm) and analyzed by high performed liquid chromatography (1100 series, Agilent, Germany) in an Aminex HPX-87H (300 × 7.8 mm, BioRad, Hercules, CA, USA) column at 60 °C with 0.003 M H2SO4 at 0.6 mL/min as mobile phase.
The total lipid content of the KW samples was performed following the Folch method [15]. Briefly, the mixtures were homogenized with 20 parts of chloroform:methanol (2:1), for one part of sample and centrifuged for 10 min at 3000 rpm and 15 °C. After filtering, 5 mL of H2O was added, forming a biphase that was separated by centrifugation. The lipid content was gravimetrically determined once the chloroform was removed by rotary evaporation (320 mbar at 40 °C).
The total phenolic content was determined using the Folin–Ciocalteau reagent (1 N) and sodium carbonate at 20%. Samples were incubated in darkness at room temperature for 60 min, and the absorbance was measured at 730 nm against a distilled water blank and a standard curve with gallic acid (Sigma, Spain).
Total starch content and amylose/amylopectin ratio were calculated using two enzymatic kits (Megazyme, Co., Wicklow, Ireland) following standard procedures [1].
2.3.2 Antioxidant properties
A representative radical scavenging capacity assay was selected, with the ABTS [2,2-azinobis(3-ethyl-benzothiazoline-6-sulfonate)] radical cation (ABTS·+) [31]. After incubation of samples with the ABTS·+ solution for 6 min, the absorbance was read at 734 nm, and values were expressed as TEAC value (Trolox equivalent antioxidant capacity).
2.4 Anti-freezing starch-based hydrogels
2.4.1 Formulation
Isolated starch (moisture content: 10.3%) was used to formulate anti-freezing hydrogels based on the recommendations previously reported [48]. Starch was gelatinized at room temperature using the soluble extract fractions obtained after hydrothermal treatment. The biopolymer (15%, w/w) was dispersed in the hydrothermal liquid phase. The mixture was gelatinized under gentle stirring for 60 min at 25 °C. Starch solutions were incubated in a freezer at − 18 °C for 24 h to develop the anti-freezing hydrogels.
2.4.2 3D printing
To illustrate the possibility of obtaining printable anti-freezing hydrogels, 3D printing trials were conducted in a Foodini 3D printer (Natural Machines, Barcelona, Spain). Two printed models with cylindrical study (15-mm diameter × 30-mm height) and spherical study (20-mm diameter) shapes were designed using Foodini creator software. The above starch/hydrothermal liquid phase solutions feed were dosed using a syringe extruder with 0.8-mm nozzle after being gellified into the printing tube at 25 °C. The printing settings were directly assigned as a new material using as the main parameters, the printing speed (1800 mm/min), ingredient flow (0.6), and gap between layers (2 mm). Several preliminary tests were performed to optimize the printing conditions taking into account the subsequent rheological features, according to the recommendations given in previous studies [10, 48]. The printed systems were placed at − 18 °C for 24 h to ensure full hydrogel maturation previously to further testing.
2.4.3 Water retention measurements
The water retention ability test of prepared hydrogels was made according to the method previously reported [39]. Briefly, starch hydrogels were cut into cylinders (15.0-mm diameter, 10.0-mm height) and placed at room temperature. The corresponding dehydration kinetics of the hydrogels were monitored for 2 weeks, following the weight changes to calculate the water content (%) = (initial wet mass − dry mass) / initial wet mass × 100.
2.4.4 Rheological behavior
Rheological testing of the starch hydrogels, in terms of elastic (G′) and viscous (G″) moduli, was conducted on a controlled-stress rheometer (MCR302, Anton Paar, Austria) using a sand-blasted parallel plate (25-mm diameter, 1-mm gap) equipped with a Peltier temperature control (− 20 to 200 °C). Frequency sweeps were performed at 10 Pa within the linear viscoelastic range (< 25 Pa) at − 18 and 25 °C.
2.5 Statistical analysis
Above experimental measurements were made at least in triplicate. One-factor analysis of variance, ANOVA, was used to statistically analyze the experimental data sets by means of PASW Statistics v.22 (IBM SPSS Statistics, New York, USA) software. A post hoc Scheffé test was made to discriminate means (95% confidence, p < 0.05), when the variance analysis indicated differences between averages.
3 Results and Discussion
The hospital selected for the study has an innovative automated segregation, grinding, mixing-pressing, and dewatering system. The lower water content in the waste material facilitates storage due to lower risks of microbial contamination and is easier to manage and transport. The possibility of periodically segregating the waste streams from different cooking batches, corresponding to well characterized and standardized diets, converts this kitchen hospital in a unique and reference model for the evaluation of novel alternatives of valorization of the different wastes.
3.1 Proximal characterization
Adequate residue classification and detailed characterization are needed to define further valorization routes [8, 45]. Each collected sample represents a mixture of two meals, dinner-breakfast (D + B) and lunch-afternoon snack (L + S), and each sample correspond to each of the different diets formulated. Before chemical characterization, sample content was described according to a typology classification of the major components (Table 1). Visual characterization confirmed the presence of both organic material and non-edible inorganic fractions, such as meat and fish bones. The particular origin of these wastes, which are generated in the preparation or pre-consumption and post-consumption stages and partially segregated, makes them different from other kitchen wastes, collected in different restaurants, which included non-edible fractions, such as paper [8].
The chemical characterization of the dried samples is shown in Table 2 and provides valuable information regarding the major groups (lipids, protein, carbohydrates). The ash content was under 3% in samples 1, 2, 11, and 13 but accounted for more than 8% in samples 3, 5, and 8, which contained some pieces from fish and chicken bones. Intermediate ash content was found in samples containing vegetable or fruit fractions, such as samples 4 and 12, which were comparable to the values in different restaurants and food services and lower than in households [8, 12].
The mineral content of samples was highly diverse among samples (Table 3). The calcium content ranged from 2.3 g/kg in sample 11 to 33.3 g/kg in sample 8. Also samples 5, 3, and 9 show high content and presence of animal and fish bones. The sodium content, between 0.12 and 0.26% of the dry weight is lower than for other restaurants, 0.33%, but not as low despite the lower processed and salt content in the diet, but to the presence of fish wastes in samples 3 and 5. This was also probably the reason, with the presence of leafy greens of the high Mg content, ranging from 395 ppm in sample 13 to 1267 ppm in sample 5, whereas in kitchen restaurants was 228–1800 ppm [8, 18]. The K content ranged from 1.4 g/kg in sample 2 to 4.5 g/kg in sample 9, in the range of those reported for other KW [8],this mineral can be found naturally in many foods, especially in unprocessed food, such as fruits, vegetables, milk, and whole grains.
The total carbon content varied between 43 and 63% of the dry weight. Comparable values have been found in hostel KW [20] and have been reported for other restaurants (50–55%) [8]. The C/N ratios in the range 8.3–14.0 have been observed in the samples from dinner-breakfast wastes, whereas ratios of 4.9–7.2 were found in lunch-snack wastes. Correspondingly, the protein content was high in KW from L + S, in the range 42.2% in sample 7 to 59.2% in sample 9. In HKW from D + B, the protein content ranged from 29.7% in sample 2 to 33.4% in sample 10. The latter samples showed values higher than those reported for household wastes, and also the lunch-snack ones are even higher [12]. C/N ratio can provide information for the valorization process, particularly those based on the biodegradation and bioconversion. Ratios in the range 13–16 have been reported, with a maximum for Italian and lower for grill restaurant [8].
Carbohydrate content of the HKW samples ranged over the range of 44 to 65%, being the highest values corresponding with dinner-breakfast wastes. Dias-Ferreira et al. [11] reported the highest waste associated to the main course and the bread, where high carbohydrate content can be found. This value is also higher in menus with fruits [8]. Data from kitchen wastes generated in different restaurants showed carbohydrate content for 44–51% (dry basis), this group being composed of simple sugar, starch, and fiber [8]. Lower value ranges were identified in household (40–55%) and in food service (35–55%) [12].
The lipid content ranged from 6–7% in samples 1, 9 and 13 to 8.6% in sample 12 and 10% in sample 3. These values are expectedly lower than in other kitchen wastes from different restaurants, in the range 17–33% [8, 12, 29] due to the presence of frying oils and sauces. Comparable values have been reported for cafeteria and household wastes [3, 30]. The lower lipid fraction in the hospital samples was due to the lower fat content of diets in a hospital compared to other types of restaurants and to the previous separation of cooking oil to be recycled in the municipal waste collection system. Further analysis of lipid classes in this study was not addressed.
Based on the information obtained in the characterization study, some suggestions could be proposed. The possibility of an early segregation of pre-consumption wastes, either corresponding to specific diets or to their mixture, could offer a great advantage allowing a rational design of fractionation and valorization strategies.
3.2 The yield of extract, total phenolic content and starch recovery
The potential of separation and fractionation operations for the selective recovery of valuable components was tested, using an aqueous-based process. Alternatively, solvent extraction for lipid recovery has been reported [7]. In the present study, a process allowing the direct use of the KW without previous drying, the use of water as solvent and the feasibility of obtaining valuable products from the protein and carbohydrate fractions were explored. For this purpose, the utilization of an aqueous-based extraction and depolymerization technique was chosen for designing flexible and efficient fractionation processes.
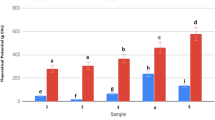
The extraction yield and composition of the extracts obtained in the range of final heating temperatures studied for the different HKW samples have been given in Table 4. The influence of severity during microwave-assisted extraction on the total extraction yield (a), protein content (b), and phenolic content (c) is shown in Fig. 1. Except for samples 2 and 6, the maximum extraction yield was obtained operating at the highest severity tested, during heating up to 200 °C. This trend was also observed for samples with high protein content (> 50%), 1, 3, 9, and 13. Up to 60% of the initial sample could be solubilized for samples 1 and 13, whereas 53 and 58% were attained in samples 3 and 9. Extraction yields higher than 70% were observed for samples 4 and 6.
Soluble starch was also recovered from the extracts after cold selective solid–liquid separation obtained at the two lowest tested final heating temperatures, accounting for 8.8 and 5.4 g/100 g extract from samples 8 and 1, respectively. Starch yield seemed to depend more on the diet than on the meal. The isolated biopolymer exhibited high total starch content of 90.5 ± 2.3% and amylose content of 21.2 ± 3.1% comparable with those reported for commonly used starches (e.g., potato, rice, corn) [51, 52]. Literature data regarding starch content have shown a wide range of values for food wastes (bread, rice, or pasta) from restaurant sector (16.2–29.4%, w/w) [8], kitchen wastes (17.4%) [46], or other food waste sources (11.5–42.1%) [16]. Although higher starch content was identified, lower total starch content was reported for the last two works with values of 77.7–80.3% and 16.3%, respectively. Despite an increasing protein extraction yield is expected with increasing temperature, the concentration in the extract is maximal at 140–160 °C due to the increase in non-proteinic compounds at higher temperatures.
As a general trend, the total phenolic content in the extract increased with temperature regardless of the diet and the meal. The content in the extract was low, up to 1.5–2.8% of the dry extract. A concomitant trend for the ABTS radical scavenging capacity was found, but the values were very low. The ABTS radical scavenging capacity assay was used to follow the solubilization of phytoconstituents from vegetable-rich samples and also could correlate with the properties of bioactive peptides solubilized during the subcritical water-extraction stage. Since protein hydrolysates and bioactive peptides exhibit a range of health-promoting properties [25]. Bioactive peptides and hydrolysates have been produced from food wastes and byproducts from terrestrial animals, fish and their byproducts [50]. Further studies of detailed biological and functional characterization are ongoing.
3.3 Valorization of the soluble fractions
For the commercial implementation of an integrated biorefinery, the design of advanced, eco-friendly, and economically profitable units is needed. The biorefining opportunities related to organic wastes rely on high-value added products associated to some emerging markets, such as (i) chemical basic-platforms, (ii) bio-based products and commodities for sustainable and renewable farming systems, such as soil amendment, plant biostimulant, and biofertilizer), and (iii) innovative biomaterials, such as bioplastics. The preliminary potential application as protein hydrolysates and as bioplastics could be suggested for two of the streams evaluated.
3.3.1 Formulation of 3D printable anti-freezing hydrogels
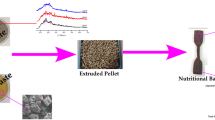
The flow diagram of the proposed valorization of selected pre- and post-consumption hospital kitchen wastes for a representative rich-starch fractions, containing the highest values of this biopolymer (sample 8, processed up to 140 °C), is shown in Fig. 2. The starch present in this D + B waste material was efficiently recovered with the proposed treatment. Biopolymers derived from renewable resources are an emerging class of advanced materials useful as adhesives, gums, binders, and emulsions. Among these biopolymers are starch and proteins with ability to tailor materials with desirable properties [19]. Starch blends are considered new economy bio-based plastics [38]. Hydrogels are attracting extensive attention due to their biocompatibility, flexibility, multifunctionality, and porosity, for food, bioengineering, soft electronics, and agricultural applications. Starch is one of the most economical and available materials for preparing hydrogels, but becoming fragile and losing elasticity at subzero temperatures. One approach to develop anti-freezing starch hydrogels that can maintain their strength and elasticity even at subzero temperatures is the formulation of water/glycerol mixtures, and also the presence of salts in the water could lower the freezing point. Anti-freezing hydrogels with the addition of CaCl2 could be formed into different 3D structures and could broaden the application of starch in various fields. Anti-freezing starch bio-based hydrogels have been attracting extensive attention for their excellent water retention ability and for the formulation of food, adhesives, medical materials, and intelligent materials [48]. Commonly, glycerol and CaCl2 were concurrently introduced to improve its mechanical, thermal, and conductive properties. These materials are highly demanded as high performing, cost effective, and safe for wearable electronic devices [24] and power sources [51, 52] with potential as substrates for transient electronics. Starch could lower the electronic waste caused by non-biodegradable and non-reusable electronic components in flexible electronic devices, due to its advantages of biodegradability, cost effectiveness, abundance, and renewability.
In this context, a selected starch-rich HKW samples was evaluated for printable anti-freezing hydrogels using the corresponding soluble extract fractions obtained after hydrothermal treatment. The rheological features of formulated and printed anti-freezing hydrogels measured at both subzero (− 18 °C) and room temperature (25 °C) have been given in Fig. 3a. All the samples exhibited predominant elastic behavior, with G′ > G″, over the tested frequency range. Both elastic and viscous moduli were almost frequency invariant, indicating a typical gel-like behavior [41]. Slight frequency dependence was identified for G″ modulus above 2.5 Hz. At fixed angular frequency, G′ and G″ moduli values were slightly higher for samples measured at subzero temperature, which is consistent with the gel network reinforcement expected at lower temperatures. This suggests that proposed KW starch-based hydrogels can maintain adequate gel mechanical performance at subzero temperatures. Note here that all printed hydrogels exhibited similar mechanical profiles, with slightly lower viscoelastic properties (Fig. 3b). Anyway, the magnitude of G′ and G″ moduli indicated intermediate strength gels, with mechanical behavior at both tested temperatures with better compressive than those obtained by the traditional hydrothermal method [48]. These results suggest that the components present in the hydrothermal liquid phases (minerals as calcium, lipids, or proteins) used here for the anti-freezing hydrogel development favor the hydrophilic cross-linking, without the extra-addition of CaCl2 or glycerol [17].
The ability of the proposed anti-freezing hydrogels to hold water in the environment was also assessed. For both formulated and printed starchy hydrogels, the water content drop from 100% at the initial stage to 75% (1 week), and then decreased slowly to 60% (2 weeks). The observed water-hold capacities were higher than those previously reported for novel anti-freezing hydrogels from potato and corn starches incorporated with CaCl2 formulated at subzero temperatures, where incubation times of 1 week at room temperature involved 55% of water and 17–25% for those control hydrogels placed in water with the same incubation time [48]. Later, the authors explained that smaller pores of the network formed at subzero temperature, which trapped water inside the hydro gel network, can help to rise the water holding ability. Ions as Ca2+ present in the hydrothermal liquid phase used as solvent have a high charge density and can form strong electrostatic interactions with water molecules, which could have reduced the free water content in the hydrogel [41].
Overall, the outcomes demonstrated that it could be possible to design anti-freezing printable hydrogels into different 3D structures as spheres or cylinders with adequate rheological performance (Fig. 3b). It should be remarked that developed hydrogels were transparent, smooth, and stereo, which is indicative of the quick prototyping ability of anti-freezing starch-based hydrogels.
3.3.2 Soluble protein and hydrolysates
The flow diagram corresponding to processing a selected protein-rich HKW (sample 9, processed up to 160 °C) has been presented in Fig. 4. The corresponding soluble extracts were further concentrated in a 5-kDa cut-off ultrafiltration membrane operating in concentration mode to a final volume concentration ratio 3. Both retentate and permeate were collected and analyzed. Under these conditions permeate with 71% peptides under 5 kDa was obtained, and the carbohydrate content was 13%. The retentate contained 51% protein and 26% carbohydrates.
Protein hydrolysates obtained from recycling wastes of plant or animal origin are good candidates to be used as plant biostimulants because of their high amino acids and soluble peptide concentrations. Biostimulants are at the moment one of the most eco-friendly sustainable strategy to increase crop production reducing the use of chemicals. Vegetable- and animal-derived protein hydrolysates are interesting biostimulants for their ability to enhance plant growth and mitigate the effects of environmental stresses [37]. Their composition is influenced by the source of proteins and the hydrolytic processes (chemical-thermal and/or enzymatic hydrolysis) utilized. Whereas enzymatically produced vegetable protein hydrolysates are mainly characterized by signaling peptides, the free amino acids are the main bioactive molecules in those from animal origin chemically produced, which are more effective at low doses, because at high doses plants need to counteract the negative effects of Na and Cl at toxic levels [33]. The enzymatically hydrolyzed animal protein-based biostimulants, represent a cost-effective approach to alleviate the negative effects of several stresses in horticultural crops, by stimulating the biosynthesis of specific hormonal pathways [9]. Conventional extraction of protein from biomass relies on acid or alkaline extraction and the further production of protein hydrolysates, composed of large peptides, small peptides, and free aminoacids, can be addressed by chemical, enzymatic, or microbial hydrolysis. The enzymatic hydrolysis offers advantages to be performed under mild conditions with precise control of the degree of hydrolysis, minimizing side reactions and the presence of toxic chemicals in the products. The development of efficient and scalable methods avoiding chemicals, enzymes, and prolonged times and could be addressed using water under subcritical conditions [27]. This is a technically efficient, inexpensive, scalable and environmentally friendly technology although less selective [2].
In this work, high amounts of protein have been identified < 5 kDa (0.250-kg protein). This could make lunch + snack HKW attractive candidates for biostimulants, since it was reported that those samples with higher protein and peptide content under 3 kDa exhibited higher impact on the soil biological properties [40]. This was related to the easier assimilation of low molecular weight proteins by soil microorganisms promoting the vegetation establishment, which can protect the soil erosion. Further studies to optimize the potential of these applications and also to advance in the utilization of the insoluble solid phase are ongoing.
4 Conclusions
The results from the present study represent a green approach to the management and valorization of kitchen wastes. A hospital kitchen with reproducible menus and with an automated organic matter segregation, grinding and drying system was selected for exploring a waste valorization scheme and two potential applications adapted to the composition of the different streams. The pressing and drying preconditioning automated stages and the higher repeatability of menus represent a model waste ideal for studying value-added solutions. However, further studies on the seasonal variation are needed and ongoing.
The studied hospital kitchen wastes showed a higher protein and lower lipid content than other kitchen wastes reported in literature. A marked difference among the different diets served in the hospital was found, the protein content being higher in the lunch-snack samples and the carbohydrate content in the dinner-breakfast ones. Microwave-assisted extraction using water as solvent solubilized between 18 and 72% samples and those with higher starch and protein content was used for the formulation of hydrogels potentially useful for 3D printable non-freezable gels.
Data availability
The data generated or analyzed during this study are included in this article.
References
AACC (2010). Approved methods of analysis (11th edn.). American Association of Cereal Chemistry, Eagan, Minnesota, USA
Álvarez-Viñas M, Rodríguez-Seoane P, Flórez-Fernández N, Torres MD, Díaz-Reinoso B, Moure A, Domínguez H (2021) Subcritical water for the extraction and hydrolysis of protein and other fractions in biorefineries from agro-food wastes and algae: a review. Food Bioprocess Technol 14(3):373–387
Bibra M, Rathinam NK, Johnson GR, Sani RK (2020) Single pot biovalorization of food waste to ethanol by Geobacillus and Thermoanaerobacter spp. Renew Energy 155:1032–1041
Brunner G (2009) Near critical and supercritical water. Part I. Hydrolytic and hydrothermal processes. J Supercrit Fluids 47:373–381
Bruno SF, Ekorong FJAA, Karkal SS, Cathrine MSB, Kudre TG (2019) Green and innovative techniques for recovery of valuable compounds from seafood by-products and discards: a review. Trends Food Sci Technol 85:10–22
Carmona-Cabello M, Garcia IL, Leiva-Candia D, Dorado MP (2018) Valorization of food waste based on its composition through the concept of biorefinery. Curr Opin Green Sustain Chem 14:67–79
Carmona-Cabello M, Sáez-Bastante J, Pinzi S, Dorado MP (2019) Optimization of solid food waste oil biodiesel by ultrasound-assisted transesterification. Fuel 2551:115817
Carmona-Cabello M, García IL, Sáez-Bastante J, Pinzi S, Koutinas AA, Dorado MP (2020) Food waste from restaurant sector – characterization for biorefinery approach. Bioresource Technology 301:122779
Casadesús A, Pérez-Llorca M, Munné-Bosch S, Polo J (2020) An enzymatically hydrolyzed animal protein-based biostimulant (pepton) increases salicylic acid and promotes growth of tomato roots under temperature and nutrient stress. Front Plant Sci 11:953
Diañez I, Gallegos C, Brito-de la Fuente E, Martínez I, Valencia C, Sánchez MC, Diaz MJ, Franco JM (2019) 3D printing in situ gelification of κ-carrageenan solutions. Food Hydrocolloids 87:321–330
Dias-Ferreira C, Santos T, Oliveira V (2015) Hospital food waste and environmental and economic indicators – a Portuguese case study. Waste Manage 46:146–154
Dou Z, Toth JD (2021) Global primary data on consumer food waste: rate and characteristics - a review. Resour Conserv Recycl 168:105332
Esteban J, Ladero M (2018) Food waste as a source of value-added chemicals and materials: a biorefinery perspective. Int J Food Sci Technol 53(5):1095–1108
Filimonau V, Ermolaev VA (2021) A sleeping giant? Food waste in the foodservice sector of Russia. J Clean Prod 297:126705
Folch J, Lees M, Sloane Stanley GH (1957) A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem 226:497–509
Fung L, Urriola PE, Baker L, Shurson GC (2019) Estimated energy and nutrient composition of different sources of food waste and their potential for use in sustainable swine feeding programs. Transl Anim Sci 3:48–57
Hedayati S, Majzoobi M, Shahidi F, Koocheki A, Farahnaky A (2016) Effects of NaCl and CaCl2 on physicochemical properties of pregelatinized and granular cold-water swelling corn starches. Food Chem 213:602–608
Ho KS, Chu LM (2019) Characterization of food waste from different sources in Hong Kong. J Air Waste Manag Assoc 69:277–288
Imam SH, Bilbao-Sainz C, Chiou BS, Glenn GM, Orts WJ (2013) Biobased adhesives, gums, emulsions, and binders: current trends and future prospects. J Adhes Sci Technol 27:1972–1997
Jayalakshmi J, Joseph K, Sukumaran V (2009) Bio hydrogen generation from kitchen waste in an inclined plug flow reactor. Int J Hydrogen Energy 34:8854–8858
Kamal H, Le CF, Salter AM, Ali A (2021) Extraction of protein from food waste: an overview of current status and opportunities. Compr Rev Food Sci Food Saf 20:2455–2475
Karmee SK (2016) Liquid biofuels from food waste: current trends, prospect and limitation. Renew Sust Energ Rev 53:945–953
López-Gómez JP, Pérez-Rivero C, Venus J (2020) Valorisation of solid biowastes: the lactic acid alternative. Process Biochem 99:222–235
Lu J, Gu J, Hu O, Fu Y, Ye D, Zhang X, Zheng Y, Hou L, Liu H, Jiang X (2021) Highly tough, freezing-tolerant, healable and thermoplastic starch/poly(vinyl alcohol) organohydrogels for flexible electronic devices. J Mater Chem A 9(34):18406–18420
Mada SB, Ugwu CP, Abarshi MM (2020) Health promoting effects of food-derived bioactive peptides: a review. Int J Pept Res Ther 26:831–848
Mahjoub B, Domscheit E (2020) Changes and challenges of an organic waste-based bioeconomy. Current Opinion in Green and Sustainable Chemistry 25:100388. https://doi.org/10.1016/j.cogsc.2020.100388
Marcet I, Álvarez C, Paredes B, Díaz M (2016) The use of sub-critical water hydrolysis for the recovery of peptides and free amino acids from food processing wastes. Review of sources and main parameters. Waste Manage 49:364–371
Maschmeyer T, Luque R, Selva M (2020) Upgrading of marine (fish and crustaceans) biowaste for high added-value molecules and bio(nano)-materials. Chem Soc Rev 49(13):4527–4563
Peinemann JC, Demichelis F, Fiore S, Pleissner D (2019) Techno-economic assessment of non-sterile batch and continuous production of lactic acid from food waste. Bioresource Technol 289:121631
Prasoulas G, Gentikis A, Konti A, Kalantzi S, Kekos D, Mamma D (2020) Bioethanol production from food waste applying the multienzyme system produced on-site by Fusarium oxysporum F3 and mixed microbial cultures. Fermentation 6:39
Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C (1999) Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Biol Med 26:1231–1237
Rogalinski T, Liu K, Albrecht T, Brunner G (2008) Hydrolysis kinetics of biopolymers in subcritical water. J Supercrit Fluids 46:335–341
Rouphael Y, Carillo P, Cristofano F, Cardarelli M, Colla G (2021) Effects of vegetal- versus animal-derived protein hydrolysate on sweet basil morpho-physiological and metabolic traits. Sci Horticult 284:110123
Sharma P, Gaur VK, Sirohi R, Varjani S, Kim SH, Wong JWC (2021) Sustainable processing of food waste for production of bio-based products for circular bioeconomy. Bioresource Technol 325:124684
Sindhu R, Gnansounou E, Rebello S, Binod P, Varjani S, Thakur IS, Nair RB, Pandey A (2019) Conversion of food and kitchen waste to value-added products. J Environ Manage 241:619–630
Sindhu R, Manju A, Mohan P, Rajesh RO, Madhavan A, Arun KB, Hazeena SH, Mohandas A, Rajamani SP, Puthiyamadam A, Binod P, Reshmy R (2020) Valorization of food and kitchen waste: an integrated strategy adopted for the production of poly-3-hydroxybutyrate, bioethanol, pectinase and 2, 3-butanediol. Bioresour Technol 310:123515
Sitohy MZ, Desoky E-SM, Osman A, Rady MM (2020) Pumpkin seed protein hydrolysate treatment alleviates salt stress effects on Phaseolus vulgaris by elevating antioxidant capacity and recovering ion homeostasis. Sci Horticult 27120:109495
Spierling S, Knüpffer E, Behnsen H, Mudersbach M, Krieg H, Springer S, Albrecht S, Herrmann C, Endres H-J (2018) Bio-based plastics - a review of environmental, social and economic impact assessments. J Clean Prod 185:476–491
Sui X, Guo H, Chen P, Zhu Y, Wen C, Gao Y, Yang J, Zhang X, Zhang L (2020) Zwitterionic osmolyte-based hydrogels with antifreezing property, high conductivity, and stable flexibility at subzero temperature. Adv Funct Mater 30:1907986
Tejada M, Benítez C, Gómez I, Parrado J (2011) Use of biostimulants on soil restoration: effects on soil biochemical properties and microbial community. Appl Soil Ecol 49:11–17
Torres MD, Fradinho P, Raymundo A, Sousa I, Falqué E, Domínguez H (2021) The key role of thermal waters in the development of innovative gelled starch-based matrices. Food Hydrocolloids 117:106697
Torres MD, Fradinho P, Rodríguez P, Falqué E, Santos V, Domínguez H (2020) Biorefinery concept for discarded potatoes: recovery of starch and bioactive compounds. J Food En 275:109886
Torres-León C, Chávez-González ML, Hernández-Almanza A, Martínez-Medina GA, Ramírez-Guzmán N, Londono-Hernández L, Aguilar CN (2021) Recent advances on the microbiological and enzymatic processing for conversion of food wastes to valuable biopoducts. Curr Opin Food Sci 38:40–45
Vila C, Campos AR, Cristovão C, Cunha AM, Santos V, Parajó JC (2008) Sustainable biocomposites based on autohydrolysis of lignocellulosic substrates. Compos Sci Technol 68:944–952
Withanage SV, Dias GM, Habib K (2021) Review of household food waste quantification methods: focus on composition analysis. J Clean Prod 279:123722
Wu M, Chen W, Hu J, Tian D, Shen F, Zeng Y, Yang G, Zhang Y, Deng S (2019) Valorizing kitchen waste through bacterial cellulose production towards a more sustainable biorefinery. Sci Total Environ 695:133898
Xu J, Zhou P, Dai L, Gui Y, Yuan L, Shen X, Zhang C, Huo K (2021) A scalable waste-free biorefinery inspires revenue from holistic lignocellulose valorization. Green Chem 23(16):6008–6019
Yang Z, Chen X, Xu Z, Ji N, Xiong L, Sun Q (2021) Anti-freezing starch hydrogels with superior mechanical properties and water retention ability for 3D printing. Int J Biol Macromol 190:382–389
Zainan NH, Thiruvenkadam S, Danquah MK, Harun R (2020) Biochemical analysis and potential applications of aqueous and solid products generated from subcritical water extraction of microalgae Chlorella pyrenoidosa biomass. J Appl Phycol 32:111–126
Zamora-Sillero J, Gharsallaoui A, Prentice C (2018) Peptides from fish by-product protein hydrolysates and its functional properties: an overview. Mar Biotechnol 20:118–130
Zhang B, Xiao Y, Wu X, Luo F, Lin Q, Ding Y (2021) Changes in structural, digestive, and rheological properties of corn, potato, and pea starches as influenced by different ultrasonic treatments. Int J Biol Macromol 185:206–218
Zhang Y, Chen Y, Alfred M, Huang F, Liao S, Chen D, Li D, Wei Q (2021) Alkaline sodium polyacrylate-starch hydrogels with tolerance to cold conditions for stretchable zinc-air batteries. Compos Part B Eng 224:109228
Acknowledgements
We acknowledge the CACTI (Centre of Scientific & Technological Support to the Research — University of Vigo — Spain) for their support with mineral determinations.
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature. The study received financial support from Xunta de Galicia (Centro Singular de Investigación de Galicia accreditation 2019–2022) and the European Union (European Regional Development Fund — ERDF) — Ref. ED431G2019/06. M. D. T. received funding from the Spanish Ministry of Economy and Competitiveness for her postdoctoral grant (RYC2018-024454-I) and Xunta de Galicia for the project (ED431F 2020/01).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Esteban-Lustres, R., Torres, M.D., Pazos, A. et al. Preliminary evaluation of pressurized hot water extraction for the solubilization of valuable components from hospital kitchen wastes. Biomass Conv. Bioref. 14, 4621–4633 (2024). https://doi.org/10.1007/s13399-022-02557-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-022-02557-3