Abstract
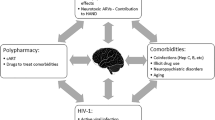
Highly active antiretroviral treatment has led to unprecedented efficacy and tolerability in people living with HIV. This effect was also observed in the central nervous system with the nowadays uncommon observation of dementias; yet in more recent works milder forms are still reported in 20–30% of optimally treated individuals. The idea of a subclinical neuronal toxicity induced by antiretrovirals has been proposed and was somehow supported by the late-emerging effects associated with efavirenz use. In this manuscript we are reviewing all the potential mechanisms by which antiretroviral drugs have been associated with in vitro, ex vivo, or in vivo toxicity to cells pertaining to the central nervous system (neurons, astrocytes, oligodendrocytes, and endothelial cells). These include direct or indirect effects and pathological pathways such as amyloid deposition, damage to small cerebral vessels, and impairment in neurotransmission. The aim of this review is therefore to provide a detailed description of the available literature in order to guide further clinical research for improving patients’ neurocognition and quality of life.

Similar content being viewed by others
References
Achim C, Adame A, Dumaop W (2009) Increased accumulation of intraneuronal amyloid beta in HIV-infected patients. J NeuroImmune Pharmacol 4(2):190–199. https://doi.org/10.1038/jid.2014.371
Akay C, Cooper M, Odeleye A, Jensen BK, White MG, Vassoler F, Gannon PJ, Mankowski J, Dorsey JL, Buch AM, Cross SA, Cook DR, Peña MM, Andersen ES, Christofidou-Solomidou M, Lindl KA, Zink MC, Clements J, Pierce RC, Kolson DL, Jordan-Sciutto KL (2014) Antiretroviral drugs induce oxidative stress and neuronal damage in the central nervous system. Journal of NeuroVirology 20(1):39–53. https://doi.org/10.1007/s13365-013-0227-1
Akay-Espinoza, C., Stern, A. L., Lee, R. N., Panvelker, N., Li, J., & Jordan-Sciutto, K. L. (2017). Differential in vitro neurotoxicity of antiretroviral drugs. Conference on Retroviruses and Opportunistic Infections, Poster 378
Amin, J., Becker, S., Belloso, W., Boffito, M., Cooper, D., Crabtree-Ramirez, B., … Dolan, M. (2015). Efficacy and safety of efavirenz 400 mg daily versus 600 mg daily: 96-week data from the randomised, double-blind, placebo-controlled, non-inferiority ENCORE1 study. Lancet Infect Dis, 15(7), 793–802. https://doi.org/10.1016/S1473-3099(15)70060-5
Andersson LM, Hagberg L, Fuchs D, Svennerholm B, Gisslén M (2001) Increased blood-brain barrier permeability in neuro-asymptomatic HIV-1-infected individuals - correlation with cerebrospinal fluid HIV-1 RNA and neopterin levels. Journal of NeuroVirology 7(6):542–547. https://doi.org/10.1080/135502801753248123
Arenas-Pinto A, Grund B, Sharma S, Martinez E, Cummins N, Fox J et al (2018) Risk of suicidal behavior with use of efavirenz: results from the strategic timing of antiretroviral treatment trial. Clin Infect Dis 67(3):420–429. https://doi.org/10.1093/cid/ciy051
Ayuso P, Neary M, Chiong J, Owen A (2019) Meta-analysis of the effect of CYP2B6, CYP2A6, UGT2B7 and CAR polymorphisms on efavirenz plasma concentrations. J Antimicrob Chemother 74(11):3281–3290. https://doi.org/10.1093/jac/dkz329
Bacq A, Balasse L, Biala G, Guiard B, Gardier AM, Schinkel A, Louis F, Vialou V, Martres MP, Chevarin C, Hamon M, Giros B, Gautron S (2012) Organic cation transporter 2 controls brain norepinephrine and serotonin clearance and antidepressant response. Mol Psychiatry 17(9):926–939. https://doi.org/10.1038/mp.2011.87
Bertrand L, Dygert L, Toborek M (2016) Antiretroviral treatment with efavirenz disrupts the blood-brain barrier integrity and increases stroke severity. Sci Rep 6(December):1–15. https://doi.org/10.1038/srep39738
Bertrand L, Velichkovska M, Toborek M (2019) Cerebral vascular toxicity of antiretroviral therapy. J NeuroImmune Pharmacol. https://doi.org/10.1007/s11481-019-09858-x
Bonfanti P, Madeddu G, Gulminetti R (2017) Discontinuation of treatment and adverse events in an Italian cohort of patients on dolutegravir We. Aids 31:455–457
Borghetti A, Baldin G, Capetti A (2017) Efficacy and tolerability of dolutegravir and two nucleos(t) ide reverse transcriptase inhibitors in HIV-1- positive, virologically suppressed patients. Aids 31(3):457–459. https://doi.org/10.1097/QAD.0000000000001351
Borghetti A, Calcagno A, Lombardi F, Cusato J, Belmonti S, D’Avolio A et al (2019) SLC22A2 variants and dolutegravir levels correlate with psychiatric symptoms in persons with HIV. J Antimicrob Chemother 74(4):1035–1043. https://doi.org/10.1093/jac/dky508
Cahn P, Pozniak AL, Mingrone H, Shuldyakov A, Brites C, Andrade-Villanueva JF, Richmond G, Buendia CB, Fourie J, Ramgopal M, Hagins D, Felizarta F, Madruga J, Reuter T, Newman T, Small CB, Lombaard J, Grinsztejn B, Dorey D, Underwood M, Griffith S, Min S (2013) Dolutegravir versus raltegravir in antiretroviral-experienced, integrase-inhibitor-naive adults with HIV: week 48 results from the randomised, double-blind, non-inferiority SAILING study. Lancet 382(9893):700–708. https://doi.org/10.1016/S0140-6736(13)61221-0
Calcagno A, Atzori C, Romito A, Vai D, Audagnotto S, Stella ML, Montrucchio C, Imperiale D, di Perri G, Bonora S (2016) Blood brain barrier impairment is associated with cerebrospinal fluid markers of neuronal damage in HIV-positive patients. Journal of NeuroVirology 22(1):88–92. https://doi.org/10.1007/s13365-015-0371-x
Calcagno A, Romito A, Atzori C, Ghisetti V, Cardellino C, Audagnotto S, Scarvaglieri E, Lipani F, Imperiale D, di Perri G, Bonora S (2017) Blood brain barrier impairment in HIV-positive naïve and effectively treated patients: immune activation versus astrocytosis. J NeuroImmune Pharmacol 12(1):187–193. https://doi.org/10.1007/s11481-016-9717-2
Centner, C. M., Bateman, K. J., & Heckmann, J. M. (2013). Manifestations of HIV infection in the peripheral nervous system. The Lancet Neurology, 12(3), 295–309. https://doi.org/10.1016/S1474-4422(13)70002-4
Chaganti J, Marripudi K, Staub LP, Rae CD, Gates TM, Moffat KJ, Brew BJ (2019) Imaging correlates of the blood-brain barrier disruption in HIV-associated neurocognitive disorder and therapeutic implications. Aids 33(12):1843–1852. https://doi.org/10.1097/QAD.0000000000002300
Ciccarelli N, Fabbiani M, Di Giambenedetto S, Fanti I, Baldonero E, Bracciale L et al (2011) Efavirenz associated with cognitive disorders in otherwise asymptomatic HIV-infected patients. Neurology 76(16):1403–1409. https://doi.org/10.1212/WNL.0b013e31821670fb
De Boer MGJ, Van Den Berk GEL, Van Holten N, Oryszcyn JE, Dorama W, Ait Moha D, Brinkman K (2016) Intolerance of dolutegravir-containing combination antiretroviral therapy regimens in real-life clinical practice. Aids 30(18):2831–2834. https://doi.org/10.1097/QAD.0000000000001279
Decloedt EH, Maartens G (2013) Neuronal toxicity of efavirenz: a systematic review. Expert Opin Drug Saf 12(6):841–846. https://doi.org/10.1517/14740338.2013.823396
Dorjee K, Choden T, Baxi SM, Steinmaus C, Reingold AL (2018) Risk of cardiovascular disease associated with exposure to abacavir among individuals with HIV: a systematic review and meta-analyses of results from 17 epidemiologic studies. Int J Antimicrob Agents 52(5):541–553. https://doi.org/10.1016/j.ijantimicag.2018.07.010
Elzi L, Erb S, Furrer H, Cavassini M, Calmy A, Vernazza P et al (2017) Adverse events of raltegravir and dolutegravir. Aids 31(13):1853–1858. https://doi.org/10.1097/QAD.0000000000001590
Eugenin EA, Clements JE, Christine Zink M, Berman JW (2011) Human immunodeficiency virus infection of human astrocytes disrupts blood-brain barrier integrity by a gap junction-dependent mechanism. J Neurosci 31(26):9456–9465. https://doi.org/10.1523/JNEUROSCI.1460-11.2011
Fettiplace A, Stainsby C, Winston A, Givens N, Puccini S, Vannappagari V, Hsu R, Fusco J, Quercia R, Aboud M, Curtis L (2017) Psychiatric symptoms in patients receiving dolutegravir. J Acquir Immune Defic Syndr 74(4):423–431. https://doi.org/10.1097/QAI.0000000000001269
Gatanaga H, Hayashida T, Tsuchiya K, Yoshino M, Kuwahara T, Tsukada H, Fujimoto K, Sato I, Ueda M, Horiba M, Hamaguchi M, Yamamoto M, Takata N, Kimura A, Koike T, Gejyo F, Matsushita S, Shirasaka T, Kimura S, Oka S (2007) Successful Efavirenz dose reduction in HIV type 1-infected individuals with cytochrome P450 2B6 *6 and *26. Clin Infect Dis 45(9):1230–1237. https://doi.org/10.1086/522175
Giunta B, Ehrhart J, Obregon DF, Lam L, Le L, Jin J et al (2011) Antiretroviral medications disrupt microglial phagocytosis of -amyloid and increase its production by neurons: implications for HIV-associated neurocognitive disorders. Molecular Brain 4(1):23. https://doi.org/10.1186/1756-6606-4-23
Green DA, Masliah E, Vinters HV, Beizai P, Moore DJ, Achim CL (2005) Brain deposition of beta-amyloid is a common pathologic feature in HIV positive patients. Aids 19(4):407–411. https://doi.org/10.1097/01.aids.0000161770.06158.5c
Grilo NM, João Correia M, Miranda JP, Cipriano M, Serpa J, Matilde Marques M, Monteiro EC, Antunes AMM, Diogo LN, Pereira SA (2017) Unmasking efavirenz neurotoxicity: time matters to the underlying mechanisms. Eur J Pharm Sci 105(December 2016):47–54. https://doi.org/10.1016/j.ejps.2017.05.010
Gutierrez J, Albuquerque ALA, Falzon L (2017) HIV infection as vascular risk: a systematic review of the literature and meta-analysis. PLoS One 12(5):1–15. https://doi.org/10.1371/journal.pone.0176686
Haas DW, Ribaudo HJ, Kim RB, Tierney C, Wilkinson GR, Gulick RM et al (2004) Pharmacogenetics of efavirenz and central nervous system side effects: an adult AIDS Clinical Trials Group study. Aids 18(18):2391–2400
Haddow LJ, Laverick R, Daskalopoulou M, McDonnell J, Lampe FC, Gilson R et al (2018) Multicenter European prevalence study of neurocognitive impairment and associated factors in HIV positive patients. AIDS Behav 22(5):1573–1583. https://doi.org/10.1007/s10461-017-1683-z
Heaton RK, Clifford DB, Franklin DR, Woods SP, Ake C, Vaida F et al (2010) HIV-associated neurocognitive disorders persist in the era of potent antiretroviral therapy: Charter Study. Neurology 75(23):2087–2096. https://doi.org/10.1212/WNL.0b013e318200d727
Hinckley S, Sherman S, Best B, Momper J, Ma Q, Letendre S, … Z-score M. (2016). Neurotoxicity screening of antiretroviral drugs with human iPSC-derived neurons high content screening high content screening mitochondrial toxicity of antiretroviral drugs neurite outgrowth during antiretroviral drug treatment. Conference on Retroviruses and Opportunistic Infections, poster 395
Hoffmann C, Welz T, Sabranski M, Kolb M, Wolf E, Stellbrink HJ, Wyen C (2017) Higher rates of neuropsychiatric adverse events leading to dolutegravir discontinuation in women and older patients. HIV Medicine 18(1):56–63. https://doi.org/10.1111/hiv.12468
Jensen B, Monnerie H, Mannell M (2015) Altered oligodendrocyte maturation and myelin maintenance: the role of anti-retrovirals in HIV-associated neurocognitive disorders. J Neuropathol Exp Neurol 74(11):1093–1118. https://doi.org/10.1016/j.physbeh.2017.03.040
Johnson DH, Gebretsadik T, Shintani A, Mayo G, Acosta EP, Stein CM, Haas DW (2013) Neuropsychometric correlates of efavirenz pharmacokinetics and pharmacogenetics following a single oral dose. Br J Clin Pharmacol 75(4):997–1006. https://doi.org/10.1111/j.1365-2125.2012.04454.x
Jones DL, Rodriguez VJ, Alcaide ML, Weiss SM, Peltzer K (2020) The use of efavirenz during pregnancy is associated with suicidal ideation in postpartum women in rural South Africa. AIDS Behav 23(1):126–131. https://doi.org/10.1007/s10461-018-2213-3.The
Latronico T, Pati I, Ciavarella R, Fasano A, Mengoni F, Lichtner M, Vullo V, Mastroianni CM, Liuzzi GM (2018) In vitro effect of antiretroviral drugs on cultured primary astrocytes: analysis of neurotoxicity and matrix metalloproteinase inhibition. J Neurochem 144(3):271–284. https://doi.org/10.1111/jnc.14269
Letendre, S., Ellis, R., Ances, B., & McCutchan, J. (2010). Neurologic complications of HIV disease and their treatment Dr. Trop HIV Med, 18(2), 45–55. https://doi.org/10.1016/j.physbeh.2017.03.040
Menard A, Montagnac C, Solas C (2017) Neuropsychiatric adverse effects on dolutegravir: an emerging concern in Europe We. Aids 31(8):1201–1203. https://doi.org/10.1097/QAD.0000000000001457
Métral M, Darling KEA, Locatelli I, Nadin I, Santos G, Brugger P, Kovari H, Cusini A, Gutbrod K, Tarr PE, Calmy A, Lecompte TD, Assal F, Monsch A, Kunze U, Stoeckle M, Schwind M, Schmid P, Pignatti R, di Benedetto C, du Pasquier R, Cavassini M, the NAMACO study group, Swiss HIV Cohort Study, Cavassini M, du Pasquier R, Métral M, Simioni S, Brugger P, Gutbrod K, Monsch AU, Kunze U, Schneitter M, Nadin I, Früh S, Schwind M, Pignatti R, Clarke S, Assal F, Derfuss T, von Arx S, Eisele G, Sacco L, Bertschi M, Hundsberger T, du Pasquier R, Calmy A, Lecompte TD, Hauser C, Cusini A, Weber R, Kovari H, Hasse B, Tarr P, Stoeckle M, Fux C, Bernasconi E, di Benedetto C, Bruno A, Schmid P, Darling K, Cavassini M, Scherrer A, Scherrer A, Vallet Y, Alves D, Locatelli I, Decosterd L, Granziera C, Krueger G, Meuli R, Vargas M (2019) The neurocognitive assessment in the Metabolic and Aging Cohort (NAMACO) study: baseline participant profile. HIV Medicine 21:30–42. https://doi.org/10.1111/hiv.12795
Mohamed M, Barker P, Skolasky R (2018) 7T brain MRS in HIV infection: correlation with cognitive impairment and performance on neuropsychological tests. AJNR Am J Neuroradiol 39(4):704–712. https://doi.org/10.1016/j.physbeh.2017.03.040
Mollan KR, Smurzynski M, Eron JJ, Daar ES, Campbell TB, Sax PE, Gulick RM, Na L, O'Keefe L, Robertson KR, Tierney C (2014) Association between efavirenz as initial therapy for HIV-1 infection and increased risk for suicidal ideation or attempted or completed suicide: an analysis of trial data. Ann Intern Med 161(1):1–10. https://doi.org/10.7326/M14-0293
Mollan KR, Tierney C, Hellwege JN, Eron JJ, Hudgens MG, Gulick RM et al (2017) Race/ethnicity and the pharmacogenetics of reported suicidality with efavirenz among clinical trials participants. J Infect Dis 216(5):554–564. https://doi.org/10.1093/infdis/jix248
Nightingale S, Winston A, Letendre S, Michael BD, McArthur JC, Khoo S, Solomon T (2014) Controversies in HIV-associated neurocognitive disorders. The Lancet Neurology 13(11):1139–1151. https://doi.org/10.1016/S1474-4422(14)70137-1
Nooka S, Ghorpade A (2017) HIV-1-associated inflammation and antiretroviral therapy regulate astrocyte endoplasmic reticulum stress responses. Cell Death Discovery 3(1):1–11. https://doi.org/10.1038/cddiscovery.2017.61
Opii W, Sultana R, Abdul H (2007) Oxidative stress and toxicity induced by the nucleoside reverse transcriptase inhibitor (NRTI) - 2′, 3′-dideoxycytidine (ddC): relevance to HIV dementia. Exp Neurol 204(1):29–38. https://doi.org/10.1038/jid.2014.371
Ortega M, Ances B (2014) Role of HIV in amyloid metabolism. J NeuroImmune Pharmacol 9(4):483–491. https://doi.org/10.1038/jid.2014.371
Pérez-Santiago J, Schrier RD, de Oliveira MF, Gianella S, Var SR, Day TRC, Ramirez-Gaona M, Suben JD, Murrell B, Massanella M, Cherner M, Smith DM, Ellis RJ, Letendre SL, Mehta SR (2016) Cell-free mitochondrial DNA in CSF is associated with early viral rebound, inflammation, and severity of neurocognitive deficits in HIV infection. Journal of NeuroVirology 22(2):191–200. https://doi.org/10.1007/s13365-015-0384-5
Raffi F, Rachlis A, Brinson C, Arasteh K, Górgolas M, Brennan C, Pappa K, Almond S, Granier C, Nichols WG, Cuffe RL, Jr JE, Walmsley S (2015) Dolutegravir efficacy at 48 weeks in key subgroups of treatment-naive HIV-infected individuals in three randomized trials. Aids 29(2):167–174. https://doi.org/10.1097/QAD.0000000000000519
Reiber H (1995) External quality assessment in clinical neurochemistry: survey of analysis for cerebrospinal fluid (CSF) proteins based on CSF/serum quotients. Clin Chem 41(2):256–263
Rempel HC, Pulliam L (2005) HIV-1 Tat inhibits neprilysin and elevates amyloid β. Aids 19(2):127–135. https://doi.org/10.1097/00002030-200501280-00004
Robertson KR, Su Z, Margolis DM, Krambrink A, Havlir DV, Evans S, Skiest DJ (2010) Neurocognitive effects of treatment interruption in stable HIV-positive patients in an observational cohort. Neurology 74(16):1260–1266. https://doi.org/10.1212/WNL.0b013e3181d9ed09
Robertson K, Liner J, Meeker RB (2012) Antiretroviral neurotoxicity. Journal of NeuroVirology 18(5):388–399. https://doi.org/10.1007/s13365-012-0120-3
Ryom L, Lundgren JD, El-Sadr W (2018) Cardiovascular disease & use of contemporary protease inhibitors: the D:A:D Study Lene. Lancet HIV 5(6):291–300
Schouten J, Su T, Wit FW, Kootstra NA, Caan MWA, Geurtsen GJ, Schmand BA, Stolte IG, Prins M, Majoie CB, Portegies P, Reiss P (2016) Determinants of reduced cognitive performance in HIV-1-infected middle-aged men on combination antiretroviral therapy. Aids 30(7):1027–1038. https://doi.org/10.1097/QAD.0000000000001017
Schweinsburg BC, Taylor MJ, Alhassoon OM, Gonzalez R, Brown GG, Ellis RJ et al (2005) Brain mitochondrial injury in human immunodeficiency virus-seropositive (HIV+) individuals taking nucleoside reverse transcriptase inhibitors. Journal of NeuroVirology 11(4):356–364. https://doi.org/10.1080/13550280591002342
Scourfield A, Zheng J, Chinthapalli S, Waters L, Martin T, Mandalia S, Nelson M (2012) Discontinuation of Atripla as first-line therapy in HIV-1 infected individuals. Aids 26(11):1399–1401. https://doi.org/10.1097/QAD.0b013e328353b047
Sharif Y, Jumah F, Coplan L, Krosser A, Sharif K, Tubbs RS (2018) Blood brain barrier: a review of its anatomy and physiology in health and disease. Clin Anat 31(6):812–823. https://doi.org/10.1002/ca.23083
Skillbäck T, Delsing L, Synnergren J, Mattsson N, Janelidze S, Nägga K, Kilander L, Hicks R, Wimo A, Winblad B, Hansson O, Blennow K, Eriksdotter M, Zetterberg H (2017) CSF/serum albumin ratio in dementias: a cross-sectional study on 1861 patients. Neurobiol Aging 59:1–9. https://doi.org/10.1016/j.neurobiolaging.2017.06.028
Soontornniyomkij V, Umlauf A, Chung SA, Cochran ML, Soontornniyomkij B, Gouaux B, Toperoff W, Moore DJ, Masliah E, Ellis RJ, Grant I, Achim CL (2014) HIV protease inhibitor exposure predicts cerebral small vessel disease. Aids 28(9):1297–1306. https://doi.org/10.1097/QAD.0000000000000262
Soontornniyomkij V, Umlauf A, Soontornniyomkij B, Gouaux B, Ellis RJ, Levine AJ et al (2018) Association of antiretroviral therapy with brain aging changes among HIV-infected adults. Aids 32(14):2005–2015. https://doi.org/10.1016/j.physbeh.2017.03.040
Tovar-y-Romo LB, Bumpus NN, Pomerantz D, Avery LB, Sacktor N, McArthur JC, Haughey NJ (2012) Dendritic spine injury induced by the 8-hydroxy metabolite of efavirenz. J Pharmacol Exp Ther 343(3):696–703. https://doi.org/10.1124/jpet.112.195701
Trunfio M, Vai D, Montrucchio C, Alcantarini C, Livelli A, Tettoni MC, Orofino G, Audagnotto S, Imperiale D, Bonora S, di Perri G, Calcagno A (2018) Diagnostic accuracy of new and old cognitive screening tools for HIV-associated neurocognitive disorders. HIV Medicine 19(7):455–464. https://doi.org/10.1111/hiv.12622
Underwood J, Robertson KR, Winston A (2015) Could antiretroviral neurotoxicity play a role in the pathogenesis of cognitive impairment in treated HIV disease? Aids 29(3):253–261. https://doi.org/10.1097/QAD.0000000000000538
Vassallo M, Fabre R, Durant J, Lebrun-Frenay C, Joly H, Ticchioni M, DeSalvador F, Harvey-Langton A, Dunais B, Laffon M, Cottalorda J, Dellamonica P, Pradier C (2017) A decreasing CD4/CD8 ratio over time and lower CSF-penetrating antiretroviral regimens are associated with a higher risk of neurocognitive deterioration, independently of viral replication. Journal of NeuroVirology 23(2):216–225. https://doi.org/10.1007/s13365-016-0490-z
Vivithanaporn P, Asahchop EL, Acharjee S, Baker GB, Power C (2016) HIV protease inhibitors disrupt astrocytic glutamate transporter function and neurobehavioral performance. Aids 30(4):543–552. https://doi.org/10.1097/QAD.0000000000000955
Walmsley SL, Antela A, Clumeck N, Duiculescu D, Eberhard A, Gutieŕrez F et al (2013) Dolutegravir plus Abacavir-Lamivudine for the treatment of HIV-1 infection. N Engl J Med 369(19):1807–1818. https://doi.org/10.1056/NEJMoa1215541
Winston A, Amin J, Clarke A, Else L, Amara A, Owen A et al (2015) Cerebrospinal fluid exposure of efavirenz and its major metabolites when dosed at 400 mg and 600 mg once daily: a randomized controlled trial. Clin Infect Dis 60(7):1026–1032. https://doi.org/10.1093/cid/ciu976
Wyen C, Hendra H, Siccardi M, Platten M, Jaeger H, Harrer T et al (2011) Cytochrome P450 2B6 (CYP2B6) and constitutive androstane receptor (CAR) polymorphisms are associated with early discontinuation of efavirenz-containing regimens. J Antimicrob Chemother 66(9):2092–2098. https://doi.org/10.1093/jac/dkr272
Yagura H, Watanabe D, Kushida H, Tomishima K, Togami H, Hirano A, Takahashi M, Hirota K, Ikuma M, Kasai D, Nishida Y, Yoshino M, Yamazaki K, Uehira T, Shirasaka T (2017) Impact of UGT1A1 gene polymorphisms on plasma dolutegravir trough concentrations and neuropsychiatric adverse events in Japanese individuals infected with HIV-1. BMC Infect Dis 17(1):1–8. https://doi.org/10.1186/s12879-017-2717-x
Acknowledgments
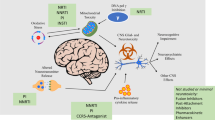
We acknowledge the illustrator Massimiliano Feroldi for his creativity and imagination in drawing the artwork “Eggs in the Brain” in Fig. 1.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
AC has received honoraria from Gilead, Janssen-Cilag, MSD, and Viiv, and he is currently receiving grants from Gilead and Viiv. SB has received honoraria from Abbvie, BMS, Gilead, Janssen-Cilag, MSD, and Viiv. The remaining authors have no conflict of interest to declare.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
De Benedetto, I., Trunfio, M., Guastamacchia, G. et al. A review of the potential mechanisms of neuronal toxicity associated with antiretroviral drugs. J. Neurovirol. 26, 642–651 (2020). https://doi.org/10.1007/s13365-020-00874-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13365-020-00874-9