Abstract
The limited tissue penetration depth and tumor hypoxic microenvironment have become the two pivotal obstacles that alleviate the antineoplastic efficacy in tumor photodynamic therapy (PDT). In the research, MnO2-decorated upconversion nanoparticles (UCSMn) have been designed to generate certain oxygen within the solid tumor, and also increase the light penetrating depth due to the optical conversion ability derived from upconversion nanoparticles. Furthermore, upconversion nanoparticles as the inner core are coated by mesoporous silica for the loading of curcumin as photosensitizer and chemotherapeutics, and then a MnO2 shell is proceeding to grow via redox method. When reaching the tumor tissue, the MnO2 nanoshells of UCSMn could be rapidly degraded into manganese ions (Mn2+) owing to the reaction with H2O2 in acidic tumor microenvironment, meanwhile producing oxygen and facilitating curcumin release. Once the tumor is illuminated by 980 nm light, the upconversion nanoparticles can transform the infrared light to visible light of 450 nm and 475.5 nm, which can be efficiently absorbed by curcumin, and then produce singlet oxygen to induce tumor cell apoptosis. Curcumin played a dual role which can not only be acted as a photosensitizer, but also a chemotherapeutic agent to further reinforce the antitumor activity. In short, the intelligent nanostructure has the potential to overcome the above-mentioned shortcomings existed in PDT and eventually do work well in the hypoxia tumors.
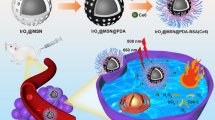
Graphical abstract
MnO2-decorated upconversion nanoparticle to solve the tissue penetration and tumor hypoxic microenvironment for tumor photodynamic therapy










Similar content being viewed by others
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
References
Agostinis P, Berg K, Cengel KA, et al. Photodynamic therapy of cancer: an update [J]. CA-Cancer J Clin. 2011;61(4):250–81.
Henderson BW, Dougherty TJ. How does photodynamic therapy work? [J]. Photochem Photobiol. 1992;55(1):145–57.
Brown SB, Brown EA, Walker I. The present and future role of photodynamic therapy in cancer treatment [J]. Lancet Oncol. 2004;5(8):497–508.
Daruwalla J, Christophi C. Hyperbaric oxygen therapy for malignancy: a review [J]. World J Surg. 2006;30(12):2112–31.
Brown JM, Wilson WR. Exploiting tumour hypoxia in cancer treatment [J]. Nat Rev Cancer. 2004;4(6):437–47.
Pan C, Ou MT, Cheng QZ, et al. Z-scheme heterojunction functionalized pyrite nanosheets for modulating tumor microenvironment and strengthening photo/chemodynamic therapeutic effects [J]. Adv Funct Mater. 2020;30(3):1906466.
Zeng WW, Zhang HJ, Deng YM, et al. Dual-response oxygen-generating mno2 nanoparticles with polydopamine modification for combined photothermal-photodynamic therapy [J]. Chem Eng J. 2020;389:124494.
Van Beek J. The Dynamic Side of the Warburg Effect: glycolytic intermediate storage as buffer for fluctuating glucose and O2 supply in tumor cells [J]. F1000 Res. 2018;7:1177.
Chen Q, Liang C, Sun X, et al. H2O2-responsive liposomal nanoprobe for photoacoustic inflammation imaging and tumor theranostics via in vivo chromogenic assay [J]. Proc Natl Acad Sci USA. 2017;114(21):5343–8.
Fan W, Bu W, Shen B, et al. Intelligent MnO2 nanosheets anchored with upconversion nanoprobes for concurrent pH-/H2O2-responsive UCL imaging and oxygen-elevated synergetic therapy [J]. Adv Mater. 2015;27(28):4155–61.
Upreti M, Jyoti A, Sethi P. Tumor microenvironment and nanotherapeutics [J]. Transl Cancer Res. 2013;2(4):309–19.
Zhang HJ, Zeng WW, Pan C, et al. SnTe@MnO2-SP nanosheet-based intelligent nanoplatform for second near-infrared light-mediated cancer theranostics [J]. Adv Funct Mater. 2019;29(37):1903791.
Ding B, Zheng P, Ma P, et al. Manganese oxide nanomaterials: synthesis, properties, and theranostic applications [J]. Adv Mater. 2020;32:190582310.
Qiu H, Tan M, Ohulchanskyy TY, et al. Recent progress in upconversion photodynamic therapy [J]. Nanomaterials. 2018;8(3445).
Hong G, Diao S, Chang J, et al. Through-skull fluorescence imaging of the brain in a new near-infrared window [J]. Nat Photonics. 2014;8(9):723–30.
Kobayashi H, Ogawa M, Alford R, et al. New strategies for fluorescent probe design in medical diagnostic imaging [J]. Chem Rev. 2010;110(5):2620–40.
Zhou Z, Song J, Nie L, et al. Reactive oxygen species generating systems meeting challenges of photodynamic cancer therapy [J]. Chem Soc Rev. 2016;45(23):6597–626.
Ai F, Ju Q, Zhang X, et al. A core-shell-shell nanoplatform upconverting near-infrared light at 808 nm for luminescence imaging and photodynamic therapy of cancer [J]. Sci Rep. 2015;5:10785.
Wilhelm S. Perspectives for upconverting nanoparticles [J]. ACS Nano. 2017;11(11):10644–53.
Park K, Lee JH. Photosensitizer effect of curcumin on uvb-irradiated HaCaT cells through activation of caspase pathways [J]. Oncol Rep. 2007;17(3):537–40.
Ailioaie LM, Ailioaie C, Litscher G. Latest innovations and nanotechnologies with curcumin as a nature-inspired photosensitizer applied in the photodynamic therapy of cancer [J]. Pharmaceutics. 2021;13(10).
Hatcher H, Planalp R, Cho J, et al. Curcumin: from ancient medicine to current clinical trials [J]. Cell Mol Life Sci. 2008;65(11):1631–52.
Wang F, Deng R, Liu X. Preparation of core-shell NaGdF4 nanoparticles doped with luminescent lanthanide ions to be used as upconversion-based probes [J]. Nat Protoc. 2014;9(7):1634–44.
Yuan J, Cen Y, Kong XJ, et al. MnO2-nanosheet-modified upconversion nanosystem for sensitive turn-on fluorescence detection of H2O2 and glucose in blood [J]. ACS Appl Mater Interfaces. 2015;7(19):10548–55.
Yang X, Xiong J, Qiu P, et al. Synthesis of a core/satellite-like multifunctional nanocarrier for pH and NIR-triggered intracellular chemothermal therapy and tumor imaging [J]. RSC Adv. 2017;7(13):7742–52.
Chatterjee S, Lee MW, Woo SH. Influence of impregnation of chitosan beads with cetyl trimethyl ammonium bromide on their structure and adsorption of Congo red from aqueous solutions [J]. Chem Eng J. 2009;155(1–2):254–9.
Selvakumar K, Kumar SMS, Thangamuthu R, et al. Physiochemical investigation of shape-designed MnO2 nanostructures and their influence on oxygen reduction reaction activity in alkaline solution [J]. J Phys Chem C. 2015;119(12):6604–18.
Liu J, Bu W, Zhang S, et al. Controlled synthesis of uniform and monodisperse upconversion core/mesoporous silica shell nanocomposites for bimodal imaging [J]. Chem Eur J. 2012;18(8):2335–41.
Chen H, Tian J, He W, et al. H2O2-activatable and O2-evolving nanoparticles for highly efficient and selective photodynamic therapy against hypoxic tumor cells [J]. J Am Chem Soc. 2015;137(4):1539–47.
Acknowledgements
We appreciated Xiaoyan Chen and Yingtao Zhong for the help in the preparation of upconversion nanoparticles.
Funding
The study was supported by the Science and Technology Program of Guangzhou, China (Grant numbers 201904010110).
Author information
Authors and Affiliations
Contributions
Yan Ma proposed the idea and participated in the design of the study. Xinru Chen collected literature search and carried out the study. Quandong Li performed the statistical analysis. Zipeng Huang provided the assistance in manuscript preparation. Wen Lin provided assistance in the manuscript editing and review. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The experimental protocol was established, according to the ethical guidelines of the Helsinki Declaration and was approved by the Guangzhou University of Chinese Medicine Institutional Animal Care and Use Committee.
Consent for publication
All the authors listed have approved the manuscript that is enclosed.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Xinru Chen and Quandong Li share first authorship of this article.
Rights and permissions
About this article
Cite this article
Chen, X., Li, Q., Huang, Z. et al. Construction and evaluation of curcumin upconversion nanocarriers decorated with MnO2 for tumor photodynamic therapy. Drug Deliv. and Transl. Res. 12, 2678–2692 (2022). https://doi.org/10.1007/s13346-022-01118-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-022-01118-5