Abstract
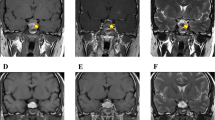
A 43-year-old man was diagnosed with acromegaly due to pituitary GH-secreting macroadenoma, and underwent craniotomy surgery. After surgery, he was given octreotide long-acting release (LAR) to treat the residual tumor. Eighteen months later, he presented polydipsia and polyuria due to diabetic ketoacidosis (DKA) and central diabetes insipidus (CDI). His casual plasma glucose level was 570 mg/dL, his HbA1c was 14.9%, and his urine was strongly positive for ketone bodies. We discuss a causal relationship among DKA, CDI, and treatment with LAR in this case with residual GH-secreting tumor from the perspective of insulin secretion and resistance.



Similar content being viewed by others
References
Colao A, Ferone D, Marzullo P, Lombardi G. Systemic complications of acromegaly: epidemiology, pathogenesis, and management. Endocr Rev. 2004;25:102–52.
Palakawong P, Arakaki R. Diabetic ketoacidosis in acromegaly: a case report. Endocr Pract. 2012;27:1–15.
Vallette S, Serri O. Octreotide LAR for the treatment of acromegaly. Expert Opin Drug Metab Toxicol. 2008;4:783–93.
Colao A, Bronstein MD, Freda P, Gu F, et al. Pasireotide versus octreotide in acromegaly: a head-to-head superiority study. J Clin Endocrinol Metab. 2014;99:791–9.
Lunetta M, Di Mauro M, Le Moli R, Nicoletti F. Effects of octreotide on glycaemic control, glucose disposal, hepatic glucose production and counterregulatory hormones secretion in type 1 and type 2 insulin treated diabetic patients. Diabetes Res Clin Pract. 1997;38:81–9.
Sato K, Takamatsu K, Hashimoto K. Short-term effects of octreotide on glucose tolerance in patients with acromegaly. Endocr J. 1995;42:739–45.
Valea A, Carsote M, Ghervan C, Georgescu C. Glycemic profile in patients with acromegaly treated with somatostatin analogue. J Med Life. 2015;8(Spec Issue):82–6.
Baldelli R, Battista C, Leonetti F, et al. Glucose homeostasis in acromegaly: effects of long-acting somatostatin analogues treatment. Clin Endocrinol (Oxf). 2003;59:492–9.
Kahara T, Ueno K, Torita M, Usuda R, Abe T. Deterioration of glycemic control during octreotide LAR treatment in an acromegalic Japanese patient with type 2 diabetes mellitus. Endocr J. 2007;54:329–33.
Shin HJ, Kim JH, Yi JH, Han SW, Kim HJ. Polyuria with the concurrent manifestation of central diabetes insipidus (CDI) & type 2 diabetes mellitus (DM). Electrolyte Blood Press. 2012;10:26–30.
Bronstein MD, Fleseriu M, Neggers S, et al. Switching patients with acromegaly from octreotide to pasireotide improves biochemical control: crossover extension to a randomized, double-blind, phase III study. Pasireotide C2305 study group. BMC Endocr Disord. 2016;16:16.
Breitschaft A, Hu K, Hermosillo Reséndiz K, Darstein C, Golor G. Management of hyperglycemia associated with pasireotide (SOM230): healthy volunteer study. Diabetes Res Clin Pract. 2014;103:458–65.
Bałdys-Waligórska A, Krzentowska-Korek A, Gołkowski F, Sokołowski G, Hubalewska-Dydejczyk A. Octreotide LAR affects the volume of pituitary adenoma in acromegalic patients. Exp Clin Endocrinol Diabetes. 2011;119:295–9.
Katznelson L, Laws ER, Melmed S, Molitch ME, Murad MH, Utz A, Wass JA. Endocrine Society. Acromegaly: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2014;99:3933–51.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure statement
There is nothing to disclose.
Human rights statement and informed consent
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964 and later versions. Informed consent or a substitute for it was obtained from all patients before they were included in the report.
About this article
Cite this article
Inaba, H., Funahashi, T., Ariyasu, H. et al. Diabetic ketoacidosis in a patient with acromegaly and central diabetes insipidus treated with octreotide long-acting release. Diabetol Int 8, 237–242 (2017). https://doi.org/10.1007/s13340-016-0301-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13340-016-0301-z