Abstract
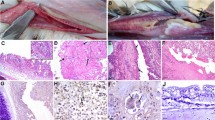
Infectious laryngotracheitis (ILT) is a viral respiratory illness in poultry that causes massive financial losses. This research aimed to isolate and identify the ILT virus in suspected outbreaks of broiler flocks in Egypt during 2020–2021, besides investigating its genetic link with other circulating strains. Real-time-PCR was used to test 57 samples taken from unvaccinated broiler farms. Ten samples are positive for ILTV, and the virus is being isolated in SPF chicken embryos. The Sanger sequencing was used to conduct (partial) sequencing of the infected cell protein4 gene (ICP4) for eight isolates. Phylogenetic analysis conducted Maximum Likelihood, comparative sequencing analysis of ICP4 of strains under study with vaccination ILT reference strains reveled that all isolates were clustered into two major groups. The (OM291843and OM291846) clustered together with the chicken embryo origin vaccine strains (IV and V group). The remaining six strains belong to the TCO vaccine(I, II and III group). The total sequence similarity between the strains under study and the various Egyptian strains varied from (97 to 100%) while the similarity with TCO or chicken embryo origin -vaccine strains ranged from (95to 100%). There were no deletions detected in the 272–283-bp region of the ICP4 gene. Detection of arginine to methionine substitutions at position 180 (R180M) and change of Serine to Asparagine at position 227 (S227N) in the (OM291843 and OM291846) which were previously described in chicken embryo origin -vaccine strains. This reveals that field strains may have evolved from vaccine strains, notably identification of non-synonymous substitutions which might be linked to the virulence strains’ attenuation. Finally, independent of geographical distribution, both chicken embryo origin-vaccine-like and TCO-Vaccine-like virus strains were circulating in Egyptian non-vaccinated broiler flocks in 2020 and 2021. Despite their genetic differences, both viruses caused significant illnesses in the field.


Similar content being viewed by others
Data Availability
The data presented in this study are available on request from the corresponding authors.
Abbreviations
- ILTV:
-
Infectious laryngotracheitis
- Real-Time-PCR:
-
Real-time-Polymeras Chain Reaction
- TCO:
-
tissue culture origin
- CEO:
-
chicken embryo origin
- S:
-
Serine
- N:
-
Asparagine
- R:
-
arginine
- M:
-
methionine
References
Epizooties OId. International animal health code: mammals, birds and bees. Office international des épizooties; 2001.
Dormitorio TV, Giambrone JJ, Macklin KS. Detection and isolation of infectious laryngotracheitis virus on a broiler farm after a disease outbreak. Avian Dis. 2013;57(4):803–7.
Aziz T. Infectious Laryngotracheitis (ILT) targets broilers. World Poult. 2010;25:17–8.
Vagnozzi A, Riblet SM, Williams SM, Zavala G, García M. Infection of broilers with two virulent strains of infectious laryngotracheitis virus: criteria for evaluation of experimental infections. Avian Dis. 2015;59(3):394–9.
Sellers HS, García M, Glisson JR, Brown TP, Sander JS, Guy JS. Mild infectious laryngotracheitis in broilers in the southeast. Avian Dis. 2004;48(2):430–6.
Ou S-C, Giambrone JJ. Infectious laryngotracheitis virus in chickens. World J Virol. 2012;1(5):142.
Gowthaman V, Kumar S, Koul M, Dave U, Murthy TGK, Munuswamy P, Tiwari R, Karthik K, Dhama K, Michalak I. Infectious laryngotracheitis: Etiology, epidemiology, pathobiology, and advances in diagnosis and control–a comprehensive review. Veterinary Q. 2020;40(1):140–61.
Fuchs W, Veits J, Helferich D, Granzow H, Teifke JP, Mettenleiter TC. Molecular biology of avian infectious laryngotracheitis virus. Vet Res. 2007;38(2):261–79.
Johnson MA, Tyack SG, Prideaux C, Kongsuwan K, Sheppard M. Nucleotide sequence of infectious laryngotracheitis virus (gallid herpesvirus 1) ICP4 gene. Virus Res. 1995;35(2):193–204.
Viejo-Borbolla A, Munoz A, Tabares E, Alcami A. Glycoprotein G from pseudorabies virus binds to chemokines with high affinity and inhibits their function. J Gen Virol. 2010;91(1):23–31.
Esparza J, Benyesh-Melnick M, Schaffer PA. Intertypic complementation and recombination between temperature-sensitive mutants of herpes simplex virus types 1 and 2. Virology. 1976;70(2):372–84.
Kozak M. Point mutations define a sequence flanking the AUG initiator codon that modulates translation by eukaryotic ribosomes. Cell. 1986;44(2):283–92.
Wang Z, Qiao Y, Chen Z, Liang Y, Cui L, Zhang Y, Li X, Xu L, Wei P, Liu S. Fos Facilitates Gallid Alpha-Herpesvirus 1 Infection by Transcriptional Control of Host Metabolic Genes and Viral Immediate Early Gene. Viruses. 2021;13(6):1110.
García M, Zavala G. Commercial vaccines and vaccination strategies against infectious Laryngotracheitis: What we have learned and knowledge gaps that remain. Avian Dis. 2019;63(2):325–34.
Oldoni I, García M. Characterization of infectious laryngotracheitis virus isolates from the US by polymerase chain reaction and restriction fragment length polymorphism of multiple genome regions. Avian Pathol. 2007;36(2):167–76.
Bagust T. Laryngotracheitis (gallid-1) herpesvirus infection in the chicken 4. latency establishment by wild and vaccine strains of ILT virus. Avian Pathol. 1986;15(3):581–95.
Bagust TJ, Johnson MA. Avian infectious laryngotracheitis: Virus-host interactions in relation to prospects for eradication. Avian Pathol. 1995;24(3):373–91.
Guy JS, Barnes HJ, Smith L. Increased virulence of modified-live infectious laryngotracheitis vaccine virus following bird-to-bird passage. Avian diseases 1991:348–355.
Groves PJ, Williamson SL, Sharpe SM, Gerber PF, Gao YK, Hirn TJ, Walkden-Brown SW. Uptake and spread of infectious laryngotracheitis vaccine virus within meat chicken flocks following drinking water vaccination. Vaccine. 2019;37(35):5035–43.
Guy JS. Laryngotracheitis. Diseases of poultry 2008:137–152.
Izuchi T, Hasegawa A. Pathogenicity of infectious laryngotracheitis virus as measured by chicken embryo inoculation. Avian diseases 1982:18–25.
Moreno A, Piccirillo A, Mondin A, Morandini E, Gavazzi L, Cordioli P. Epidemic of infectious laryngotracheitis in Italy: characterization of virus isolates by PCR–restriction fragment length polymorphism and sequence analysis. Avian Dis. 2010;54(4):1172–7.
García M, Volkening J, Riblet S, Spatz S. Genomic sequence analysis of the United States infectious laryngotracheitis vaccine strains chicken embryo origin (CEO) and tissue culture origin (TCO). Virology. 2013;440(1):64–74.
Shehata AA, Halami MY, Sultan HH, Abd El-Razik AG, Vahlenkamp TW. Chicken embryo origin-like strains are responsible for Infectious laryngotracheitis virus outbreaks in Egyptian cross-bred broiler chickens. Virus Genes. 2013;46(3):423–30.
OIE. Disease Information. World organization for animal health. In.: URL https://www.oie.int/en/animal-healthin-the-world/wahis-portal-animal-health-data/; 2021.
Tantawi H, El Batrawi A, Bastami M, Youssef Y, Fawzia M. Avian infectious laryngo-tracheitis in Egypt. I. Epidemiology, virus isolation and identification. Vet Res Commun. 1983;6(1):281–7.
Ali A, Mansour S, Mohamed M, Ali H, Shahin A. Molecular characterization of thymidine kinase and glycoprotein G genes from a possible vaccine induced infectious laryngotracheitis outbreak in Egypt. Pak Vet J. 2014;34:381–5.
NOURHAN N, MAHMOUD S, RADWAN A, HAMED R, MAHMOUD AE-M, ADEL N, EL-ZAHED HM. Isolation and molecular characterization of circulating infectious laryngotracheitis(ILT) virus in Egypt, j. Egypt vet med Assoc. 2019;79:743–59.
Bayoumi M, El-Saied M, Amer H, Bastami M, Sakr EE, El-Mahdy M. Molecular characterization and genetic diversity of the infectious laryngotracheitis virus strains circulating in Egypt during the outbreaks of 2018 and 2019. Arch Virol. 2020;165(3):661–70.
York JJ, Fahey K. Diagnosis of infectious laryngotracheitis using a monoclonal antibody ELISA. Avian Pathol. 1988;17(1):173–82.
Islam M, Khan M, Islam M, Hassan J. Isolation and characterization of infectious laryngotracheitis virus in layer chickens. Bangladesh J Veterinary Med. 2010;8(2):123–30.
Hamad Mohammed A, Sood Mustafa A, Hussein Najeeb Mohammed A-SA, Majeed MMH, Muhaidi M. Detection of Infectious Laryngotracheitis by real-time PCR in Baghdad.
Magouz A, Medhat S, Abou Asa S, Desouky A. Detection of infectious laryngotracheitis virus (Gallid herpesvirus-1) from clinically infected chickens in Egypt by different diagnostic methods. Iran J veterinary Res. 2018;19(3):194.
Ou S-C, Giambrone J, Macklin K. Detection of infectious laryngotracheitis virus from darkling beetles and their immature stage (lesser mealworms) by quantitative polymerase chain reaction and virus isolation. J Appl Poult Res. 2012;21(1):33–8.
Wolfrum N. Infectious laryngotracheitis: an update on current approaches for prevention of an old disease. J Anim Sci. 2020;98(Supplement_1):27–35.
Molini U, Aikukutu G, Khaiseb S, Kahler B, Van der Westhuizen J, Cattoli G, Dundon WG. Investigation of infectious laryngotracheitis outbreaks in Namibia in 2018. Trop Anim Health Prod. 2019;51(7):2105–8.
Hall T, Biosciences I, Carlsbad C. BioEdit: an important software for molecular biology. GERF Bull Biosci. 2011;2(1):60–1.
Burland TG. DNASTAR’s Lasergene sequence analysis software. In: Bioinformatics methods and protocols. Springer; 2000. pp. 71–91.
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S: MEGA6: molecular evolutionary genetics analysis version 6.0. Molecular biology and evolution 2013, 30(12):2725–2729.
Kumar S, Stecher G, Tamura K: MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Molecular biology and evolution 2016, 33(7):1870–1874.
Rodríguez-Avila A, Oldoni I, Riblet S, García M. Replication and transmission of live attenuated infectious laryngotracheitis virus (ILTV) vaccines. Avian Dis. 2007;51(4):905–11.
Chacón JL, Ferreira AJP. Differentiation of field isolates and vaccine strains of infectious laryngotracheitis virus by DNA sequencing. Vaccine. 2009;27(48):6731–8.
Spatz SJ, Garcia M, Riblet S, Ross TA, Volkening JD, Taylor TL, Kim T, Afonso CL. MinION sequencing to genotype US strains of infectious laryngotracheitis virus. Avian Pathol. 2019;48(3):255–69.
Barboza-Solis C, Contreras AP, Palomino-Tapia VA, Joseph T, King R, Ravi M, Peters D, Fonseca K, Gagnon CA, Van der Meer F. Genotyping of Infectious Laryngotracheitis Virus (ILTV) Isolates from Western Canadian Provinces of Alberta and British Columbia Based on Partial Open Reading Frame (ORF) a and b. Animals. 2020;10(9):1634.
García M. Current and future vaccines and vaccination strategies against infectious laryngotracheitis (ILT) respiratory disease of poultry. Vet Microbiol. 2017;206:157–62.
Guy JS, Barnes HJ, Morgan LM. Virulence of infectious laryngotracheitis viruses: comparison of modified-live vaccine viruses and North Carolina field isolates. Avian diseases 1990:106–113.
Kotiw M, Wilks C, May J. The effect of serial in vivo passage on the expression of virulence and DNA stability of an infectious laryngotracheitis virus strain of low virulence. Vet Microbiol. 1995;45(1):71–80.
de Macedo Couto R, Preis IS, Braga JFV, Brasil BS, Drummond MG, da Silva Martins NR, Ecco R. Molecular characterization of infectious laryngotracheitis virus in naturally infected egg layer chickens in a multi-age flock in Brazil. Arch Virol. 2015;160(1):241–52.
Chacon JL, Mizuma MY, Piantino Ferreira AJ. Characterization by restriction fragment length polymorphism and sequence analysis of field and vaccine strains of infectious laryngotracheitis virus involved in severe outbreaks. Avian Pathol. 2010;39(6):425–33.
Barnes M, Gary R. Bioinformatics for Geneticists. West Sussex, England. In.: John Wiley & Sons; 2003.
Menendez KR, García M, Spatz S, Tablante NL. Molecular epidemiology of infectious laryngotracheitis: a review. Avian Pathol. 2014;43(2):108–17.
Veits J, Lüschow D, Kindermann K, Werner O, Teifke JP, Mettenleiter TC, Fuchs W. Deletion of the non-essential UL0 gene of infectious laryngotracheitis (ILT) virus leads to attenuation in chickens, and UL0 mutants expressing influenza virus haemagglutinin (H7) protect against ILT and fowl plague. J Gen Virol. 2003;84(12):3343–52.
Acknowledgements
The authors extend a cordial thanks to all the farms owners, and veterinarians who help in this study.
Funding
This research received no external funding. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Author information
Authors and Affiliations
Contributions
Conceptualization, Zeinab mossad and Ali Mahmoud Zanaty methodology, Moussa,M. Saied, Mustafa M. Fathy.; software, Zeinabmossad.; validation Ali Mahmoud Zanaty.; analysis, Zeinabmossad investigation, Ali Mahmoud Zanaty and J Moussa,M. Saied resources Mustafa M. Fathy data curation, Mustafa M. Fathy, writing—original draft preparation Zeinab mossad and Ali Mahmoud Zanaty writing review and editing, Moussa,M. Saied, Mustafa M. Fathy. funding acquisition, All authors read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflict of interest.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Written informed consent has been obtained from the farm owners to publish this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
mossad, Z., Moussa, S.A., Saied, M. et al. Molecular and genetic detection of infectious laryngeotrachitis disease virus in broiler farms after a disease outbreak in Egypt. VirusDis. 33, 404–412 (2022). https://doi.org/10.1007/s13337-022-00792-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13337-022-00792-w