Abstract
Introduction
Medications used to treat type 2 diabetes (T2D) often require dose escalation to optimize effectiveness. Physician and patient perceptions of treatment characteristics of T2D medications have previously been examined, but little is known about perceptions of escalation to the optimal dose for each patient. This study examined physicians’ perceptions of dose escalation for medications used to treat T2D.
Methods
Data on dose escalation and other factors influencing decision-making for treatment of T2D were collected via an online survey of endocrinologists and primary care physicians in the USA.
Results
The sample included 501 physicians (348 primary care physicians and 153 endocrinologists). Dose escalation was not frequently considered by physicians as a primary factor keeping patients’ from reaching treatment goals (mentioned as a factor by only 7.6% of the sample) or a barrier to prescribing T2D medication (16.2%). Factors more likely to keep patients from reaching treatment goals included an unhealthy diet (86.6%) and medication adherence (77.4%). The most common reasons that physicians reported for escalating dose levels were the need for better glycemic control (reported by 89.8% of the sample), ability to decrease the total number of medications by increasing the dose of one medication (39.9%), and the need for the patient to lose weight (39.3%). Data reported by primary care physicians and endocrinologists followed similar patterns.
Conclusions
Although common with T2D treatments, escalating the dose of T2D medication was not perceived by physicians to be a significant barrier to attaining treatment goals or prescribing medication. Multiple factors contribute to the decision to escalate the dose of T2D medication.
Plain Language Summary
In early phases of initiating medication treatment for a patient with type 2 diabetes (T2D), it is common for physicians to increase from a lower initial dose to a higher end dose to maximize treatment benefit. This process is known as dose escalation. The purpose of this study was to examine physicians’ perceptions of dose escalation for medications used to treat T2D. An online survey was designed to identify reasons why physicians in the US may choose to escalate or not escalate a dose of medication for T2D. In addition, physicians were asked about factors that keep patients from reaching treatment goals to identify whether the requirement for dose escalation is perceived to be a common barrier to successful treatment. The sample included 501 physicians (348 primary care, 153 endocrinologists). Dose escalation was not frequently considered to be a primary factor keeping patients’ from reaching treatment goals or a barrier to prescribing medication for T2D. Dose escalation decisions are complex, driven by a range of factors such as glycemic control medication tolerability, the patient’s body mass index, treatment guidelines, comorbidities, characteristics of the patient’s entire treatment regimen, and potential cardiovascular benefits.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Because dose escalation is a common attribute of medications used to treat type 2 diabetes (T2D), it is important to understand its impact. |
Little is known about physicians’ perceptions of escalation to the optimal dose for each patient. |
The purpose of this study was to conduct an online survey of endocrinologists and primary care physicians in the US to examine their perceptions of dose escalation for medications used to treat T2D. |
What was learned from the study? |
Results suggest that most physicians, including endocrinologists and primary care physicians, do not perceive dose escalation to be a significant challenge. |
Dose escalation was not perceived to be a significant barrier to attaining treatment goals or prescribing medication. |
Decisions to escalate a dose are complex, driven by a range of factors such as glycemic control, medication tolerability, the patient’s body mass index, treatment guidelines, comorbidities, characteristics of the patient’s entire treatment regimen, and potential cardiovascular benefits. |
Introduction
A growing body of literature has focused on attributes of medications used to treat type 2 diabetes (T2D). Attributes examined in previous research include dose frequency, dose flexibility, glucose monitoring, adverse event profile, requirements for reconstituting the medication, and ease of preparing and using injection devices [1,2,3,4,5,6,7,8,9,10,11,12,13,14]. Attributes of the treatment process have been shown to affect medication adherence, which can have an impact on treatment outcomes [15,16,17,18,19,20,21]. In addition to the impact on patients, these medication attributes can also affect physicians’ perceptions of treatments for T2D [22], which directly influence their choice of medications to prescribe for their patients.
Relatively limited research has examined the impact of dose escalation. Dose escalation is the process of increasing a fixed dose of medication from a lower initial dose to a higher end dose to optimize the medication’s acceptability and efficacy [23]. Dose escalation is commonly required during the early phases of treatment with oral and injectable T2D therapies, such as metformin, liraglutide, tirzepatide, dulaglutide, and semaglutide [24,25,26]. Dose escalation is different from the individualized dose adjustments in response to changes in a physiological parameter, often called “dose titration.” For example, patients treated with multiple daily insulin injections regularly adjust their dose based on blood glucose levels [27].
Because dose escalation is a common attribute of medications used to treat T2D, it is important to understand its impact. In a previous study examining the patient perspective, dose escalation was perceived to be one of the least important characteristics of treatment for T2D [27]. However, physicians’ perceptions of dose escalation remain largely unknown. Therefore, the purpose of this study was to examine physicians’ perceptions of dose escalation for medications used to treat T2D. An online survey was designed to identify reasons why physicians may choose to escalate or not escalate a dose of medication for T2D. In addition, physicians were asked about factors that keep patients from reaching treatment goals to identify whether the requirement for dose escalation is perceived to be a common barrier to successful treatment.
Methods
Study Design
In this cross-sectional study, physicians completed an online survey designed to assess their perceptions of dose escalation. Before completing the survey, physicians provided electronic consent and completed online screening questions to determine whether they were eligible to participate. The online survey was designed to take approximately 20–30 min to complete, and physicians who completed the survey were reimbursed. Participants provided informed consent before completing the survey. All procedures and materials were approved by a central institutional review board (22131-01A, Ethical and Independent Review Services), which was conducted in accordance with the Declaration of Helsinki. All surveys were completed from July to September 2022.
Participants
Participants were primary care physicians and endocrinologists licensed to practice medicine in the USA. To be eligible for this study, physicians were required to have been in medical practice for ≥ 1 year, treated an average of ≥ 10 patients with T2D per month, and prescribed injectable T2D medication (i.e., insulin or glucagon-like peptide-1 receptor agonists) for ≥ 3 patients in the 6 months prior to survey completion. Physicians were excluded if they were practicing medicine in a state where the Sunshine Act prohibits participation (i.e., Vermont and Massachusetts). An estimated sample size of approximately 400 to 500 physicians (approximately 70% primary care physicians, 30% endocrinologists) was targeted, roughly equally distributed across four US regions (Northeast, Midwest, South, and West). As the planned analyses were descriptive without a key statistical comparison, no power analysis was conducted when determining the sample size target. The sample size target was determined on the basis of similar surveys published in the past as well as practical implications.
Participants were recruited through a healthcare provider (HCP) panel built over a period of approximately 12 years by sourcing HCP contact data through direct physician outreach, conferences, LinkedIn outreach, ZoomInfo, institution websites, and PubMed. HCP status is confirmed at the time of opting into the panel by verifying their National Provider Identifier and implementing other quality control metrics. Panelist data are periodically cross-checked against online databases to ensure profiling remains accurate as time passes. The recruitment strategy for most of the study was to conduct a spam-resistant continual email campaign, sending study invitations to verified primary care physicians and endocrinologists in the HCP panel. However, for the initial soft launch of 15 physicians, a controlled email campaign was used to avoid over-recruitment and pause data collection as needed to identify any potential issues with the survey prior to full launch. For the full launch, the continual email campaign was initiated, usually sending emails every 2 days during the study period. The email invitation briefly described the study and how much time it would take. A link was provided in the email invitation for the participant to click and be screened. If the screening questions determined that the participant was eligible, the participant would continue by completing the online physician survey. For this study, a total of > 30,000 email invitations were sent, and recruitment was discontinued when the sample size target was reached.
Online Physician Survey
To inform development of the survey, four clinical experts (three physicians with experience treating T2D and one clinical researcher who designs trials of medication for T2D) were interviewed about dose escalation. They were asked about factors they consider when deciding whether to escalate the dose of T2D medication, advantages and disadvantages of dose escalation, and the importance of dose escalation relative to other attributes of medication used to treat T2D. The content of the online survey was based on input provided during these qualitative interviews with physicians and a clinical researcher.
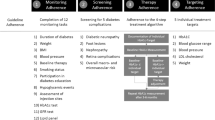
The survey began with instructions for completion. Respondents were instructed to answer the questions while thinking about “the broad range of antihyperglycemic medications for type 2 diabetes, including oral treatment like metformin, empagliflozin, and oral semaglutide; basal and meal-time insulin; and non-insulin injectable medications like liraglutide, injectable semaglutide, and dulaglutide.” Then, a series of questions were administered to determine whether the physicians met study inclusion criteria. Physicians who met criteria continued by completing three additional background questions (see physician characteristics in Table 1). The next set of questions assessed the importance of dose escalation relative to other medication attributes. These items asked physicians to select from a list of medication attributes (presented in Tables 2 and 3) to indicate which attributes most commonly prevent patients from reaching treatment goals and which attributes were most commonly perceived as a barrier to prescribing medication.
The final series of questions was designed to provide insight into physicians’ decisions regarding dose escalation. For example, one question asked physicians to select from among a list of factors (presented in Table 4) they consider when deciding whether to escalate a dose of medication for T2D. Another item asked physicians to report the most common reasons for escalating a dose over the past 6 months, again by selecting from a list of potential reasons (presented in Table 5).
After each of the questions where physicians selected multiple responses from a list of options (i.e., Tables 2, 3, 4, 5), physicians were asked to rank their selections in order of importance. The exact wording of the key questions is presented in footnotes below the relevant tables.
After completing the draft survey, it was formatted for online completion and administered to the first 15 participants (i.e., 10 primary care physicians and 5 endocrinologists). Data collection was paused after these initial 15 participants so that results could be examined to ensure that the survey was functioning as intended. Results of the interim analysis led to two edits prior to continuing with data collection. First, the item assessing gender was moved to the screening section earlier in the survey so that gender could be considered as part of the screening criteria. Second, one question was deleted because it appeared to be potentially confusing. After making these two edits, the full data collection was allowed to proceed until the sample size target was met. Because no survey content was changed or added following the initial 15 completions, results from the pilot participants were included in the final dataset.
Statistical Methods
Descriptive statistics were used to summarize participants’ responses and to characterize the sample in terms of sociodemographic characteristics and clinical background. For continuous variables, the mean, median, standard deviation, and range were calculated. Categorical variables are reported as frequencies and percentages. Analyses were conducted with SAS software version 9.4 (SAS Institute, Cary, NC).
Results
Sample Characteristics
Of the 602 physicians who were screened, 501 (83.2%) met criteria for study inclusion, including 348 (57.8%) primary care physicians and 153 (25.4%) endocrinologists (Fig. 1). The most common reasons for ineligibility were not being a primary care physician or endocrinologist (n = 47, 46.5%) and not seeing a sufficient number of patients with T2D per month (n = 13, 12.9%). The majority of physicians were male (n = 320, 63.9% of those who completed the survey), and most worked in either an individual or small group practice (n = 208, 41.5%) or a multi-specialty group practice (n = 217, 43.3%). Participants were roughly evenly distributed across the Northeast (n = 130, 25.9%), Midwest (n = 117, 23.4%), South (n = 139, 27.7%), and West (n = 115, 23.0%) regions of the US (Table 1).
Factors Preventing Attainment of T2D Treatment Goals
Unhealthy diet (86.6%), medication adherence problems (77.4%), and lack of exercise (71.9%) were the factors most commonly cited by physicians as preventing patients from reaching T2D treatment goals (Table 2). In contrast, few physicians (7.6%) selected “difficulty escalating to an effective dose” as a factor that prevented patients from reaching treatment goals. Of the 38 physicians (7.6%) who ranked “difficulty escalating to an effective dose” as one of the five most important factors preventing patients from reaching T2D treatment goals, only one (2.6%) ranked it as the most important factor. Reports of the primary care physicians and endocrinologists followed similar patterns.
Barriers to Prescribing T2D Medication
Over two-thirds of physicians (68.7%) reported that health plan formulary restrictions were a barrier to prescribing T2D medications (Table 3). Other commonly reported barriers included an injectable route of administration (45.9%), concerns about the risk of medication-related adverse events (39.9%), patient preference (37.1%), and the tendency of some T2D medications to cause weight gain (32.1%). In comparison, “challenges associated with escalating to an effective dose” was reported to be a barrier to prescription less frequently (16.2%). When physicians were asked to rank the factors they consider to be barriers to prescription, health plan formulary restrictions were most commonly cited (by 67.4% of the sample) as the most important factor. Dose escalation was reported to be the primary barrier by 9.9% of the sample.
Factors Contributing to Decisions About Dose Escalation
To identify factors that physicians consider when deciding whether to escalate a dose of medication for T2D, they were asked to select the five most important factors from the list of 18 factors presented in Table 4. “Glycemic control and HbA1c” were selected as one of the five most important factors by 446 physicians (89.0% of the sample). Of these 446, 74.9% reported that this was the most important factor contributing to dose escalation decisions. “Medication tolerability and adverse events” appears to be a second primary factor, selected by 60.5% of the sample. While no other factors were selected by a majority of the sample, the other common factors included severity of the patient’s diabetes (42.3%), the patient’s current weight or body mass index (BMI) (38.3%), treatment guidelines (37.3%), and comorbidities (34.5%). Responses to this question followed similar patterns for the primary care physicians and endocrinologists (Table 4).
Respondents were also asked about the most common reasons they have escalated a dose for patients during the past 6 months (Table 5). Consistent with results in Table 4, they reported most commonly escalating a dose because their patients required more glycemic control (89.8%). Other reasons that were somewhat commonly cited included the ability to decrease total medication burden by increasing the dose of one medication (39.9%), the need for more weight loss (39.3%), and the fact that there were no tolerability concerns at a lower dose, which made it possible to increase the dose (38.7%). Physicians were also asked about the factors that cause them not to escalate a dose for one of their patients. The most commonly cited reasons were the potential for adverse events at a higher dose (68.5%), additional glycemic control was not needed (66.3%), dosing limitations associated with comorbidities such as chronic kidney disease (49.7%), and the patient’s age (25.0%). Reasons for escalating and not escalating a dose followed similar patterns for the primary care physicians and endocrinologists.
Discussion
Previous research has examined patient and physician perceptions of a wide range of attributes of medications for treatment of T2D [9, 14]. For example, medication attributes shown to have an impact on patient preference include glycemic control, adverse event profile, dose frequency, injection device, dose flexibility, and medication preparation requirements [14]. However, the current study is the first to focus on dose escalation for the treatment of T2D. Results of the current survey suggest that endocrinologists and primary care physicians may not perceive dose escalation to be a significant challenge. These findings add to a previous study indicating that patients perceived dose escalation to be one of the least important characteristics of treatments for T2D [27].
Only 16.2% of physicians in this sample reported that dose escalation was a potential barrier to prescribing medication for their patients with T2D. Furthermore, only 7.6% considered dose escalation requirements to be one of the top five factors preventing patients from reaching treatment goals. The factors that were most commonly believed to interfere with treatment goals tended to be related to patient behavior, such as unhealthy diet, medication adherence, lack of exercise, and missing healthcare visits. In addition, a range of treatment process attributes were perceived to interfere with treatment goals more commonly than dose escalation (e.g., the need to titrate doses based on glucose levels, dose frequency, dose timing requirements). In general, these results were similar across the subgroups of endocrinologists and primary care physicians. Overall, these findings help place dose escalation in context of other T2D medication attributes. From the physician perspective, requirements for dose escalation appear to be a less important concern than the effects of the medication as well as other treatment process attributes.
Results also provide insight into physicians’ decision-making regarding dose escalation. Responses to several questions in this survey indicate that decisions to escalate a dose are complex, driven by a range of factors. The need for increased glycemic control was the most commonly reported reason that physicians escalate a dose, but they reported considering multiple factors when making these decisions, including medication tolerability, the patient’s current weight or BMI, treatment guidelines, comorbidities, characteristics of the patient’s entire treatment regimen, and potential cardiovascular benefits.
Findings should be interpreted in the context of several limitations. First, the survey was conducted with a sample of physicians who treat T2D; therefore, results cannot provide insight into the patient perspective. It is possible that patients could have different views of dose escalation requirements, and future research is needed to examine the patient perspective.
Second, because this survey was designed to be administered efficiently to a large number of physicians, structured responses were provided for each question. This structured quantitative approach does not allow for the rich detailed responses that can be gathered in qualitative interviews or questions with free-text response fields. It is possible that a less structured approach could identify additional factors contributing to dose escalation decisions. For example, frequency of interaction with their HCPs as well as quality of shared decision-making with these HCPs [28, 29] could be additional factors that have an impact on ultimate treatment goals. In addition, the survey was designed to be brief, which means that some potentially relevant questions were not addressed. For example, the number of participants who recently performed a dose escalation is not known.
Finally, there may be limitations associated with the representativeness and generalizability of this sample of physicians in the US. Although recruitment targets ensured a geographically diverse sample from across the US, the HCP panel recruitment approach does not necessarily ensure that the sample is nationally representative. Furthermore, physicians working in other countries with different healthcare systems could have different priorities related to dose escalation. Therefore, generalizability of the current results to other countries is unknown, and future research should examine issues associated with dose escalation outside the US.
Conclusions
This study adds to previous research on physicians’ perceptions of attributes of treatments for T2D. Results suggest that physicians consider multiple factors when deciding whether to escalate a dose for their patients, including effectiveness, tolerability, and a range of patient characteristics. Overall, responses to this survey suggest that physicians who treat T2D do not perceive the requirement for dose escalation to be a significant barrier to achieving treatment goals or prescribing a specific medication.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Boye KS, Matza LS, Stewart KD, Jordan J, et al. Patient preferences and health state utilities associated with dulaglutide and semaglutide injection devices among patients with type 2 diabetes in italy. J Med Econ. 2019;22(8):806–13.
Gelhorn HL, Bacci ED, Poon JL, Boye KS, et al. Evaluating preferences for profiles of glucagon-like peptide-1 receptor agonists among injection-naive type 2 diabetes patients in Japan. Patient Prefer Adherence. 2016;10:1337–48.
Gelhorn HL, Osumili B, Brown K, Ross MM, et al. The impact of substantial improvements in HbA1c and weight loss on the medication preferences of people with type 2 diabetes. Patient Prefer Adherence. 2023;17:793–805.
Gelhorn HL, Poon JL, Davies EW, Paczkowski R, et al. Evaluating preferences for profiles of GLP-1 receptor agonists among injection-naive type 2 diabetes patients in the UK. Patient Prefer Adherence. 2015;9:1611–22.
Matza LS, Boye KS, Currie BM, Paczkowski R, et al. Patient perceptions of injection devices used with dulaglutide and liraglutide for treatment of type 2 diabetes. Curr Med Res Opin. 2018;34(8):1457–64.
Matza LS, Boye KS, Jordan JB, Norrbacka K, et al. Patient preferences in Italy: Health state utilities associated with attributes of weekly injection devices for treatment of type 2 diabetes. Patient Prefer Adherence. 2018;12:971–9.
Matza LS, Boye KS, Stewart KD, Coyne KS, et al. Assessing patient preference between the dulaglutide pen and the semaglutide pen: a crossover study (PREFER). Diabetes Obes Metab. 2020;22(3):355–64.
Matza LS, Boye KS, Stewart KD, Davies EW, Paczkowski R. Health state utilities associated with attributes of weekly injection devices for treatment of type 2 diabetes. BMC Health Serv Res. 2017;17(1):774.
Matza LS, Curtis SE, Jordan JB, Adetunji O, et al. Physician perceptions of GLP-1 receptor agonists in the UK. Curr Med Res Opin. 2016;32(5):857–64.
Matza LS, Osumili B, Stewart KD, Perez-Nieves M, et al. Patient preferences and health state utilities associated with mealtime insulin concentrations among patients with diabetes in Italy. Diabetes Ther. 2020;11(1):319–30.
Polster M, Zanutto E, McDonald S, Conner C, Hammer M. A comparison of preferences for two GLP-1 products–liraglutide and exenatide–for the treatment of type 2 diabetes. J Med Econ. 2010;13(4):655–61.
Savarese G, Sharma A, Pang C, Wood R, Soleymanlou N. Patient preferences for newer oral therapies in type 2 diabetes. Int J Cardiol. 2023;371:526–32.
Seo J, Heidenreich S, Aldalooj E, Poon JL, et al. Patients’ preferences for connected insulin pens: a discrete choice experiment among patients with type 1 and type 2 diabetes. Patient. 2023;16(2):127–38.
Thieu VT, Robinson S, Kennedy-Martin T, Boye KS, Garcia-Perez LE. Patient preferences for glucagon-like peptide 1 receptor-agonist treatment attributes. Patient Prefer Adherence. 2019;13:561–76.
Hixson-Wallace JA, Dotson JB, Blakey SA. Effect of regimen complexity on patient satisfaction and compliance with warfarin therapy. Clin Appl Thromb Hemost. 2001;7(1):33–7.
Morris LS, Schulz RM. Medication compliance: the patient’s perspective. Clin Ther. 1993;15(3):593–606.
Raue PJ, Schulberg HC, Heo M, Klimstra S, Bruce ML. Patients’ depression treatment preferences and initiation, adherence, and outcome: a randomized primary care study. Psychiatr Serv. 2009;60(3):337–43.
Schaller M, Sigurgeirsson B, Sarkany M. Patient-reported outcomes from two randomised studies comparing once-weekly application of amorolfine 5% nail lacquer to other methods of topical treatment in distal and lateral subungual onychomycosis. Mycoses. 2017;60(12):800–7.
Shikiar R, Rentz A, Barone J, Duncanson F, Katz E. Patient satisfaction with ofloxacin (f) and polymyxin b/neomycin/hydrocortisone (c) in the treatment of otitis externa: results from two randomized clinical trials. J Manag Care Med. 2002;6(3):24–7.
Shikiar R, Rentz AM. Satisfaction with medication: an overview of conceptual, methodologic, and regulatory issues. Value Health. 2004;7(2):204–15.
Shingler SL, Bennett BM, Cramer JA, Towse A, et al. Treatment preference, adherence and outcomes in patients with cancer: literature review and development of a theoretical model. Curr Med Res Opin. 2014;30(11):2329–41.
Toroski M, Kebriaeezadeh A, Esteghamati A, Karyani AK, et al. Patient and physician preferences for type 2 diabetes medications: a systematic review. J Diabetes Metab Disord. 2019;18(2):643–56.
Frias JP, Nauck MA, Van J, Benson C, et al. Efficacy and tolerability of tirzepatide, a dual glucose-dependent insulinotropic peptide and glucagon-like peptide-1 receptor agonist in patients with type 2 diabetes: a 12-week, randomized, double-blind, placebo-controlled study to evaluate different dose-escalation regimens. Diabetes Obes Metab. 2020;22(6):938–46.
Almandoz JP, Lingvay I, Morales J, Campos C. Switching between glucagon-like peptide-1 receptor agonists: rationale and practical guidance. Clin Diabetes. 2020;38(4):390–402.
Gallwitz B, Giorgino F. Clinical perspectives on the use of subcutaneous and oral formulations of semaglutide. Front Endocrinol (Lausanne). 2021;12: 645507.
Gorgojo-Martinez JJ, Mezquita-Raya P, Carretero-Gomez J, Castro A, et al. Clinical recommendations to manage gastrointestinal adverse events in patients treated with GLP-1 receptor agonists: a multidisciplinary expert consensus. J Clin Med. 2022. https://doi.org/10.3390/jcm12010145.
Boye KS, Jordan JB, Malik RE, Currie BM, Matza LS. Patient perceptions of and preferences between characteristics of injectable diabetes treatments. Diabetes Ther. 2021;12(9):2387–403.
Rodriguez-Gutierrez R, Gionfriddo MR, Ospina NS, Maraka S, et al. Shared decision making in endocrinology: present and future directions. Lancet Diabetes Endocrinol. 2016;4(8):706–16.
Ruissen MM, Montori VM, Hargraves IG, Branda ME, et al. Problem-based shared decision-making in diabetes care: a secondary analysis of video-recorded encounters. BMJ Evid Based Med. 2023;28(3):157–63.
Acknowledgements
The authors thank the physician participants for taking the time to complete this online survey.
Medical Writing and Editorial Assistance.
The authors thank MedPanel, Inc. (Waltham, MA), for sample recruitment and collection of electronic data; Robyn Cyr for statistical programming; John Plant for medical writing input; Resmi Thekkedath for assistance with the IRB submission; Amara Tiebout for editorial review and support. Robyn Cyr, John Plant, Resmi Thekkedath, and Amara Tiebout are employed by Evidera, a company that received funding from Eli Lilly and Company for time spent on this research. The survey was developed by the authors. Therefore, no external permissions were required to use or implement the survey.
Funding
Funding for this study and the Rapid Service Fee was provided by Eli Lilly and Company.
Author information
Authors and Affiliations
Contributions
All authors collaborated on the study design, analysis plan, interpretation of data, and outline of the manuscript. Louis S. Matza drafted the manuscript, and Kristina S. Boye, Raleigh Malik, and Jessica B. Jordan provided input and approval.
Corresponding author
Ethics declarations
Conflict of Interest
Louis S. Matza and Jessica B. Jordan are employees of Evidera, a company that received funding from Eli Lilly and Company for time spent conducting this research. Kristina S. Boye and Raleigh Malik are employees of and own stock in Eli Lilly and Company.
Ethical Approval
Participants provided informed consent before completing the survey. All procedures and materials were approved by a central institutional review board (22131-01A, Ethical and Independent Review Services), which was conducted in accordance with the Declaration of Helsinki. All surveys were completed from July to September 2022.
Additional information
Prior Presentation: Parts of these study results were presented in a poster format at the American Diabetes Association 83rd Annual Scientific Sessions in San Diego, CA from 23–26 June 2023. The citation for this work is as follows: Boye K, Jordan J, Malik R, Cyr RA, Matza LS. 1054-P: A Physician Survey Focusing on Dose Escalation for Type 2 Diabetes Medications. Diabetes. 2023;72(Supplement1) https://doi.org/10.2337/db23-1054-P.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Boye, K.S., Jordan, J.B., Malik, R. et al. Physician Perceptions of Dose Escalation for Type 2 Diabetes Medications in the United States. Diabetes Ther 15, 381–393 (2024). https://doi.org/10.1007/s13300-023-01499-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-023-01499-x