Abstract
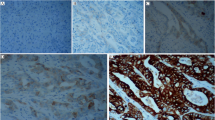
Hepatoma-derived growth factor (HDGF) is an acidic heparin-binding protein involved in tumor progression and poor prognosis of kinds of cancers. Aimed at investigating the functions of HDGF in intrahepatic cholangiocarcinoma (IHCC), we detected the expression of HDGF by immunohistochemistry in 83 patients. Associations of HDGF with clinicopathologic features, microvascular density (MVD), and overall survival rates were further analyzed by Chi-square method, univariate or multivariate analysis. HDGF functions in IHCC proliferation, invasion, and angiogenesis were detected by MTT, transwell, and tube formation assays, respectively. As a result, we found that HDGF-positive expression rate in IHCC was 51.8 % (43/83) in IHCC. HDGF expression was significantly correlated to MVD (P = 0.031), lymphatic invasion (P = 0.030), distant metastasis (P = 0.002), and TNM stage (P = 0.037). HDGF was further identified as an independent prognostic factor in IHCC with Kaplan-Meier method (P = 0.003) and Cox-regression model (P = 0.008). Moreover, both intracellular and extracellular HDGF were proved to promote the proliferation, invasion, and angiogenesis of IHCC cell lines. In conclusion, HDGF was identified as an independent prognostic biomarker in IHCC. HDGF can promote IHCC cells progression, including proliferation, invasion, and angiogenesis, indicating HDGF could become a new promising and potential drug target of IHCC.





Similar content being viewed by others
References
Nakamura H, Izumoto Y, Kambe H, Kuroda T, Mori T, Kawamura K, et al. Molecular cloning of complementary DNA for a novel human hepatoma-derived growth factor. Its homology with high mobility group-1 protein. J Biol Chem. 1994;269:25143–9.
Enomoto H, Yoshida K, Kishima Y, Kinoshita T, Yamamoto M, Everett AD, et al. Hepatoma-derived growth factor is highly expressed in developing liver and promotes fetal hepatocyte proliferation. Hepatology. 2002;36:1519–27.
Chang KC, Tai MH, Lin JW, Wang CC, Huang CC, Hung CH, et al. Hepatoma-derived growth factor is a novel prognostic factor for gastrointestinal stromal tumors. Int J Cancer J Int Cancer. 2007;121:1059–65.
Uyama H, Tomita Y, Nakamura H, Nakamori S, Zhang B, Hoshida Y, et al. Hepatoma-derived growth factor is a novel prognostic factor for patients with pancreatic cancer. Clin Cancer Res: Off J Am Assoc Cancer Res. 2006;12:6043–8.
Yamamoto S, Tomita Y, Hoshida Y, Takiguchi S, Fujiwara Y, Yasuda T, et al. Expression of hepatoma-derived growth factor is correlated with lymph node metastasis and prognosis of gastric carcinoma. Clin Cancer Res: An Off J Am Assoc Cancer Res. 2006;12:117–22.
Hu TH, Huang CC, Liu LF, Lin PR, Liu SY, Chang HW, et al. Expression of hepatoma-derived growth factor in hepatocellular carcinoma. Cancer. 2003;98:1444–56.
Ren H, Tang X, Lee JJ, Feng L, Everett AD, Hong WK, et al. Expression of hepatoma-derived growth factor is a strong prognostic predictor for patients with early-stage non-small-cell lung cancer. J Clin Oncol: Off J Am Soc Clin Oncol. 2004;22:3230–7.
Abouzied MM, El-Tahir HM, Prenner L, Haberlein H, Gieselmann V, Franken S. Hepatoma-derived growth factor. Significance of amino acid residues 81–100 in cell surface interaction and proliferative activity. J Biol Chem. 2005;280:10945–54.
Okuda Y, Nakamura H, Yoshida K, Enomoto H, Uyama H, Hirotani T, et al. Hepatoma-derived growth factor induces tumorigenesis in vivo through both direct angiogenic activity and induction of vascular endothelial growth factor. Cancer Sci. 2003;94:1034–41.
Everett AD, Narron JV, Stoops T, Nakamura H, Tucker A. Hepatoma-derived growth factor is a pulmonary endothelial cell-expressed angiogenic factor. Am J Physiol Lung Cell Mol Physiol. 2004;286:L1194–201.
Kishima Y, Yamamoto H, Izumoto Y, Yoshida K, Enomoto H, Yamamoto M, et al. Hepatoma-derived growth factor stimulates cell growth after translocation to the nucleus by nuclear localization signals. J Biol Chem. 2002;277:10315–22.
Palmer WC, Patel T. Are common factors involved in the pathogenesis of primary liver cancers? A meta-analysis of risk factors for intrahepatic cholangiocarcinoma. J Hepatol. 2012;57:69–76.
Sempoux C, Jibara G, Ward SC, Fan C, Qin L, Roayaie S, et al. Intrahepatic cholangiocarcinoma: new insights in pathology. Sem Liver Dis. 2011;31:49–60.
Patel T. Increasing incidence and mortality of primary intrahepatic cholangiocarcinoma in the United States. Hepatology. 2001;33:1353–7.
Patel T. Cholangiocarcinoma—controversies and challenges. Nat Rev Gastroenterol Hepatol. 2011;8:189–200.
Xu YF, Yang XQ, Lu XF, Guo S, Liu Y, Iqbal M, et al. Fibroblast growth factor receptor 4 promotes progression and correlates to poor prognosis in cholangiocarcinoma. Biochem Biophys Res Commun. 2014;446:54–60.
Rimm DL, Camp RL, Charette LA, Costa J, Olsen DA, Reiss M. Tissue microarray: a new technology for amplification of tissue resources. Cancer J. 2001;7:24–31.
Liu YF, Zhao R, Guo S, Wang XQ, Lian PL, Chen YG, et al. Expression and clinical significance of hepatoma-derived growth factor as a prognostic factor in human hilar cholangiocarcinoma. Ann Surg Oncol. 2011;18:872–9.
Gasparini G, Weidner N, Bevilacqua P, Maluta S, Dalla Palma P, Caffo O, et al. Tumor microvessel density, p53 expression, tumor size, and peritumoral lymphatic vessel invasion are relevant prognostic markers in node-negative breast carcinoma. J Clin Oncol: Off J Am Soc Clin Oncol. 1994;12:454–66.
Erbersdobler A, Isbarn H, Dix K, Steiner I, Schlomm T, Mirlacher M, et al. Prognostic value of microvessel density in prostate cancer: a tissue microarray study. World J Urol. 2010;28:687–92.
Thelen A, Scholz A, Weichert W, Wiedenmann B, Neuhaus P, Gessner R, et al. Tumor-associated angiogenesis and lymphangiogenesis correlate with progression of intrahepatic cholangiocarcinoma. Am J Gastroenterol. 2010;105:1123–32.
Guo Z, He Y, Wang S, Zhang A, Zhao P, Gao C, et al. Various effects of hepatoma-derived growth factor on cell growth, migration and invasion of breast cancer and prostate cancer cells. Oncol Rep. 2011;26:511–7.
Kolokythas A, Cox DP, Dekker N, Schmidt BL. Nerve growth factor and tyrosine kinase a receptor in oral squamous cell carcinoma: is there an association with perineural invasion? J Oral Maxillofac Surg: Off J Am Assoc Oral Maxillofac Surg. 2010;68:1290–5.
Yoshikawa D, Ojima H, Iwasaki M, Hiraoka N, Kosuge T, Kasai S, et al. Clinicopathological and prognostic significance of EGFR, VEGF, and HER2 expression in cholangiocarcinoma. Br J Cancer. 2008;98:418–25.
Yang Y, Li H, Zhang F, Shi H, Zhen T, Dai S, et al. Clinical and biological significance of hepatoma-derived growth factor in Ewing’s sarcoma. J Pathol. 2013;231:323–34.
Chen SC, Kung ML, Hu TH, Chen HY, Wu JC, Kuo HM, et al. Hepatoma-derived growth factor regulates breast cancer cell invasion by modulating epithelial–mesenchymal transition. J Pathol. 2012;228:158–69.
Everett AD, Lobe DR, Matsumura ME, Nakamura H, McNamara CA. Hepatoma-derived growth factor stimulates smooth muscle cell growth and is expressed in vascular development. J Clin Investig. 2000;105:567–75.
Everett AD, Bushweller J. Hepatoma derived growth factor is a nuclear targeted mitogen. Curr Drug Targets. 2003;4:367–71.
Everett AD, Stoops T, McNamara CA. Nuclear targeting is required for hepatoma-derived growth factor-stimulated mitogenesis in vascular smooth muscle cells. J Biol Chem. 2001;276:37564–8.
Ooi BN, Mukhopadhyay A, Masilamani J, Do DV, Lim CP, Cao XM, et al. Hepatoma-derived growth factor and its role in keloid pathogenesis. J Cell Mol Med. 2010;14:1328–37.
Tsang TY, Tang WY, Tsang WP, Co NN, Kong SK, Kwok TT. Mechanistic study on growth suppression and apoptosis induction by targeting hepatoma-derived growth factor in human hepatocellular carcinoma HepG2 cells. Cell Physiol Biochem: Int J Exp Cell Physiol Biochem Pharmacol. 2009;24:253–62.
Tsai HE, Liu GS, Kung ML, Liu LF, Wu JC, Tang CH, et al. Downregulation of hepatoma-derived growth factor contributes to retarded lung metastasis via inhibition of epithelial-mesenchymal transition by systemic POMC gene delivery in melanoma. Mol Cancer Ther. 2013;12:1016–25.
Zhang J, Ren H, Yuan P, Lang W, Zhang L, Mao L. Down-regulation of hepatoma-derived growth factor inhibits anchorage-independent growth and invasion of non-small cell lung cancer cells. Cancer Res. 2006;66:18–23.
Ren H, Chu Z, Mao L. Antibodies targeting hepatoma-derived growth factor as a novel strategy in treating lung cancer. Mol Cancer Ther. 2009;8:1106–12.
Thirant C, Galan-Moya EM, Dubois LG, Pinte S, Chafey P, Broussard C, et al. Differential proteomic analysis of human glioblastoma and neural stem cells reveals hdgf as a novel angiogenic secreted factor. Stem Cells. 2012;30:845–53.
Hsu SS, Chen CH, Liu GS, Tai MH, Wang JS, Wu JC, et al. Tumorigenesis and prognostic role of hepatoma-derived growth factor in human gliomas. J Neuro-Oncol. 2012;107:101–9.
von Heijne G. A new method for predicting signal sequence cleavage sites. Nucleic Acids Res. 1986;14:4683–90.
Nakamura H, Kambe H, Egawa T, Kimura Y, Ito H, Hayashi E, et al. Partial purification and characterization of human hepatoma-derived growth factor. Clin Chim Acta Int J Clin Chem. 1989;183:273–84.
Acknowledgment
This study is funded by the Outstanding Young Scientist Award Fund supported by Shandong Province (no. BS2012YY021).
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guo, S., Liu, Hd., Liu, Yf. et al. Hepatoma-derived growth factor: a novel prognostic biomarker in intrahepatic cholangiocarcinoma. Tumor Biol. 36, 353–364 (2015). https://doi.org/10.1007/s13277-014-2651-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-014-2651-0