Abstract
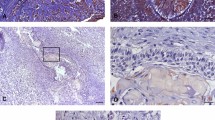
Ameloblastoma is a locally invasive odontogenic tumor with a high recurrence rate. Its local invasiveness is aided by angiogenesis, which can be correctly estimated by CD34. On the other hand, maspin decreases the local invasive and metastatic capability of cancer cells and functions as an angiogenesis inhibitor. We aim to assess the association between maspin expression and microvessel density in ameloblastoma. Twenty-five formalin-fixed paraffin-embedded (FFPE) blocks of ameloblastoma cases were prepared for antibody processing to CD34 and maspin. Positive immunohistochemical staining was marked by brown cytoplasmic/membrane coloration for CD34 and by nuclear/cytoplasmic coloration for maspin. At the ×40 magnification, we counted blood vessels in two areas of dimension; 300 × 400 μm (area A) and 150 × 200 μm (area B) adjacent to the tumor region to assess relative dispersion of the vessels bordering the tumor. The overall approximate microvessel density (MVD) for area A = 11 (minimum 2, maximum 21) and that for area B = 5 (minimum 1, maximum 10). The MVD in the area A of plexiform ameloblastoma was similar to that of the unicystic, while the hemangiomatous variant had the highest MVD for area A. Maspin positivity was present only in the cytoplasm of ameloblast, stellate reticulum, and the fibrous connective tissue in varying proportions. There was no evidence of the anti-angiogenesis effect of maspin in ameloblastoma from this study. The significance of cytoplasmic localization of maspin in the ameloblasts and stellate reticulum cells needs further investigation.


Similar content being viewed by others
References
Jordan RCK, Speight PM. Current concepts of odontogenic tumours. Diagn Histopathol. 2009;15(6):303–10.
Folkman J. Tumor angiogenesis: therapeutic implications. N Engl J Med. 1971;285:1182–6.
Kumamoto H, Ohki K, Ooya K. Association between vascular endothelial growth factor (VEGF) expression and tumor angiogenesis in ameloblastomas. J Oral Pathol Med. 2002;31:28–34.
Nagatsuka H, Hibi K, Gunduz M, Tsujigiwa H, Tamamura R, Sugahara T, et al. Various immunostaining patterns of CD31, CD34 and endoglin and their relationship with lymph node metastasis in oral squamous cell carcinomas. J Oral Pathol Med. 2005;34:70–6.
Lanza F, Healy L, Sutherland DR. Structural and functional features of the CD34 antigen: an update. J Biol Regul Homeost Agents. 2001;15:1–13.
Seifi S, Shafaie S, Ghadiri S. Microvessel density in follicular cysts, keratocystic odontogenic tumours and ameloblastomas. Asian Pacific J Cancer Prev. 2011;12:351–6.
Sager R, Sheng S, Pemberton P, Hendrix MJ. Maspin. A tumor suppressing serpin. Adv Exp Med Biol. 1997;425:77–88.
Lockett J, Yin S, Li X, Meng Y, Sheng S. Tumor suppressive maspin and epithelial homeostasis. J Cell Biochem. 2006;97(4):651–60.
Goulet B, Chan G, Chambers AF, Lewis JD. An emerging role for the nuclear localization of maspin in the suppression of tumor progression and metastasis. Biochem Cell Biol. 2012;90(1):22–38.
Ghanaati S, Barbeck M, Lorenz J, Stuebinger S, Seitz O, Landes C, et al. Synthetic bone substitute material comparable with xenogeneic material for bone tissue regeneration in oral cancer patients: first and preliminary histological, histomorphometrical and clinical results. Ann Maxillofac Surg. 2013;3(2):126–38.
Dunstan S, Powe DG, Wilkinson M, et al. The stroma of oral squamous cell carcinomas show increased vascularity compared with adjacent host tissue. Br J Cancer. 1997;75:559–65.
Guzmán-Medrano R, Arreola-Rosales RL, Shibayama M, Silva-Olivares DA, Bologna-Molina R, Rodríguez MA. Tumor-associated macrophages and angiogenesis: a statistical correlation that could reflect a critical relationship in ameloblastoma. Pathol Res Pract. 2012;208(11):672–6.
Sherlin HJ, Natesan A, Ram P, Ramani P, Thiruvenkadam C. Immunohistochemical profiling of ameloblastoma using cytokeratin, vimentin, smooth muscle actin, CD34 and S-100. Ann Maxillofac Surg. 2013;3(1):51–7.
Nouaem MI. Association between vascular endothelial growth factor (VEGF) expression and tumor angiogenesis in benign and malignant ameloblastoma. Egypt Dent J. 2005;51(17):186.
Waldron CA, El-Mofty SK. A histopathologic study of 116 ameloblastomas with special reference to the desmoplastic variant. Oral Surg Oral Med Oral Pathol. 1987;63(4):441–51.
Adebiyi KE, Ugboko VI, Omoniyi-Esan GO, Ndukwe KC, Oginni FO. Clinicopathological analysis of histological variants of ameloblastoma in a suburban Nigerian population. Head Face Med. 2006;2:42.
Tete S, Mastrangelo F, Grimaldi S, et al. Immunohistochemical evaluation of CD31 in human cystic radicular lesions and in keratocyst. Int J Immunopathol Pharmacol. 2005;18:39–45.
Bailey CM, Khalkhali-Ellis Z, Seftor EA, Hendrix MJ. Biological functions of maspin. J Cell Physiol. 2006;209:617–24.
Sheng S, Carey J, Seftor EA, Dias L, Hendrix MJ, Sager R. Maspin acts at the cell membrane to inhibit invasion and motility of mammary and prostatic cancer cells. Proc Natl Acad Sci U S A. 1996;93:11669–74.
Kumamoto H, Ooya K. Immunohistochemical detection of uPA, uPAR, PAI-1, and maspin in ameloblastic tumors. J Oral Pathol Med. 2000;36(8):488–94.
Sijana HD, Alexander K, Xiaohua L, Margarida B, Yonghong M, Ivory D, et al. Identification of an intrinsic determinant critical for maspin subcellular localization and function. PLoS One. 2013;8(11):e74502.
Acknowledgement
The authors wish to thank Mrs. Verena Hoffmann for all the technical assistance provided during the course of this study.
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Udeabor, S.E., Adisa, A.O., Kolude, B. et al. Expression of CD34 and Maspin in ameloblastoma from a West African subpopulation. Tumor Biol. 35, 7727–7731 (2014). https://doi.org/10.1007/s13277-014-1977-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-014-1977-y