Abstract
Background
Bupivacaine, a common local anesthetic, can cause neurotoxicity and permanent neurological disorders. Crocin has been widely reported as a potential neuroprotective agent in neural injury models.
Objective
The aim of this study was to investigate the role and regulatory mechanism of crocin underlying bupivacaine-induced neurotoxicity.
Method
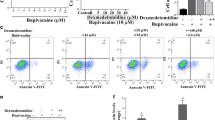
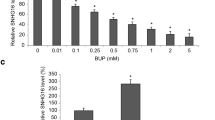
Human neuroblastoma SH-SY5Y cells were treated with bupivacaine and/or crocin for 24 h, followed by detecting cell viability using 3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide assay. The effect of crocin or bupivacaine on SH-SY5Y cell proliferation was measured by Ki67 immunofluorescence assay. The levels of apoptosis-related proteins and the markers in the PI3K/Akt signaling pathway were examined using western blot analysis. The activities of caspase 3, catalase (CAT), superoxide dismutase (SOD), malondialdehyde (MDA) and glutathione peroxidase (GSH-Px) were tested using respective commercial assay kits. Flow cytometry analysis was executed for detecting SH-SY5Y cell apoptosis.
Result
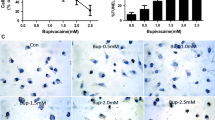
Crocin attenuated bupivacaine-induced neurotoxicity in SH-SY5Y cells. Meanwhile, crocin inhibited SH-SY5Y cell apoptosis induced by bupivacaine via repressing the activity of caspase-3, reducing Bax expression, and elevating Bcl-2 expression. Moreover, crocin mitigated oxidative stress in SH-SY5Y cells by increasing the content of CAT, SOD, GSH-Px and reducing the content of MDA. Additionally, crocin protected against bupivacaine-induced dephosphorylation of Akt and GSK-3β. The protective effects of crocin against bupivacaine-induced neurotoxicity in SH-SY5Y cells were counteracted by the Akt inhibitor.
Conclusion
These results suggested that crocin may exert a neuroprotective function by promoting cell proliferation and suppressing apoptosis and oxidative stress in SH-SY5Y cells. Thus, crocin might become a promising drug for the treatment of bupivacaine-induced neurotoxicity.






Similar content being viewed by others
Data Availability
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
References
Baghishani F, Mohammadipour A, Hosseinzadeh H, Hosseini M, Ebrahimzadeh-Bideskan A (2018) The effects of tramadol administration on hippocampal cell apoptosis, learning and memory in adult rats and neuroprotective effects of crocin. Metab Brain Dis 33:907–916
Baradaran Rahim V, Khammar MT, Rakhshandeh H, Samzadeh-Kermani A, Hosseini A, Askari VR (2019) Crocin protects cardiomyocytes against LPS-Induced inflammation. Pharmacol Rep 71:1228–1234
Becker DE, Reed KL (2012) Local anesthetics: review of pharmacological considerations. Anesth Prog 59:90–101 quiz 102–103
Borgeat A, Aguirre J (2010) Update on local anesthetics. Curr Opin Anaesthesiol 23:466–471
Byram SC, Byram SW, Miller NM, Fargo KN (2017) Bupivacaine increases the rate of motoneuron death following peripheral nerve injury. Restor Neurol Neurosci 35:129–135
Cela O, Piccoli C, Scrima R, Quarato G, Marolla A, Cinnella G, Dambrosio M, Capitanio N (2010) Bupivacaine uncouples the mitochondrial oxidative phosphorylation, inhibits respiratory chain complexes I and III and enhances ROS production: results of a study on cell cultures. Mitochondrion 10:487–496
Chen L, Qi Y, Yang X (2015) Neuroprotective effects of crocin against oxidative stress induced by ischemia/reperfusion injury in rat retina. Ophthalmic Res 54:157–168
Chen X, Chen H, He Y, Fu S, Liu H, Wang Q, Shen J (2020) Proteomics-guided study on Buyang Huanwu Decoction for its neuroprotective and neurogenic mechanisms for transient ischemic stroke: involvements of EGFR/PI3K/Akt/Bad/14-3-3 and Jak2/Stat3/Cyclin D1 signaling cascades. Mol Neurobiol 57:4305–4321
Furuhashi T, Sakamoto K (2017) Regulation of AKT activity prevents autonomic nervous system imbalance. Physiol Behav 168:20–23
Galbes O, Bourret A, Nouette-Gaulain K, Pillard F, Matecki S, Py G, Mercier J, Capdevila X, Philips A (2010) N-acetylcysteine protects against bupivacaine-induced myotoxicity caused by oxidative and sarcoplasmic reticulum stress in human skeletal myotubes. Anesthesiology 113:560–569
Hadipour M, Bahari Z, Afarinesh MR, Jangravi Z, Shirvani H, Meftahi GH (2021) Administering crocin ameliorates anxiety-like behaviours and reduces the inflammatory response in amyloid-beta induced neurotoxicity in rat. Clin Exp Pharmacol Physiol 48:877–889
Hashemzaei M, Rezaee R, Nabatzehi M, Tsarouhas K, Konstantinos Nikolouzakis T, Lazopoulos G, Tsatsakis DAS, Shahraki A J (2020) Anti-hypertensive effect of crocin and hesperidin combination in high-fat diet treated rats. Exp Ther Med 19:3840–3844
Hatziagapiou K, Kakouri E, Lambrou GI, Bethanis K, Tarantilis PA (2019) Antioxidant Properties of Crocus Sativus L. and its constituents and relevance to neurodegenerative Diseases; Focus on Alzheimer’s and Parkinson’s Disease. Curr Neuropharmacol 17:377–402
Heidari S, Mehri S, Hosseinzadeh H (2017) Memory enhancement and protective effects of crocin against D-galactose aging model in the hippocampus of Wistar rats. Iran J Basic Med Sci 20:1250–1259
Ji J, Yan X, Li Z, Lai Z, Liu J (2015) Therapeutic effects of intrathecal versus intravenous monosialoganglioside against bupivacaine-induced spinal neurotoxicity in rats. Biomed Pharmacother 69:311–316
Johnson ME, Saenz JA, DaSilva AD, Uhl CB, Gores GJ (2002) Effect of local anesthetic on neuronal cytoplasmic calcium and plasma membrane lysis (necrosis) in a cell culture model. Anesthesiology 97:1466–1476
Li H, Tang Z, Chu P, Song Y, Yang Y, Sun B, Niu M, Qaed E, Shopit A, Han G et al (2018) Neuroprotective effect of phosphocreatine on oxidative stress and mitochondrial dysfunction induced apoptosis in vitro and in vivo: involvement of dual PI3K/Akt and Nrf2/HO-1 pathways. Free Radic Biol Med 120:228–238
Liu Y, Nan F, Lu K, Wang Y, Liu Y, Wei S, Wu R, Wang Y (2017) Identification of key genes in endometrioid endometrial adenocarcinoma via TCGA database. Cancer Biomark 21:11–21
Lv D, Bai Z, Yang L, Li X, Chen X (2016) Lipid emulsion reverses bupivacaine-induced apoptosis of h9c2 cardiomyocytes: PI3K/Akt/GSK-3β signaling pathway. Environ Toxicol Pharmacol 42:85–91
Ma R, Wang X, Lu C, Li C, Cheng Y, Ding G, Liu L, Ding Z (2010) Dexamethasone attenuated bupivacaine-induced neuron injury in vitro through a threonine-serine protein kinase B-dependent mechanism. Neuroscience 167:329–342
Maurice JM, Gan Y, Ma FX, Chang YC, Hibner M, Huang Y (2010) Bupivacaine causes cytotoxicity in mouse C2C12 myoblast cells: involvement of ERK and akt signaling pathways. Acta Pharmacol Sin 31:493–500
Ochiai T, Ohno S, Soeda S, Tanaka H, Shoyama Y, Shimeno H (2004) Crocin prevents the death of rat pheochromyctoma (PC-12) cells by its antioxidant effects stronger than those of alpha-tocopherol. Neurosci Lett 362:61–64
Pham TH, Page YL, Percevault F, Ferrière F, Flouriot G, Pakdel F (2021) Apigenin, a partial antagonist of the Estrogen receptor (ER), inhibits ER-Positive breast Cancer cell proliferation through Akt/FOXM1 signaling. Int J Mol Sci 22:470
Scholzen T, Gerdes J (2000) The Ki-67 protein: from the known and the unknown. J Cell Physiol 182:311–322
Sekimoto K, Tobe M, Saito S (2017) Local anesthetic toxicity: acute and chronic management. Acute Med Surg 4:152–160
Shafahi M, Vaezi G, Shajiee H, Sharafi S, Khaksari M (2018) Crocin inhibits apoptosis and astrogliosis of Hippocampus neurons against methamphetamine neurotoxicity via antioxidant and anti-inflammatory mechanisms. Neurochem Res 43:2252–2259
Trachootham D, Lu W, Ogasawara MA, Nilsa RD, Huang P (2008) Redox regulation of cell survival. Antioxid Redox Signal 10:1343–1374
Verlinde M, Hollmann MW, Stevens MF, Hermanns H, Werdehausen R, Lirk P (2016) Local Anesthetic-Induced neurotoxicity. Int J Mol Sci 17:339
Wang X, Zhang X, Cheng Y, Li C, Zhang W, Liu L, Ding Z (2010) Alpha-lipoic acid prevents bupivacaine-induced neuron injury in vitro through a PI3K/Akt-dependent mechanism. Neurotoxicology 31:101–112
Wang LY, Li X, Han YZ (2015) Neuroprotection by epigallo catechin gallate against bupivacaine anesthesia induced toxicity involves modulation of PI3/Akt/PTEN signalling in N2a and SH-SY5Y cells. Int J Clin Exp Med 8:15065–15075
Wang JF, Xu HJ, He ZL, Yin Q, Cheng W (2020) Crocin Alleviates Pain Hyperalgesia in AIA Rats by Inhibiting the Spinal Wnt5a/β-Catenin Signaling Pathway and Glial Activation. Neural Plast 2020:4297483
Wang L, Guo W, Guan H, Yan N, Cai X, Zhu L (2021a) Local anesthetic bupivacaine inhibits proliferation and metastasis of hepatocellular carcinoma cells via suppressing PI3K/Akt and MAPK signaling. J Biochem Mol Toxicol 35:e22871
Wang T, Zheng L, Zhang W (2021b) Hesperidin alleviates bupivacaine anesthesia-induced neurotoxicity in SH-SY5Y cells by regulating apoptosis and oxidative damage. J Biochem Mol Toxicol 35:e22787
Wen X, Xu S, Liu H, Zhang Q, Liang H, Yang C, Wang H (2013) Neurotoxicity induced by bupivacaine via T-type calcium channels in SH-SY5Y cells. PLoS ONE 8:e62942
Wright JL, Durieux ME, Groves DS (2008) A brief review of innovative uses for local anesthetics. Curr Opin Anaesthesiol 21:651–656
Yang X, Huo F, Liu B, Liu J, Chen T, Li J, Zhu Z, Lv B (2017) Crocin inhibits oxidative stress and pro-inflammatory response of Microglial cells Associated with Diabetic Retinopathy through the activation of PI3K/Akt signaling pathway. J Mol Neurosci 61:581–589
Yu JS, Cui W (2016) Proliferation, survival and metabolism: the role of PI3K/AKT/mTOR signalling in pluripotency and cell fate determination. Development 143:3050–3060
Yu LX, Zhang BL, Yang MY, Liu H, Xiao CH, Zhang SG, Liu R (2019) MicroRNA-106b-5p promotes hepatocellular carcinoma development via modulating FOG2. Onco Targets Ther 12:5639–5647
Zang M, Hou J, Huang Y, Wang J, Ding X, Zhang B, Wang Y, Xuan Y, Zhou Y (2021) Crocetin suppresses angiogenesis and metastasis through inhibiting sonic hedgehog signaling pathway in gastric cancer. Biochem Biophys Res Commun 576:86–92
Zhang S, Xue R, Geng Y, Wang H, Li W (2020a) Fisetin prevents HT22 cells from high Glucose-Induced Neurotoxicity via PI3K/Akt/CREB signaling pathway. Front Neurosci 14:241
Zhang X, Zhang X, Dang Z, Su S, Li Z, Lu D (2020b) Cognitive Protective Mechanism of Crocin Pretreatment in Rat Submitted to Acute High-Altitude Hypoxia Exposure. Biomed Res Int 2020:3409679
Zhang L, Zhang L, Guo F (2021) MiRNA-494-3p regulates Bupivacaine-Induced neurotoxicity by the CDK6-PI3K/AKT signaling. Neurotox Res 39:2007–2017
Zhao T, Wang Q (2020) Capillarisin protects SH-SY5Y cells against bupivacaine-induced apoptosis via ROS-mediated PI3K/PKB pathway. Life Sci 259:118279
Zhao W, Liu Z, Yu X, Lai L, Li H, Liu Z, Li L, Jiang S, Xia Z, Xu SY (2016) iTRAQ proteomics analysis reveals that PI3K is highly associated with bupivacaine-induced neurotoxicity pathways. Proteomics 16:564–575
Zheng T, Shi Y, Zhang J, Peng J, Zhang X, Chen K, Chen Y, Liu L (2019) MiR-130a exerts neuroprotective effects against ischemic stroke through PTEN/PI3K/AKT pathway. Biomed Pharmacother 117:109117
Acknowledgements
The authors appreciate all the participants providing supports for this study.
Funding
This work was supported by Zhejiang Medical and Health Science and Technology Plan Project (Grant no. 2021KY1002) and Wuhan Medical Research Project and Youth Project (Grant no. WX20Q21).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
None.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, L., Chen, Z., Li, J. et al. Crocin alleviates neurotoxicity induced by bupivacaine in SH-SY5Y cells with inhibition of PI3K/AKT signaling. Genes Genom 46, 149–160 (2024). https://doi.org/10.1007/s13258-023-01431-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13258-023-01431-4