Abstract
Purpose
Recent studies have demonstrated that miRNAs play a vital role in regulating myocardial ischemia/reperfusion injury (MIRI). MiR-217 has been proven to be implicated in cardiac diseases such as chronic heart failure and cardiac myxoma. However, the role of miR-217 in MIRI is not clear.
Methods
A mouse MIRI model was established and the myocardial infarct size was evaluated by TTC staining. The expression level of miR-217 in I/R group was determined by real-time polymerase chain reaction. Subsequently, MIRI mice and H9C2 cells were administrated with miR-217 inhibitor in vivo and in vitro, respectively. The levels of TNF-α and IL-6 were measured by commercially available ELISA kits. Blood and cell samples were collected for the measurement of lactate dehydrogenase (LDH) level and caspase-3 activity. Cell viability was assessed with the CCK-8 assay. We then explored the detailed molecular mechanisms by TargetScan 7.1 database and further studies were performed to prove the prediction by dual-luciferase reporter assay.
Results
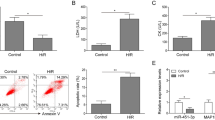
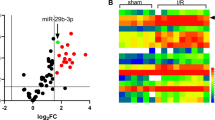
Larger stainless infarct areas were observed in the MIRI group, accompanied by inceased serum LDH activity, indicating the mouse MIRI model was successfully established. MiR-217 was up-regulated in MIRI mice and hypoxia/reoxygenation-treated H9C2 cells. MiR-217 knockdown alleviated the MIRI in MIRI mouse model, and also attenuated the myocardial hypoxia/reoxygenation injury in H9C2 cells. Moreover, dual specificity protein phosphatase 14 (DUSP14) was proved to be a target of miR-217. Besides, further study indicated that inhibition of miR-217 protected against MIRI through inactivating NF-κB and MAPK pathways via targeting DUSP14.
Conclusions
MiR-217 inhibition protected against MIRI through inactivating NF-κB and MAPK pathways by targeting DUSP14. This study may provide valuable diagnostic and factors and therapeutic agents for MIRI.






Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article.
References
Adameova, A. D., S. K. Bhullar, V. Elimban, and N. S. Dhalla. Activation of β 1-adrenoceptors may not be involved in arrhythmogenesis in ischemic heart disease. Rev. Cardiovasc. Med. 19(3):97–101, 2018.
Bulluck, H., D. M. Yellon, and D. J. Hausenloy. Reducing myocardial infarct size: challenges and future opportunities. Heart. 102(5):341–348, 2016. https://doi.org/10.1136/heartjnl-2015-307855.
Chen, C., D. A. Ridzon, A. J. Broomer, Z. Zhou, D. H. Lee, J. T. Nguyen, et al. Real-time quantification of microRNAs by stem-loop RT-PCR. Nucleic Acids Res. 33(20):e179, 2005. https://doi.org/10.1093/nar/gni178.
Chen, Z., D. Wu, L. Li, and L. Chen. Apelin/APJ system: a novel therapeutic target for myocardial ischemia/reperfusion injury. DNA Cell Biol. 35(12):766–775, 2016. https://doi.org/10.1089/dna.2016.3391.
de Lucia, C., K. Komici, G. Borghetti, G. D. Femminella, L. Bencivenga, A. Cannavo, et al. microRNA in cardiovascular aging and age-related cardiovascular diseases. Front. Med. 4:74, 2017. https://doi.org/10.3389/fmed.2017.00074.
Fan, Z. X., and J. Yang. The role of microRNAs in regulating myocardial ischemia reperfusion injury. Saudi Med. J. 36(7):787–793, 2015. https://doi.org/10.15537/smj.2015.7.11089.
Frank, A., M. Bonney, S. Bonney, L. Weitzel, M. Koeppen, and T. Eckle. Myocardial ischemia reperfusion injury: from basic science to clinical bedside. Semin. Cardiothorac. Vasc. Anesth. 16(3):123–132, 2012. https://doi.org/10.1177/1089253211436350.
Guo, J., Z. Feng, Z. Huang, H. Wang, and W. Lu. MicroRNA-217 functions as a tumour suppressor gene and correlates with cell resistance to cisplatin in lung cancer. Mol. Cells. 37(9):664–671, 2014. https://doi.org/10.14348/molcells.2014.0121.
Hausenloy, D. J., and D. M. Yellon. Time to take myocardial reperfusion injury seriously. N. Engl. J. Med. 359(5):518–520, 2008. https://doi.org/10.1056/NEJMe0803746.
Hausenloy, D. J., and D. M. Yellon. Ischaemic conditioning and reperfusion injury. Nat. Rev. Cardiol. 13(4):193–209, 2016. https://doi.org/10.1038/nrcardio.2016.5.
Hwang, H. W., and J. T. Mendell. MicroRNAs in cell proliferation, cell death, and tumorigenesis. Br. J. Cancer. 96(Suppl):R40–R44, 2007.
Klinger, S., C. Poussin, M. B. Debril, W. Dolci, P. A. Halban, and B. Thorens. Increasing GLP-1-induced beta-cell proliferation by silencing the negative regulators of signaling cAMP response element modulator-alpha and DUSP14. Diabetes 57(3):584–593, 2008. https://doi.org/10.2337/db07-1414.
Lin, B., J. Xu, D. G. Feng, F. Wang, J. X. Wang, and H. Zhao. DUSP14 knockout accelerates cardiac ischemia reperfusion (IR) injury through activating NF-kappaB and MAPKs signaling pathways modulated by ROS generation. Biochem. Biophys. Res. Commun. 501(1):24–32, 2018. https://doi.org/10.1016/j.bbrc.2018.04.101.
Liu, N. B., M. Wu, C. Chen, M. Fujino, and J. S. Huang. Novel molecular targets participating in myocardial ischemia-reperfusion injury and cardioprotection. Cardiol. Res. Pract. 2019:6935147, 2019. https://doi.org/10.1155/2019/6935147.
Marti, F., A. Krause, N. H. Post, C. Lyddane, B. Dupont, M. Sadelain, et al. Negative-feedback regulation of CD28 costimulation by a novel mitogen-activated protein kinase phosphatase, MKP6. J. Immunol. 166(1):197–206, 2001. https://doi.org/10.4049/jimmunol.166.1.197.
Mens, M. M. J., and M. Ghanbari. Cell cycle regulation of stem cells by MicroRNAs. Stem Cell Rev. Rep. 14(3):309–322, 2018. https://doi.org/10.1007/s12015-018-9808-y.
National Research Council Institute for Laboratory Animal R. Guide for the Care and Use of Laboratory Animals. Washington, DC: National Academies Press, 1996.
Nie, X., J. Fan, H. Li, Z. Yin, Y. Zhao, B. Dai, et al. miR-217 promotes cardiac hypertrophy and dysfunction by targeting PTEN. Mol. Ther. Nucleic Acids. 12:254–266, 2018. https://doi.org/10.1016/j.omtn.2018.05.013.
Oerlemans, M. I., A. van Mil, J. Liu, E. van Eeuwijk, K. D. Ouden, P. A. Doevendans, et al. Inhibition of miR-223 reduces inflammation but not adverse cardiac remodelling after myocardial ischemia-reperfusion in vivo. Non-coding RNA Investig. 2(15), 2018.
Ong, S. G., W. H. Lee, L. Theodorou, K. Kodo, S. Y. Lim, D. H. Shukla, et al. HIF-1 reduces ischaemia-reperfusion injury in the heart by targeting the mitochondrial permeability transition pore. Cardiovasc. Res. 104(1):24–36, 2014. https://doi.org/10.1093/cvr/cvu172.
Pan, Z., X. Sun, J. Ren, X. Li, X. Gao, C. Lu, et al. miR-1 exacerbates cardiac ischemia-reperfusion injury in mouse models. PLoS ONE. 7(11):e50515, 2012. https://doi.org/10.1371/journal.pone.0050515.
Pan, B., J. Yang, X. Wang, K. Xu, and T. Ikezoe. miR-217 sensitizes chronic myelogenous leukemia cells to tyrosine kinase inhibitors by targeting pro-oncogenic anterior gradient 2. Exp. Hematol. 68(80–8):e2, 2018. https://doi.org/10.1016/j.exphem.2018.09.001.
Wang, L., H. Huang, Y. Fan, B. Kong, H. Hu, K. Hu, et al. Effects of downregulation of microRNA-181a on H2O2-induced H9c2 cell apoptosis via the mitochondrial apoptotic pathway. Oxid. Med. Cell. longev. 2014:960362, 2014. https://doi.org/10.1155/2014/960362.
Wang, X., W. Mao, C. Fang, S. Tian, X. Zhu, L. Yang, et al. Dusp14 protects against hepatic ischaemia-reperfusion injury via Tak1 suppression. J. Hepatol. 2017. https://doi.org/10.1016/j.jhep.2017.08.032.
Wang, X., X. Zhang, X. P. Ren, J. Chen, H. Liu, J. Yang, et al. MicroRNA-494 targeting both proapoptotic and antiapoptotic proteins protects against ischemia/reperfusion-induced cardiac injury. Circulation. 122(13):1308–1318, 2010. https://doi.org/10.1161/circulationaha.110.964684.
Wei, R., R. Zhang, Y. Xie, L. Shen, and F. Chen. Hydrogen suppresses hypoxia/reoxygenation-induced cell death in hippocampal neurons through reducing oxidative stress. Cell. Physiol. Biochem. 36(2):585–598, 2015. https://doi.org/10.1159/000430122.
Yang, C. Y., L. L. Chiu, C. C. Chang, H. C. Chuang, and T. H. Tan. Induction of DUSP14 ubiquitination by PRMT5-mediated arginine methylation. FASEB J. 2018:fj201800244RR, 2018. https://doi.org/10.1096/fj.201800244rr.
Yang, C. Y., J. P. Li, L. L. Chiu, J. L. Lan, D. Y. Chen, H. C. Chuang, et al. Dual-specificity phosphatase 14 (DUSP14/MKP6) negatively regulates TCR signaling by inhibiting TAB 1 activation. J. Immunol. 192(4):1547–1557, 2014. https://doi.org/10.4049/jimmunol.1300989.
Yellon, D. M., and D. J. Hausenloy. Myocardial reperfusion injury. N. Engl. J. Med. 357(11):1121–1135, 2007. https://doi.org/10.1056/NEJMra071667.
Zhang, J., C. Wang, and H. Xu. miR-217 suppresses proliferation and promotes apoptosis in cardiac myxoma by targeting Interleukin-6. Biochem. Biophys. Res. Commun. 490(3):713–718, 2017. https://doi.org/10.1016/j.bbrc.2017.06.106.
Zhang, H. S., T. C. Wu, W. W. Sang, and Z. Ruan. MiR-217 is involved in Tat-induced HIV-1 long terminal repeat (LTR) transactivation by down-regulation of SIRT1. Biochimica et Biophysica Acta 1823(5):1017–1023, 2012. https://doi.org/10.1016/j.bbamcr.2012.02.014.
Zhang, Q., Y. Yuan, J. Cui, T. Xiao, and D. Jiang. MiR-217 promotes tumor proliferation in breast cancer via targeting DACH1. J. Cancer 6(2):184–191, 2015. https://doi.org/10.7150/jca.10822.
Zhao, W. G., S. N. Yu, Z. H. Lu, Y. H. Ma, Y. M. Gu, and J. Chen. The miR-217 microRNA functions as a potential tumor suppressor in pancreatic ductal adenocarcinoma by targeting KRAS. Carcinogenesis. 31(10):1726–1733, 2010. https://doi.org/10.1093/carcin/bgq160.
Zheng, H., Q. Li, R. Chen, J. Zhang, Y. Ran, X. He, et al. The dual-specificity phosphatase DUSP14 negatively regulates tumor necrosis factor- and interleukin-1-induced nuclear factor-kappaB activation by dephosphorylating the protein kinase TAK1. J. Biol. Chem. 288(2):819–825, 2013. https://doi.org/10.1074/jbc.M112.412643.
Zhou, T., D. K. Xiang, S. N. Li, L. H. Yang, L. F. Gao, and C. Feng. MicroRNA-495 ameliorates cardiac microvascular endothelial cell injury and inflammatory reaction by suppressing the NLRP3 inflammasome signaling pathway. Cell. Physiol. Biochem. 49(2):798–815, 2018. https://doi.org/10.1159/000493042.
Conflict of interest
The authors declare that they have no competing interests, and all authors should confirm its accuracy.
Ethics Approval
The animal use protocol listed below has been reviewed and approved by the Animal Ethical and Welfare Committee.
Author information
Authors and Affiliations
Contributions
YFL conceived and designed the experiments, LPF analyzed and interpreted the results of the experiments, JLW and QYN performed the experiments.
Corresponding author
Additional information
Associate Editor Hanjoong Jo oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, Y., Fei, L., Wang, J. et al. Inhibition of miR-217 Protects Against Myocardial Ischemia–Reperfusion Injury Through Inactivating NF-κB and MAPK Pathways. Cardiovasc Eng Tech 11, 219–227 (2020). https://doi.org/10.1007/s13239-019-00452-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13239-019-00452-z