Abstract
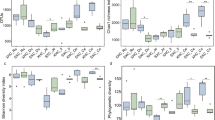
The Tibetan swine (TIS) is a non-ruminant herbivore with high disease resistance. Also, it has the ability to digest plants with high fiber content. However, it is not known whether any relationship exist between these characteristics of the TIS and its cecal microbiota. Thus, this study aims to investigate the cecal microbiota of the adult TIS using high-throughput sequencing techniques in order to explore possible relationships between these unique characteristics of the TIS (high disease resistance and ability to digest high fiber plants) and its cecal microbiota. PIC pigs (lean type) were chosen as controls. The results show that 75,069 valid sequences of the 16S rRNA gene at V4-V5 region were obtained in the cecal content of TIS. They were composed of 15 phyla, 70 genera and divided into 660 Operational Taxonomic Units (OTUs). Bacteroidetes and Firmicutes were the predominant phyla in both breeds, but TIS had more Bacteroidetes than Firmicutes. Also, 42.4% of the cecal bacteria were found to be unclassified and uncultured. Many cellulolytic bacteria were also found in the two breeds. TIS (88.10%) had much higher abundance in the core bacterial communities than PIC pigs (81.29%), and the proportion of Bacteroides and Spirochaetes that can effectively degrade cellulose were 6.01 and 6.40% higher than PIC pigs, respectively, while Proteobacteria that are closely related to gastrointestinal diseases were 1.61% lower than PIC pigs. Thus, the disease resistance of the TIS and its ability to digest plants with high fiber content may be related to high abundance of core bacterial communities as well as the large number of unknown and unclassified bacteria.







Similar content being viewed by others
References
Bian G, Ma L, Su Y, Zhu W (2013) The microbial community in the feces of the white rhinoceros (Ceratotherium simum) as determined by barcoded pyrosequencing analysis. PLoS One 8:e70103. https://doi.org/10.1371/journal.pone.0070103
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK, Fierer N, Pena AG, Goodrich JK, Gordon JI, Huttley GA, Kelley ST, Knights D, Koenig JE, Ley RE, Lozupone CA, McDonald D, Muegge BD, Pirrung M, Reeder J, Sevinsky JR, Tumbaugh PJ, Walters WA, Widmann J, Yatsunenko T, Zaneveld J, Knight R (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7:335–336. https://doi.org/10.1038/Nmeth.F.303
Castillo M, Martin-Orue SM, Anguita M, Perez JF, Gasa J (2007) Adaptation of gut microbiota to corn physical structure and different types of dietary fibre. Livest Sci 109:149–152. https://doi.org/10.1016/j.livsci.2007.01.129
Chakravorty S, Helb D, Burday M, Connell N, Alland D (2007) A detailed analysis of 16S ribosomal RNA gene segments for the diagnosis of pathogenic bacteria. J Microbiol Methods 69:330–339. https://doi.org/10.1016/j.mimet.2007.02.005
Chen H, Mao XB, Che LQ, Yu B, He J, Yu J, Han GQ, Huang ZQ, Zheng P, Chen DW (2014) Impact of fiber types on gut microbiota, gut environment and gut function in fattening pigs. Anim Feed Sci Technol 195:101–111. https://doi.org/10.1016/j.anifeedsci.2014.06.002
Edgar RC (2013) UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nat Methods 10:996. https://doi.org/10.1038/nmeth.2604
Edgar RC, Haas BJ, Clemente JC, Quince C, Knight R (2011) UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 27:2194–2200. https://doi.org/10.1093/bioinformatics/btr381
Edrington TS, Dowd SE, Farrow RF, Hagevoort GR, Callaway TR, Anderson RC, Nisbet DJ (2012) Development of colonic microflora as assessed by pyrosequencing in dairy calves fed waste milk. J Dairy Sci 95:4519–4525. https://doi.org/10.3168/jds.2011-5119
Jami E, Mizrahi I (2012) Composition and similarity of bovine rumen microbiota across individual animals. PLoS One 7:e33306. https://doi.org/10.1371/journal.pone.0033306
Kim HB, Borewicz K, White BA, Singer RS, Sreevatsan S, Tu ZJ, Isaacson RE (2012) Microbial shifts in the swine distal gut in response to the treatment with antimicrobial growth promoter, tylosin. Proc Natl Acad Sci U S A 109:15485–15490. https://doi.org/10.1073/pnas.1205147109
Kohl KD, Miller AW, Marvin JE, Mackie R, Dearing MD (2014) Herbivorous rodents (Neotoma spp.) harbour abundant and active foregut microbiota. Environ Microbiol 9:2869–2878. https://doi.org/10.1111/1462-2920.12376
Leser TD, Amenuvor JZ, Jensen TK, Lindecrona RH, Boye M, Moller K (2002) Culture-independent analysis of gut bacteria: the pig gastrointestinal tract microbiota revisited. Appl Environ Microbiol 68:673–690. https://doi.org/10.1128/aem.68.2.673-690.2002
Li RW, Wu ST, Baldwin RL, Li WZ, Li CJ (2012) Perturbation dynamics of the rumen microbiota in response to exogenous butyrate. PLoS One 7. https://doi.org/10.1371/journal.pone.0029392
Liu H, Ivarsson E, Dicksved J, Lundh T, Lindberg JE (2012) Inclusion of chicory (Cichorium intybus L.) in pigs’ diets affects the intestinal microenvironment and the gut microbiota. Appl Environ Microbiol 78:4102–4109. https://doi.org/10.1128/AEM.07702-11
Liu J, Wang JK, Zhu W, Pu YY, Guan LL, Liu JX (2014) Monitoring the rumen pectinolytic bacteria Treponema saccharophilum using real-time PCR. FEMS Microbiol Ecol 87:576–585. https://doi.org/10.1111/1574-6941.12246
Looft T, Johnson TA, Allen HK, Bayles DO, Alt DP, Stedtfeld RD, Sul WJ, Stedtfeld TM, Chai B, Cole JR, Hashsham SA, Tiedje JM, Stanton TB (2012) In-feed antibiotic effects on the swine intestinal microbiome. Proc Natl Acad Sci U S A 109:1691–1696. https://doi.org/10.1073/pnas.1120238109
Looft T, Allen HK, Cantarel BL, Levine UY, Bayles DO, Alt DP, Henrissat B, Stanton TB (2014) Bacteria, phages and pigs: the effects of in-feed antibiotics on the microbiome at different gut locations. ISME J 8:1566–1576. https://doi.org/10.1038/ismej.2014.12
Ma L, Yang WP, Meng FX, Ji SY, Xin HY, Cao BY (2015) Characterization of an acidic cellulase produced by Bacillus subtilis BY-4 isolated from gastrointestinal tract of Tibetan pig. J Taiwan Inst Chem Eng 56:67–72. https://doi.org/10.1016/j.jtice.2015.04.025
Meng FX, Ma L, Ji SY, Yang WP, Cao BY (2014) Isolation and characterization of Bacillus subtilis strain BY-3, a thermophilic and efficient cellulase-producing bacterium on untreated plant biomass. Lett Appl Microbiol 59:306–312. https://doi.org/10.1111/lam.12276
Niu Q, Li PH, Hao SS, Zhang YQ, Kim SW, Li HZ, Ma X, Gao S, He LC, Wu WJ, Huang XG, Hua JD, Zhou B, Huang RH (2015) Dynamic distribution of the gut microbiota and the relationship with apparent crude fiber digestibility and growth stages in pigs. Sci Rep 5. https://doi.org/10.1038/Srep09938
Pajarillo EAB, Chae JP, Balolong MP, Kim HB, Seo KS, Kang DK (2014) Pyrosequencing-based analysis of fecal microbial communities in three purebred pig lines. J Microbiol 52:646–651. https://doi.org/10.1007/s12275-014-4270-2
Pitta DW, Kumar S, Veiccharelli B, Parmar N, Reddy B, Joshi CG (2014) Bacterial diversity associated with feeding dry forage at different dietary concentrations in the rumen contents of Mehshana buffalo (Bubalus bubalis) using 16S pyrotags. Anaerobe 25:31–41. https://doi.org/10.1016/j.anaerobe.2013.11.008
Shinkai T, Ueki T, Kobayashi Y (2010) Detection and identification of rumen bacteria constituting a fibrolytic consortium dominated by Fibrobacter succinogenes. Anim Sci J 81:72–79. https://doi.org/10.1111/j.1740-0929.2009.00698
Sun DL, Jiang X, Wu QLL, Zhou NY (2013) Intragenomic heterogeneity of 16S rRNA genes causes overestimation of prokaryotic diversity. Appl Environ Microbiol 79:5962–5969. https://doi.org/10.1128/aem.01282-13
Turnbaugh PJ, Hamady M, Yatsunenko T, Cantarel BL, Duncan A, Ley RE, Sogin ML, Jones WJ, Roe BA, Affourtit JP, Egholm M, Henrissat B, Heath AC, Knight R, Gordon JI (2009) A core gut microbiome in obese and lean twins. Nature 457:480–484. https://doi.org/10.1038/nature07540
Walujkar SA, Dhotre DP, Marathe NP, Lawate PS, Bharadwaj RS, Shouche YS (2014) Characterization of bacterial community shift in human ulcerative colitis patients revealed by Illumina based 16S rRNA gene amplicon sequencing. Gut Pathogens 6. https://doi.org/10.1186/1757-4749-6-22
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 73:5261–5267. https://doi.org/10.1128/aem.00062-07
Wenk C (2001) The role of dietary fibre in the digestive physiology of the pig. Anim Feed Sci Technol 90:21–33. https://doi.org/10.1016/s0377-8401(01)00194-8
Wu SG, Wang GT, Angert ER, Wang WW, Li WX, Zou H (2012) Composition, diversity, and origin of the bacterial community in grass carp intestine. PLoS One 7. https://doi.org/10.1371/journal.pone.0030440
Xiao Y, Yan HL, Diao H, Yu B, He J, Yu J, Zheng P, Mao XB, Luo YH, Chen DW (2017) Early gut microbiota intervention suppresses DSS-induced inflammatory responses by deactivating TLR/NLR signalling in pigs. Sci Rep 7:3224. https://doi.org/10.1038/s41598-017-03161-6
Xin H, Ji SY, Peng JY, Han P, An XP, Wang S, Cao BY (2017) Isolation and characterisation of a novel antibacterial peptide from a native swine intestinal tract-derived bacterium. Int J Antimicrob Agents 49:427–436. https://doi.org/10.1016/j.ijantimicag.2016.12.012
Yan H, Potu R, Lu H, de Almeida VV, Stewart T, Ragland D, Armstrong A, Adeola O, Nakatsu CH, Ajuwon KM (2013) Dietary fat content and fiber type modulate hind gut microbial community and metabolic markers in the pig. PLoS One 8. https://doi.org/10.1371/journal.pone.0059581
Yang WP, Meng FX, Peng JY, Han P, Fang F, Ma L, Cao BY (2014) Isolation and identification of a cellulolytic bacterium from the Tibetan pig’s intestine and investigation of its cellulase production. Electron J Biotechnol 17:262–267. https://doi.org/10.1016/j.ejbt.2014.08.002
Zhang LL, Mu CL, He XY, Su Y, Mao SY, Zhang J, Smidt H, Zhu WY (2016) Effects of dietary fibre source on microbiota composition in the large intestine of suckling piglets. FEMS Microbiol Lett 363. https://doi.org/10.1093/femsle/fnw138
Zhu L, Wu Q, Dai J, Zhang S, Wei F (2011) Evidence of cellulose metabolism by the giant panda gut microbiome. Proc Natl Acad Sci U S A 108:17714–17719. https://doi.org/10.1073/pnas.1017956108
Acknowledgments
We thank Hongzhou Wang (Tibetan swine Slaughterhouse of Shaanxi Huayi Industrial Co., Ltd.) and Junfang Yan (Shaanxi Benxiang Pig Slaughterhouse) for their help in sampling. We are also grateful to Juan Qin (Shanghai Majorbio Bio-Pharm Technology Co., Ltd.) for technical help.
Funding
This study was sponsored by grants from the Key Agricultural Science and Technology Promotion Project in Shaanxi province, China (ZDKJ-2014-27) and the Annual National Undergraduate Innovative Entrepreneurial Training Program of China (201410712008), and Science and Technology Plan Project in Henan Province (162102310475).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None declare
Additional information
Highlights
• Comparing the cecal microbiota of a Tibetan and PIC (lean-type) pig.
• The core bacterial communities were different in these two breeds.
• Tibetan swine had a higher proportion of digestion related bacteria phyla.
Weiping Yang and Haiyun Xin are joint first authors.
Rights and permissions
About this article
Cite this article
Yang, W., Xin, H., Cao, F. et al. The significance of the diversity and composition of the cecal microbiota of the Tibetan swine. Ann Microbiol 68, 185–194 (2018). https://doi.org/10.1007/s13213-018-1329-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13213-018-1329-z